Does osmolarity equal to molarity
the osmolarity of a 1M solution of CaCl2 is 1×3 = 3 osmol/L. This equation can be also used to calculate the osmolarity of solutions whose solutes that do not dissociate such as glucose, urea, glycerol, …. Their osmolarity equals their molarity because n=1.
How are molarity and osmotic concentration related?
The osmotic pressure of a solution is proportional to the molar concentration of the solute particles in solution.
How do I calculate molarity?
To calculate the molarity of a solution, you divide the moles of solute by the volume of the solution expressed in liters. Note that the volume is in liters of solution and not liters of solvent. When a molarity is reported, the unit is the symbol M and is read as “molar”.
What is the molarity and osmolarity?
Whereas molarity measures the number of moles of solute per unit volume of solution, osmolarity measures the number of osmoles of solute particles per unit volume of solution.How do you calculate moles from molar concentration?
To calculate the number of moles in a solution given the molarity, we multiply the molarity by total volume of the solution in liters.
Why is molarity used in osmotic pressure calculations?
Osmotic Pressure and Solute Concentration In contrast to the need to employ solute molality to calculate the effects of a non-volatile solute on changes in the freezing and boiling points of a solution, we can use solute molarity to calculate osmotic pressures.
How do you find molar concentration with pressure and temperature?
Use the rearranged ideal gas law to determine the concentration. For example, with a temperature of 298 K and a pressure of 0.031 atm, the formula is 0.031 atm / (0.0821 atm * liter/mole * K) * (298 K). This is equal to 0.0013 mol/L, or moles per liter.
How do you calculate osmolarity?
The equation: Posm =2 [Na(+)]+glucose (mg/dL)/18+BUN (mg/dL)//2.8 is also the simplest and best formula to calculate plasma osmolality. The concentration of only effective osmoles evaluates effective osmolality or tonicity as: Eosm =2 [Na(+)]+glucose/18. The normal range of plasma tonicity is 275-295mOsm/kg of water.How can molar mass be calculated?
To calculate the molar mass of a compound, calculate the molar mass of each element in the compound, then multiply the element’s atomic mass by the number of atoms of that element in the compound. Add the molar masses of each element in the compound to calculate the molar mass of the compound!
What is the difference between molarity and osmolarity quizlet?osmolarity is greater than its molarity. a correction coefficient that accounts for ions interaction at physiological concentrations (conditions). Osmolarity is the number of moles of solute per liter of solution. Osmolality is the number of moles of solute per kilogram of water.
Article first time published onIs tonicity the same as osmolarity?
Tonicity is equal to the osmolality less the concentration of these ineffective solutes and provides the correct value to use. Osmolality is a property of a particular solution and is independent of any membrane. Tonicity is a property of a solution in reference to a particular membrane.
What osmolarity is isotonic?
The osmolarity and sodium concentration of isotonic fluids are similar to that of plasma and extracellular fluid. Normal plasma osmolarity is 290 to 310 mOsm/L for dogs and 311 to 322 mOsm/L for cats, and isotonic fluids generally have an osmolality in the range of 270 to 310 mOsm/L.
How do you calculate osmolarity of glucose?
The molecular weight of glucose is 180 g. Finally, we know that the sugar molecule remains intact and does not break into smaller bits, so 1 M = 1 OsM. Now that everything is in the right units, we just need to divide moles by litres to get the osmolarity: 0.15 mole / 0.25 litre = 0.60 OsM.
What is osmolarity vs osmolality?
Osmolarity and osmolality are frequently confused and incorrectly interchanged. Osmolarity refers to the number of solute particles per 1 L of solvent, whereas osmolality is the number of solute particles in 1 kg of solvent. For dilute solutions, the difference between osmolarity and osmolality is insignificant.
How do you find moles from liters and molarity?
- Find the number of moles of solute dissolved in solution,
- Find the volume of solution in liters, and.
- Divide moles solute by liters solution.
How do you calculate molarity of HCl?
Molarity is a ratio between moles of solute and volume of solution. By dividing the number of moles of HCl by the volume (L) of solution in which it was dissolved, we will obtain the molarity of the acid solution.
How do you find molarity without liters?
There are different methods for finding molarity. If you don’t have volume you can use another formula, like mass divided by relative molecular mass. How can I calculate volume in molarity? Molarity = no of moles of solute/volume of solvent.
How do you find moles from volume and molarity?
Compute the volume of a solution in liters, given the number of moles and molarity, by dividing the number of moles by the molarity in units of moles per liter. For example, a solution containing 6.0 moles and a having a molarity of 3.0 moles per liter has a volume of 2.0 moles per liter.
