Does ninhydrin react with proline
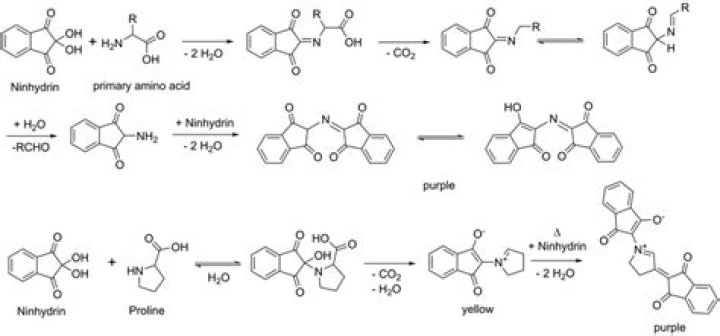
Imino acids, e.g. proline and hydroxyproline, react with ninhydrin to give a yellow color. At higher temperatures (“~100°C), the yellow compound (X) is trans- formed to the purple-red compound (XI) 1°.
What color is proline with ninhydrin?
In most cases, a blue or violet compound (proline and hydroxyproline give a yellow color) is formed owing to reaction of the liberated NH3 with ninhydrin, as shown in Figure 2-11. Color and CO2 production provide a basis for the quantitative determination of amino acids.
Will all amino acids give a positive ninhydrin test?
Ninhydrin Test Free amino groups will react with the ninhydrin reagent to yield a purple solution. Almost all amino acids contain a free amino group (except proline and hydroxyproline). Some proteins also give a positive test with ninhydrin.
Why does Proline turn yellow in the ninhydrin test?
Proline gives a yellow colour because it is a secondary amine. Most amino acids are primary amines with the general structure H2NCHRCOOH . Except for proline and hydroxyproline, all the α-amino acids are oxidized by ninhydrin to give the same intensely colored purple anion. … The product is a yellow-coloured zwitterion.Does ninhydrin react with secondary amines?
The reaction of ninhydrin with secondary amines gives an iminium salt, which is also coloured, and this is generally yellow–orange in color.
Which amino acid give yellow colour in ninhydrin test Why?
The amino acid which gives yellow colour with Ninhydrin in paper chromatography is Proline.
What colour would you expect the proline ninhydrin reaction to yield and why?
Imino acids, e.g. proline and hydroxyproline, react with ninhydrin to give a yellow color. At higher temperatures (“~100°C), the yellow compound (X) is trans- formed to the purple-red compound (XI) 1°.
Is proline an imino acid?
Proline. Proline contains a secondary amine group, called an imine, instead of a primary amine group. For this reason, proline is called an imino acid. Since the three-carbon R-group of proline is fused to the α-nitrogen group, this compound has a rotationally constrained rigid-ring structure.Is proline an alpha amino acid?
Proline is one of the α-amino acids that are used by living organisms as the building blocks of proteins. The L-isomer of proline, which is the only form that is involved in protein synthesis, is one of the 20 standard amino acids common in animal proteins and required for normal functioning in humans.
What is the mechanism of ninhydrin reaction?Answer: The ninhydrin reaction mechanism is essentially an oxidation and reduction reaction process. When we add drops of the ninhydrin solution to the given test sample, the ninhydrin acts as an oxidizing agent. It reacts with the amino group of the compound, leading to delamination.
Article first time published onWould proline give a positive biuret test?
This is because it has an amide group which bends over the copper ion giving the color pink or violet off to the visual eye. Will methionine give a positive Biuret test? The Biuret test will give a positive for peptide bonds. Methionine is a free amino acid and does not have peptide bonds.
Which amino acids will give different colored results to ninhydrin test?
Ninhydrin Test Result Interpretation For ammonia, primary/secondary amines, and amino acids, deep purple colour is obtained. For hydroxyproline and proline, a yellow colour is obtained. For asparagine, brown colour is obtained.
How are amino acid detected using ninhydrin?
In summary, ninhydrin, which is originally yellow, reacts with amino acid and turns deep purple. It is this purple color that is detected in this method. Ninhydrin will react with a free alpha-amino group, NH2-C-COOH. This group is contained in all amino acids, peptides, or proteins.
Does ninhydrin react with tertiary amines?
It’s a stable hydrated product of indane-1,2,3-trione. It is a white to light yellow solid that can dissolve in many solvents such as water, ethanol, and acetone at room temperature. Except for tertiary amines, it can react with ammonia, primary and secondary amines, and peptides, forming a beautiful purple product.
Can ninhydrin test distinguish amino acids and proteins?
