Does sucrose dissolve in methanol
For example, it is known that sucrose is insoluble in absolute methanol and eth anol, and its solubility in water–alcohol mixtures is the higher, the higher is the water content of the mix ture [1].
Will sugar dissolve in methanol?
Lactose, sucrose, maltose, galactose, fructose, and many other carbohydrates and derivatives are found highly soluble in cold absolute methanol containing sufficient calcium chloride.
Does alcohol dissolve sucrose?
Sugar Dissolving in Different Liquids Sugar dissolves well in water because water is very polar and interacts with the polar areas of sucrose. Sugar does not dissolve very well in alcohol because alcohol has a large part that is pretty non-polar.
What would dissolve in methanol?
NameFormulaSolubilityMethanolCH3OHmiscibleEthanolC2H5OHmisciblePropanolC3H7OHmiscibleIs sucrose solubility in ethanol?
4.5 Solubility Sucrose octaacetate is readily soluble in organic solvents like acetonitrile, benzene, pyridine, and methanol. It has a solubility of about 12% (v/v) in 95% ethanol at room temperature [6].
Is methanol a solvent?
Methanol is primarily used as an industrial solvent to help create inks, resins, adhesives, and dyes. It is also used as a solvent in the manufacture of important pharmaceutical ingredients and products such as cholesterol, streptomycin, vitamins and hormones.
Is sucrose a white sugar?
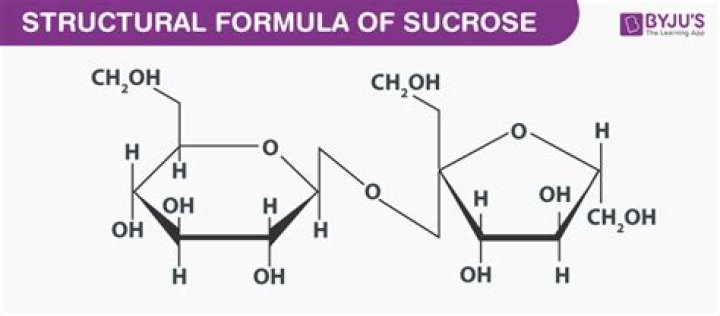
Sucrose is crystallised white sugar produced by the sugar cane plant and can be found in households and foods worldwide. Sucrose is a disaccharide made up of 50% glucose and 50% fructose and is broken down rapidly into its constituent parts. Due to its glucose content, sucrose has a GI value of 65.
Does methanol have a low solubility?
For primary alcohols, the trend is the longer the chain, the less soluble. However, at room temperature, methanol, ethanol and propanol are all completely miscible with water. At lower temperature, below the freezing point of water, there is finite solubility.Why is water a good solvent for methanol?
Methanol is soluble in water. Like water, methanol has relatively strong hydrogen bonding acting between its molecules. … The forces of attraction between the ions and water molecules are strong enough to overcome the ion-ion and water-water force of attraction.
Does sucrose dissolve in acid?The solubility of sucrose in mixtures of propionic acid or ester with water and octanoic acid with tert-pentanol was determined enzymically. Sucrose was hydrolyzed in 1.2 M HCl for 1 h at 50 °C and neutralized with 1.2 M NaOH.
Article first time published onWhy do sucrose molecules dissolve in water?
The sucrose molecules are attracted to one another by positive and negative polar areas. The polar water molecules attract the oppositely charged polar areas of the sucrose molecules and pull them away, resulting in dissolving.
Is sucrose more soluble in water or hexane?
Sucrose has a high solubility in water as compared to hexane as sucrose is a polar molecule and hexane have no hydrogen bonding.
Why does sucrose not dissolve in hexane?
These bonds will form HYDROGEN BONDS and allow the compounds to DISSOLVE IN WATER. Ethanol, glycerin, sucrose, and urea are POLAR AND CANNOT DISSOLVE IN HEXANE HEXANE DOES NOT HAVE HYDROGEN BONDING. The bonding will only occur in OH or NH gropus.
Why is sucrose soluble?
The bond between the oxygen and hydrogen atoms (O–H bond) in sugar (sucrose) gives the oxygen a slight negative charge and the hydrogen a slight positive charge. … The polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes sucrose dissolve in water.