How do you calculate pressure from molarity?
PV=nRT (which is the formula for pressure for ideal gases) can be used to approximate pressure in real gases. We can convert this to concentration by dividing by volume and getting P=concRT –> since n/V = mol/L which is molarity which is concentration.
How do you convert molar mass to molarity?
- • Always distinguish between amount of. substance in moles (grams) and. concentration of substance in mol/l (g/l)
- • For conversion from mass to molarity. divide the mass (g or g/l) with molar mass. (relative AW/MW/FW)
- • For conversion from molarity to mass. multiply the molarity (mol or mol/l) with.
How do you find molar mass given molarity and volume?
- First you must calculate the number of moles in this solution, by rearranging the equation. No. Moles (mol) = Molarity (M) x Volume (L) = 0.5 x 2. = 1 mol.
- For NaCl, the molar mass is 58.44 g/mol. Now we can use the rearranged equation. Mass (g) = No. Moles (mol) x Molar Mass (g/mol) = 1 x 58.44. = 58.44 g.
How do you find the osmotic pressure of a combined solution?
- π is the osmotic pressure.
- i is the van’t Hoff factor.
- C is the molar concentration of the solute in the solution.
- R is the universal gas constant.
- T is the temperature.
How do you calculate moles in chemistry?
If you want to know how many moles of a material you have, divide the mass of the material by its molar mass. The molar mass of a substance is the mass in grams of one mole of that substance. This mass is given by the atomic weight of the chemical unit that makes up that substance in atomic mass units (amu).
How do you calculate osmolarity from osmolality?
- mOsmol/kg = n x mmol/L.
- mOsmol/kg = (n x mg/dL x 10) ÷ mol wt.
- mOsmol/kg = (n x mEq/L) ÷ valence.
How do you calculate Osmolal gap?
Plasma sodium concentration:mmol/LMeasured Osmolality, plasma:mOsm/kg H2OMeasured ETOH, plasma:mg/dLOsmolality Gap:
How do you calculate osmolarity of IV fluids?
Add together the volumes of each component in order to obtain the total volume of the admixture. Divide the total number of milliosmoles from step [2] by the total volume from step [3], then multiply by 1,000 to obtain an estimate of the osmolarity of the admixture in units of milliosmoles per liter.
Which is the best explanation of osmolarity quizlet?
Osmolarity is an estimation of osmolar concentration and derived from the measured Na+, K+, urea and glucose concentrations in the blood or urine. Diffusion is the passive movement of solute across a permeable membrane.
What is the osmolarity of cells?
Osmolarity is defined as the number of ionic species in molarity that has a characteristic range depending on the species examined. … For example, cell culture media are usually adjusted to 290–320 mOsmol to prevent any osmotic stress to cells.
What is osmolarity and what contributes to it?
The most important factor in determining osmolarity is the molarity of the solution — the more moles of solute, the more osmoles of ions are present. … Consequently, all else being equal, a solution of calcium chloride will have a higher osmolarity than a solution of sodium chloride.
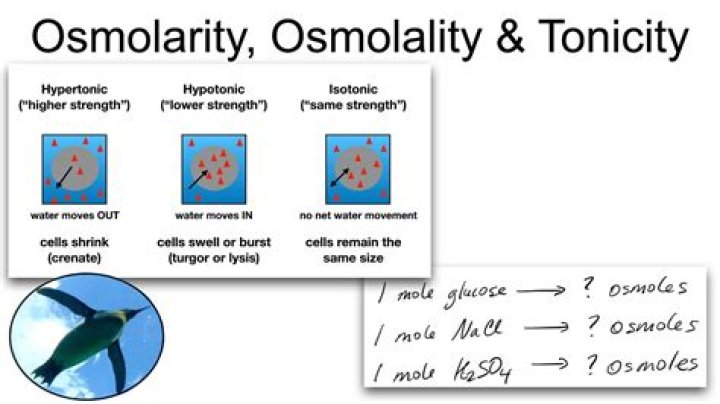
How do you calculate osmolarity and tonicity?
“Whereas a solution’s osmolarity is based solely on its total solute concentration, its tonicity is determined by how it affects cell volume, which depends not only on the solute concentration but also on the solute permeability of cell membranes. A solution is said to be isotonic when it does not alter cell volume.
Can you calculate tonicity?
The latter phenomenon is called osmosis. Tonicity is a measure of the relative concentration of non-penetrating solute on either side of a membrane. … Next, divide the number of grams of solute by the molar mass of the compound to get the number of moles. Calculate the molarity of the solution.