Uses of Ninhydrin Test Ninhydrin test is used to detect the presence of amino acids in unknown samples. This test is also used in solid-phase peptide synthesis to monitor the protection for amino acid analysis of proteins. As the ninhydrin test is quite sensitive, it is commonly used to detect fingerprints.
What does ninhydrin degrade amino acids into?
one ninhydrin reacts with an amino acid in a such a way that the ninhydrin gets stuck to the amino acid’s nitrogen through a “Schiff base.” that’s really awkward, so the amino acid ditches carbon dioxide CO₂ (decarboxylates)
Why the purple Colour of the spots appear after spraying with ninhydrin?
Amino acids are colorless compounds. In order to see the spots on the chromatogram, you will apply a solution of ninhydrin to the paper. Ninhydrin will react with the amino acid to produce a purple compound. … Like the amino acids, the medicine tablet ingredients will not be visible after the plate has been developed.
Why does ninhydrin stain the skin Blue?
Ninhydrin changes to a blue color in the presence of ammonia (NH3) or amine functional groups (NH or NH2. Ninhydrin can be use to detect proteins and amino acids because they contain amine functional groups.
Which Colour is obtained when protein is treated with ninhydrin solution Mcq?
Explanation: when protein is mixed with a dilute solution of ninhydrin, a purple or blue colour is produced. This test is done by all proteins, dipeptides and also by amino acids.
Why do we use ninhydrin in chromatography?
Ninhydrin (or fluorescamine) is very useful in chromatographic methods for the analysis of amino acids. … This double migration process gives a better separation of the amino acids than a single migration and results in concentration of the different amino acids in rather well-defined spots.
What is the purpose of ninhydrin reagent?
Ninhydrin is a tricyclic 1,2,3-trione, which functions as an amino acid reagent. It is a vital organic building block, which exposes latent fingerprints on porous surfaces like paper, cardboard and raw wood. Ninhydrin is used to detect primary and secondary amines.
Is proline neutral?
Amino acidprolineSingle Letter CodePThree Letter CodeProCharge (+/-/ neutral)neutralPolaritynonpolar
Is proline acidic basic or neutral?
Structures of Amino AcidsR = any number carbons in a hydrocarbon chain *CHIME plug-in required to view these images.ProlineproNeutral Non-polarSerineserNeutral PolarThreoninethrNeutral Polar
What makes proline different from other amino acids?
Proline is unique in that it is the only amino acid where the side chain is connected to the protein backbone twice, forming a five-membered nitrogen-containing ring. … For this reason, Proline can often be found in very tight turns in protein structures (i.e. where the polypeptide chain must change direction).
Why does proline cause a kink?
Helices. … Prolines in alpha helices after the first turn (4th residue) cause a kink in the helix. This kink is caused by proline being unable to complete the H-bonding chain of the helix and steric or rotamer effects that keep proline from adapting the prefered helical geometry.
Why are proline and glycine helix breakers?
Tight turns and loose, flexible loops link the more “regular” secondary structure elements. … Proline and glycine are sometimes known as “helix breakers” because they disrupt the regularity of the α helical backbone conformation; however, both have unusual conformational abilities and are commonly found in turns.
Why are proline and glycine know as helix breakers?
Glycine is considered as relatively small (looking at the side group) and is known as a “helix breaker” because it disrupts the regularity of the α helical backbone conformation. … The role of proline and glycine in determining the backbone flexibility of a channel-forming peptide.
Why can we not use UV for the amino acids?
Imaging Proteins by UV Absorbance As such, protein fluorescence requires very powerful UV light sources and very sensitive cameras because the fluorescent emission from proteins is so weak. However, powerful UV light sources can destroy the protein due to long exposure times required to obtain significant data.
What are the two basic reactions that are involved in estimation of amino acid by ninhydrin method?
- alpha-amino acid + Ninhydrin —> Reduced ninhydrin +Alpha amino acid +H2O. …
- alpha-amino acid + H2O —> alpha-keto acid +NH3 …
- alpha-keto acid + NH3 —> aldehyde + CO2
Would you expect biuret reagent to react with amino acids?
Specifically, peptide bonds (C-N bonds) in proteins complex with Cu2+ in Biuret reagent and produce a violet color. A Cu2+ must complex with four to six peptide bonds to produce a color; therefore, free amino acids do not positively react. … Biuret reagent is an alkaline solution of 1% CuSO4, copper sulfate.
Are all amino acids reactive with the biuret reagent Why or why not?
Single amino acids and dipeptides do not give the biuret reaction, but tripeptides and larger polypeptides or proteins will react to produce a light blue to violet complex that absorbs light at 540 nm. … The intensity of the color produced is proportional to the number of peptide bonds participating in the reaction.



