Do all gases obey Boyles Law
Our characterization of these results has been that all gases obey the same equations—Boyle’s law, Charles’ law, and the ideal gas equation—and do so exactly. … At very high gas densities, the classical gas laws can be very poor approximations.
Does Boyle's law apply to liquids and gases?
In contrast to gases, liquids are not compressible as their particles are already very close together. Boyle’s law only applies to gases.
Are gas law only applicable to gases?
The gas laws apply only to ideal gases. Ideal gases are gases whose behaviour is interpretable. … The gas particles travel randomly.
Why Boyle's law is applicable only for ideal gas?
Boyle’s law is applicable for an. a) Adiabatic process. … Hint: Boyle’s law states that the volume of an ideal gas is inversely proportional to the pressure in the given volume. This is only possible if no gas escapes the container in which it is kept and the temperature of the container is held constant.Which gas does not obey Boyle's Law?
Some gases such as NO2 and NF2 do not obey Boyles Law at any pressure.
How does Boyle's law apply to space?
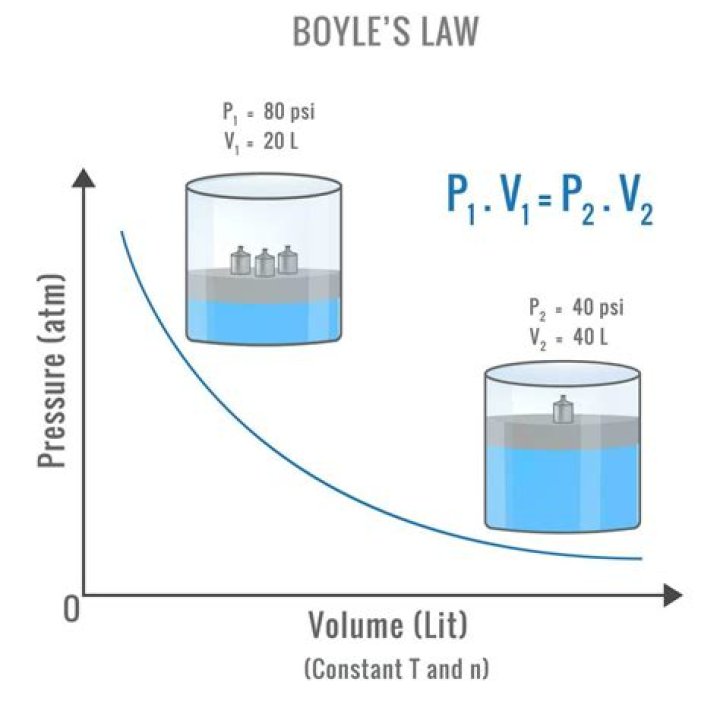
Boyle in 1662, states that at a fixed temperature, the volume of gas is inversely proportional to the pressure exerted by the gas. In other words, when a gas is pumped into an enclosed space, it will shrink to fit into that space, but the pressure that gas puts on the container will increase.
Do all gases obey Charles Law?
Our characterization of these results has been that all gases obey the same equations—Boyle’s law, Charles’ law, and the ideal gas equation—and do so exactly.
Does ideal gas law apply to liquids?
The Ideal Gas Law cannot be applied to liquids. … But we know that a liquid has a constant volume, so the Ideal Gas Law cannot apply to a liquid. The Ideal Gas Law doesn’t even apply to “real” gases like hydrogen, oxygen, and nitrogen.How does Boyle's law describe the relationship between gas pressure and volume?
Boyle’s law—named for Robert Boyle—states that, at constant temperature, the pressure P of a gas varies inversely with its volume V, or PV = k, where k is a constant. … Charles (1746–1823)—states that, at constant pressure, the volume V of a gas is directly proportional to its absolute (Kelvin) temperature T, or V/T = k.
Why does Boyle's law work for a gas but not for a liquid?Boyle’s law states that pressure, , is inversely proportional to the volume, , of a gas if the temperature and the amount of gas remain unchanged in a closed system . One way is by changing the amount of gas, such as in the human lung, which is not a closed system, hence not subject to Boyle’s law.
Article first time published onUnder what condition Boyle's law is applicable?
Boyle’s law is applicable for an isothermal process, wherein temperature of gas remains constant.
Can you compare two different gases in Boyle's law?
Explanation: Boyle’s law states that at constant temperature the given volume of an ideal gas is inversely proportional to the mass of the gas present in that particular container. So in order to compare two different gases we can use Boyle’s law.
Which gas law is a combination of all gas laws?
