Does salt have high melting point
Sodium chloride has a high melting and boiling point There are strong electrostatic attractions between the positive and negative ions, and it takes a lot of heat energy to overcome them. Ionic substances all have high melting and boiling points.
Why does salt have a high melting point?
Sodium chloride has a high melting and boiling point There are strong electrostatic attractions between the positive and negative ions, and it takes a lot of heat energy to overcome them. Ionic substances all have high melting and boiling points.
Why does salt have a higher melting point than sugar?
You can melt salt, you just need much more heat energy than it takes melt sugar. Sugar (sucrose) will melt at around 160C, but table salt (sodium chloride) melts at 801C. So, if you are trying to melt salt on your stove top, you probably can’t.
Does salt have a high or low melting point?
NaCl is a solid at room temperature, with a very high melting point (801 °C), similar to the melting points of silver (961.78 °C) and gold (1064.18 °C), although much lower than the decomposition temperature of diamond (3550 °C). An interesting difference between diamond and sodium chloride occurs on heating.What salt has the highest melting point?
Oscar L. MgO has the highest melting point, by a large margin.
What determines the melting point of salt?
As with boiling points, the melting point of a solid is dependent on the strength of those attractive forces. Sodium chloride (NaCl) is an ionic compound that consists of a multitude of strong ionic bonds. Sodium chloride melts at 801°C.
Why does salt not melt when heated?
Why It Matters Only a little bit of energy is required to loosen these intermolecular interactions enough to allow the formation of a free-flowing liquid, so its melting point is relatively low. The ions in a salt crystal are held together by strong ionic bonds.
Does salt have a fixed boiling point?
Water boils at 100oC at 1atm of pressure, but a solution of salt water does not . When table salt is added to water the resulting solution has a higher boiling point than the water did by itself. … Therefore, the salt-water solution will not boil at 100oC.What is the melting point of salt water?
Some metals, like tin, do cause little explosions, but that’s because their melting point is so low (231.9 degrees Celsius). But salt melts at around 800 degrees Celsius – almost 200 degrees higher than aluminium – which means that it should easily be hot enough to trigger the Leidenfrost effect.
Does salt decrease boiling point?Adding salt to water is going to do two things to water’s physical properties: it will raise the boiling point and it will lower the specific heat. … But lowering the water’s specific heat — AKA, the amount of energy needed to change an object’s temperature — will cause the salt water to heat up faster!
Article first time published onDoes salt have low melting point?
If salt is dissolved in the water, the rate of detachment of the ice molecules is unaffected but the rate at which water molecules attach to the ice surface is decreased, mainly because the concentration of water molecules in the liquid (molecules per cubic centimeter) is lower. Hence, the melting point is lower.
Why do salts have high melting and boiling point?
One of the major physical properties is that they have a high melting and boiling points. This happens because there is a strong electrostatic force of attraction between the oppositely charged ions. … ) form a strong ionic bonding which takes a lot of energy to break. Hence, the high melting and boiling point.
What is the same as the melting point of salt?
The melting point of sodium chloride, commonly referred to as table salt, at standard pressure is 801 degrees Celsius. This represents the phase transition from solid, crystalline salt to a liquid. Liquid salt turns to a gas, or boil, at 1,413 degrees Celsius.
Which is likely to have the highest melting point?
The chemical element with the lowest melting point is Helium and the element with the highest melting point is Carbon. The unity used for the melting point is Celsius (C).
Which one will have highest melting point?
The metal with the highest melting point is tungsten, at 3,414 °C (6,177 °F; 3,687 K); this property makes tungsten excellent for use as electrical filaments in incandescent lamps.
How do you determine the highest melting point?
In general, the greater the charge, the greater the electrostatic attraction, the stronger the ionic bond, the higher the melting point.
What happens to salt at high temperatures?
The simplest example of a molten salt would be to heat sodium chloride (“table salt”) to a red heat (greater than 801° C, or 1474° F)1 upon which it would melt into a liquid. This liquid is stable, has a heat capacity similar to water (by volume) and flows much like water does.
Does salt dissolve in heat?
Yes, salt and other ionic compounds like it will dissolve faster the hotter the water it is dissolved in. This is because hot temperatures make atoms move quicker and the quicker they move, the easier they come apart!
Can we heat salt?
When you HEAT salt (NaCl), it melts and then vaporizes. But these physical changes require very very high temperatures. When you INTRODUCE salt to a FLAME, you see a bright yellow coloration to the flame. This is due to excitation of electrons in sodium atoms (sodium ions or Na+ DO NOT have a 3s electrons).
Which factor is generally responsible for high melting points?
