Does density change with mass
The density of an object can change if either the mass or volume of the object is changed. … If an object is more dense than water, it will sink; if it is less dense than water, it will float.
Does mass depend on density?
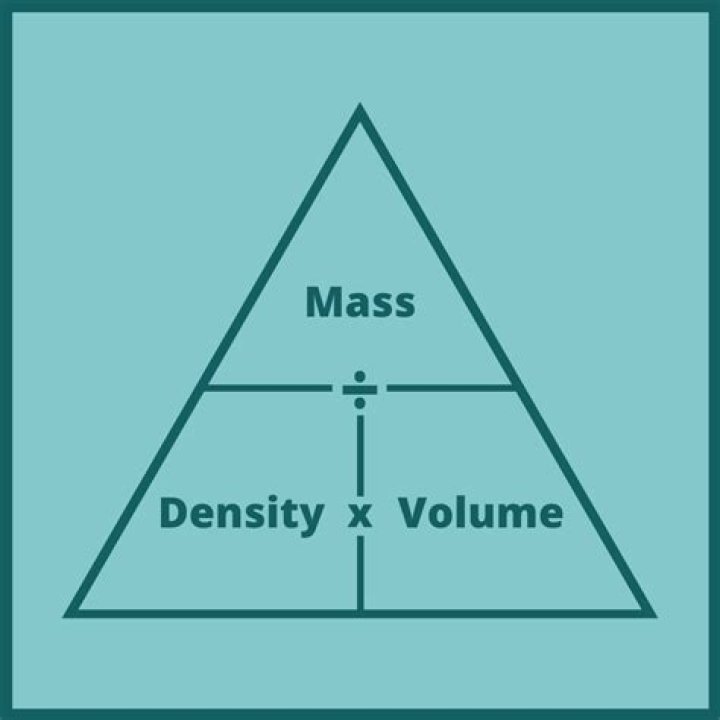
Density (d) depends upon the mass (m) and volume (v) of a substance. The mass depends upon the amount of matter in a substance, whereas volume is the amount of space that a substance takes up. Mathematically, density is equal to a ratio where d = m/v.
Why does density increase as mass increases?
We know that density is given by mass divided by volume. According to this formula, the density is directly proportional to the mass when the volume is constant and inversely proportional to the volume when mass is constant. … Thus, at constant volume, the mass of a substance increases when the density increases.
Does higher density mean higher mass?
Density is a derived unit from two other units: mass and volume. Density is by definition, the amount of mass per unit volume. … Density tells scientists how “heavy” a substance is. If a substance has a higher density, it is heavier.How is mass affected by density and volume?
Mass, volume and density are three of an object’s most basic properties. Mass is how heavy something is, volume tells you how big it is, and density is mass divided by volume.
Why density does not depend on mass and volume?
Unlike mass and volume, increasing the amount of material measured does not increase or decrease density. … However, since volume deviates with changes in temperature and pressure, density can also change with temperature and pressure.
Is density and mass density same?
Mathematically, density is defined as mass divided by volume. Mass density, usually just called “density”, is mass (the amount of matter) per volume, while weight density is the amount of gravity exerted per volume.
What is the relationship between density and mass?
Formula. The mathematical relationship between mass and density is often given by the formula: Density=Mass/Volume. This can be rewritten Mass=Density x Volume.Does higher density mean more atoms?
A substance with smaller more massive atoms that are close together is going to have a higher density. How might these factors work together to cause a substance to have a low density? A substance with larger, lighter atoms that are farther apart is going to have a lower density.
What happens to density if mass and volume decrease?If the mass of the object stays the same but the volume of the object decreases then its density becomes greater. … If the volume of the object stays the same but the mass of the object increases then its density becomes greater.
Article first time published onWhat happens increase density?
When density increases, volume decreases. When volume increases, density decreases.
How do you find mass with only density?
The formula for density is the mass of an object divided by its volume. In equation form, that’s d = m/v , where d is the density, m is the mass and v is the volume of the object.
Does density increase as volume increases?
Density decreases when volume increases if mass is kept constant. Density is defined as mass divided by volume.
Does density change with gravity?
NO. Gravity depends on how many atoms are in a substance, ie , MASS. Now you can increase the density of an object by shrinking it down, but it will still have the same pull .
How does density affect atomic mass?
Explanation: Atoms with a lower atomic mass have a greater density than the atom with the higher atomic weight because the atoms are tightly packed together. The volume of a material can change with temperature and pressure.
Is mass more dense than volume?
Density = Mass/Volume also means that the larger the volume of an object compared to its mass, the less dense it is. For example, if a marble weighs the same as a much larger foam ball, the marble has a higher density than the foam ball, despite its smaller size.
What affects the atomic mass?
The number of neutrons in a nucleus affects the mass of the atom but not its chemical properties. It is usual to characterize different isotopes by giving the sum of the number of protons and neutrons in the nucleus—a quantity called the atomic mass number. …
Is density a mass?
density, mass of a unit volume of a material substance. The formula for density is d = M/V, where d is density, M is mass, and V is volume. Density is commonly expressed in units of grams per cubic centimetre. … For example, the density of air is 1.2 kilograms per cubic metre.
How are density and mass different?
The term mass is used to mean the amount of matter contained in an object. Density alludes to the closeness of the atoms, in substance, i.e. how tightly atoms are packed. Mass is the measure of the amount of inertia. Conversely, density is the degree of compactness.
How is density different from weight and mass?
Density has the components of mass and volume, while weight is concerned with mass and gravity. 2. Division is used to find an object’s density by dividing the mass over volume, while weight is the product of mass and gravity (by virtue of multiplication). … The units to express density and weight also differ.
Does density decrease if mass decreases?
usual density increases with increase in mass and decrease with increase in volume. so density is directly proportional to mass and inverse proportion to volume.
What happens to density if you increase the mass but keep the volume the same?
Density is defined as mass per unit volume. So keeping volume constant we can say density is directly proporsional to mass, and increasing mass will definitely increase density. As density=mass/volume,density is directly proportional to mass and inversely proportional to volume.
When volume increases does mass increase?
As the volume of the material increases, the mass will also increase. The greater the volume of the object the greater the number of atoms present. This will result in the object having greater mass.
Why does density decrease as volume increases?
Density changes with temperature because volume changes with temperature. Density is mass divided by volume. As you heat something up, the volume usually increases because the faster moving molecules are further apart. Since volume is in the denominator, increasing the volume decreases the density.
How do you calculate mass with volume and density?
Divide the mass by the density of the substance to determine the volume (mass/density = volume). Remember to keep the units of measure consistent. For example, if the density is given in grams per cubic centimeter, then measure the mass in grams and give the volume in cubic centimeters.
How do you find volume if you have mass and no density?
Usually, if the density is not given then we can use the density of water i.e. 1g/cc as a standard or reference but here only mass is given, so the other way to find volume is the water displacement method.
How do you find the mass?
One way to calculate mass: Mass = volume × density. Weight is the measure of the gravitational force acting on a mass. The SI unit of mass is “kilogram”.
Does density increase or decrease?
If the amount of matter is increased without changing the volume, then the density increases (Fig. 2.2 A to 2.2 B). If volume increases without an increase in mass, then the density decreases (Fig.