The combined gas law combines the three gas laws: Boyle’s Law, Charles’ Law, and Gay-Lussac’s Law. It states that the ratio of the product of pressure and volume and the absolute temperature of a gas is equal to a constant. When Avogadro’s law is added to the combined gas law, the ideal gas law results.
What is the difference between Boyle's Law and Charles Law?
In Charles law, temperature and volume of the gas are kept at constant pressure. Whereas in Boyle’s law, pressure and volume of the gas are kept at a constant temperature. In Boyle’s law, pressure and volume vary inversely whereas, in Charles law, pressure and volume vary directly.
How are Boyle's law and the ideal gas law similar?
Boyle’s law—named for Robert Boyle—states that, at constant temperature, the pressure P of a gas varies inversely with its volume V, or PV = k, where k is a constant. … Though this law describes the behaviour of an ideal gas, it closely approximates the behaviour of real gases.
Does Air obey Boyle's Law?
We can breathe air in and out of our lungs because of Boyle’s law. According to Boyle’s law, if a given amount of gas has a constant temperature, increasing its volume decreases its pressure, and vice-versa.
Is Boyle's Law inverse or direct?
Boyle’s Law describes the inverse relationship between the pressure and volume of a fixed amount of gas at a constant temperature.
At which condition all gases obey Charles Law?
At low pressure and high temperature, real gases behave as ideal because at low pressure the volume of the molecules tend to become negligible as compared to the total volume of the gas and at high temperature the intermolecular forces between molecules become less significant therefore, satisfying the postulates of …
Does blowing up a balloon agree with Boyle's law?
No. Boyle’s law simply states that volume and pressure are inversely proportional.
What gas law is applied in hot air balloon?
Charles’s Law says that the volume of a gas is directly related to the temperature of that gas, similarly when a gas is heated, like a burner in a hot air balloon, the gas expands. So when the air inside the balloon expands, it becomes less dense and provides the lift for the hot air balloon.
What are the properties of gases involved in Boyle's law?
Explanation: Boyle’s law states that, At a constant temperature and the number of moles of a gas, the pressure is inversely proportional to the volume of a gas.
When volume on a gas goes up what happens to its pressure?
Each time they collide with the walls they exert a force on them. More collisions mean more force, so the pressure will increase. When the volume decreases, the pressure increases. This shows that the pressure of a gas is inversely proportional to its volume.
What is an expression of Boyle's Law K constant?
Simply put, Boyle’s states that for a gas at constant temperature, pressure multiplied by volume is a constant value. The equation for this is PV = k, where k is a constant. At a constant temperature, if you increase the pressure of a gas, its volume decreases. If you increase its volume, the pressure decreases.
Are all gases ideal gases?
Ideal gas molecules themselves take up no volume. There are no gases that are exactly ideal, but there are plenty of gases that are close enough that the concept of an ideal gas is an extremely useful approximation for many situations.
Is Boyle's Law valid at very high temperatures?
Boyle’s Laws is applicable to limited high temperatures as Boyle’s law states the inverse relationship between pressure and volume of a gas when the temperature is held constant.
Are all the equations of state known for all substances?
There is no single equation of state that predicts the behavior of all substances under all conditions.
Does adiabatic process obey Boyle's Law?
Answer: An ideal gas undergoes an adiabatic process obeying the relation PV^4/3 = constant. If its initial temperature is.
Which gas law is valid for isobaric process?
An isobaric process is a thermodynamic process in which pressure stays constant: ΔP = 0. For an ideal gas, this means the volume of a gas is proportional to its temperature (historically, this is called Charles’ law ).
Which two variables must be held constant for Boyle's law to apply?
Boyle’s Law holds true only if the number of molecules (n) and the temperature (T) are both constant. Boyle’s Law is used to predict the result of introducing a change in volume and pressure only, and only to the initial state of a fixed quantity of gas.
How can Boyle's law be derived from the combined gas law?
When we put Boyle’s law, Charles’ law, and Gay-Lussac’s law together, we come up with the combined gas law, which shows that: Pressure is inversely proportional to volume, or higher volume equals lower pressure. … Volume is directly proportional to temperature, or higher temperature equals higher volume.
Which shows combined relationship of Boyle's Law and Charles Law?
The combined gas law is an amalgamation of the three previously known laws which are- Boyle’s law PV = K, Charles law V/T = K, and Gay-Lussac’s law P/T = K. Therefore, the formula of combined gas law is PV/T = K, Where P = pressure, T = temperature, V = volume, K is constant.