In general, ionic compounds have high melting points because the electrostatic forces connecting the ions – the ion-ion interaction – are strong. In organic compounds, the presence of polarity, especially hydrogen bonding, usually leads to a higher melting point.
What does high melting point mean?
A higher melting point indicates greater intermolecular forces and therefore less vapour pressure. Melting point test is not required for every chemical. Usually it is conducted for solid materials under normal conditions.
Why does salt water have a higher boiling point?
In order for water to boil, its vapor pressure has to equal the pressure of the atmosphere, Giddings said. … When salt is added, it makes it harder for the water molecules to escape from the pot and enter the gas phase, which happens when water boils, Giddings said. This gives salt water a higher boiling point, she said.
Does salt increase melting point of ice?
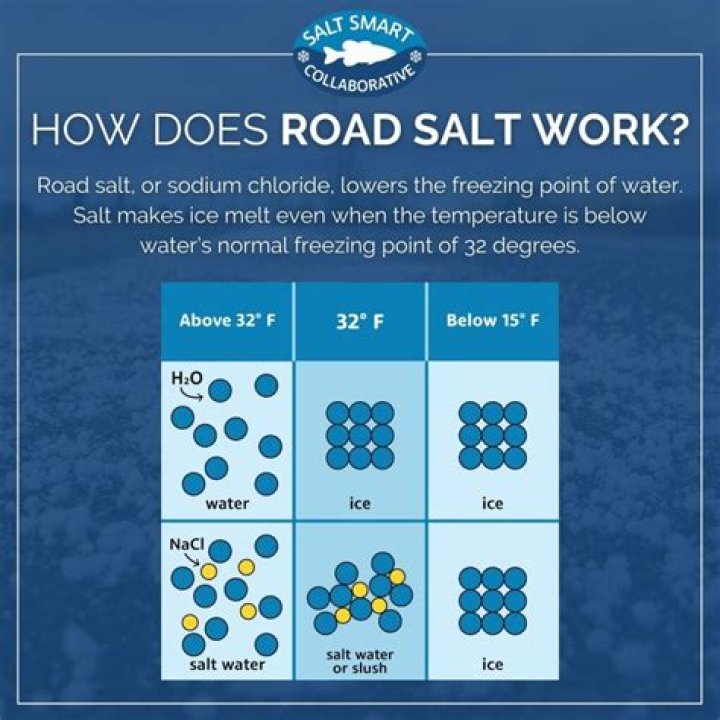
What’s the Point? Water can exist in different states; ice is the solid state of water. The melting point of pure water ice is 32°F (0°C). Adding salt — or other substances — to ice lowers the melting point of ice.
Does salt slow down ice melting?
Adding salt to the ice/water mix causes a temperature drop that slows the melting rate and increases the freezing rate [3]. The net result is that the ice melts more and more slowly after the initial addition of salt. Why does salt melt ice? In pure water, at 0°C, ice melts just as fast as water freezes.
Is table salt melts completely?
When polar molecules enter water, they attract water molecules, which surround them and carry them off into solution. Salt dissolves so well because it completely dissociates into positive and negative ions in water.
Do water and salt have the same boiling point?
Adding salt does not lower the boiling point of water. Actually, the opposite is true. … The usual boiling point of water is 100 °C or 212 °F at 1 atmosphere of pressure (at sea level). You would have to add 58 grams of salt just to raise the boiling point of a liter of water by one half of a degree Celsius.
Why does salt water have a lower heat capacity?
When we dissolve NaCl in water, the ions are held in a rigid cage of water molecules. … It takes less energy to activate these molecules, so the specific heat of the water decreases. The greater the concentration of NaCl, the lower the specific heat capacity of the solution.
What salt has the lowest melting point?
Although calcium chloride has the lowest melting temperature of minus 20 degrees Fahrenheit, it is also the most corrosive and can damage lawns, trees and shrubs since the runoff solution created when using the product seeps into the ground.
What is the melting and boiling point of salt?
NamesMelting point800.7 °C (1,473.3 °F; 1,073.8 K)Boiling point1,465 °C (2,669 °F; 1,738 K)Solubility in water360 g/1000 g pure water at T = 25 °CSolubility in ammonia21.5 g/L at T = ?
How does rock salt melt ice?
Rock salt works by lowering the freezing point of water, forming a brine solution of salt and water. The solution flows under the ice and breaks the bond between the ice and the surface, such as pavement. There are other formulations of ice melt, as well, that may be a more environmentally friendly solution.
Why does sodium fluoride have a high melting point?
Electrostatic force varies with the inverse square of distance, so the ions are more strongly attracted to each other in NaF than in NaCl, NaBr or NaI. Stronger attractions between the ions in the crystal lattice means a higher temperature is required to melt the crystal.