Does sucrose dissolve electricity in water?
Substances like sodium chloride which strongly conduct electricity in aqueous solution are called strong electrolytes. All of the bonds in the sucrose molecule are strong covalent bonds. … Substances like sucrose which do not conduct electricity in aqueous solution are called non-electrolytes.
Is sucrose soluble in petroleum ether?
3. Table sugar, sucrose, is soluble in water, but is insoluble in petroleum ether (an organic solvent similar to gasoline).
Is calcium hydroxide a solvent?
Calcium hydroxide is a widely used antimicrobial agent in endodontics. The solvent used for this agent influences both the physical and chemical properties of the material, including its viscosity and ion release pattern.
Is sucrose soluble in organic solvent?
Properties. Sucrose octapropionate is only slightly soluble in water (less than 0.1 g/L) but is soluble in many common organic solvents such as isopropanol and ethanol, from which it can be crystallized by evaporation of the solvent.
Does sucrose break down into glucose?
Enzymes in your mouth partially break down sucrose into glucose and fructose. However, the majority of sugar digestion happens in the small intestine ( 4 ). The enzyme sucrase, which is made by the lining of your small intestine, splits sucrose into glucose and fructose.
Is sucrose reducing sugar?
4.4 Chemistry Sucrose is a non-reducing sugar and must first be hydrolyzed to its components, glucose and fructose, before it can be measured in this assay.
What kind of solvent is methanol?
Methanol is the primary alcohol that is the simplest aliphatic alcohol, comprising a methyl and an alcohol group. It has a role as an amphiprotic solvent, a fuel, a human metabolite, an Escherichia coli metabolite, a mouse metabolite and a Mycoplasma genitalium metabolite.
What products is methanol found in?
Products that contain methanol include windshield washer fluid, gas line antifreeze, carburetor cleaner, copy machine fluid, perfumes, food warming fuel, and other types of fuels.
What is the main use of methanol?
Methanol — the simplest alcohol (CH3OH) — is a chemical building block for hundreds of everyday products, including plastics, paints, car parts and construction materials. Methanol also is a clean energy resource used to fuel cars, trucks, buses, ships, fuel cells, boilers and cook stoves.
Which solvent is best for extraction?
Hexane – Hexane is a popular solvent for extraction as it has a very low VOC, is safe to be used with goods produced for consumption, and it produces no foul odor or poisonous fumes. All of this coupled with hexane’s low boiling point, it is considered a safe, fast, easy to use solvent for extraction.
What is the strongest solvent?
As per the general information floating around in the web and the details given in some of the books water is the strongest solvent amongst others. It is also sometimes called the “universal solvent” as it can basically dissolve most of the substances than any other liquid. Water is a good solvent due to its polarity.
What are some commonly used solvent for extraction?
Solvents commonly used in extraction of medicinal plants are polar solvent (e.g., water, alcohols), intermediate polar (e.g., acetone, dichloromethane), and nonpolar (e.g., n-hexane, ether, chloroform).
Is methanol soluble in alcohol?
IUPAC namecommon namemp (°C)triphenylmethanoltriphenylcarbinol162IUPAC namebp (°C)solubility in watermethanol65miscibleethanol78miscible
Does ethanol dissolve in methanol?
Each alcohol consists of a carbon chain (always nonpolar) and a OH group (which is polar). … Solubility of alcohols is therefore determined by the stronger of the two forces. Because of the strength of the attraction of the OH group, first three alcohols (methanol, ethanol and propanol) are completely miscible.
Which compound is most soluble in methanol?
CompoundCH3CH2OHIUPAC NameEthanolCommon NameEthyl alcoholMelting Poing (oC)-114.7Boiling Point (oC)78.5
Why is sucrose insoluble in dichloromethane?
no (Although dichloromethane and sucrose are both polar molecules, dichloromethane is not an organic solvent (sucrose is an organic compound). Additionally, sucrose has hydrogen bonding which is much more polar than the dipole-dipole interactions of dichloromethane. Thus, sucrose does not dissolve in dichloromethane.)
Is sucrose solution an electrical conductor?
Sucrose is a covalent molecule when it dissolves in water it doesn’t form ions so it can’t conduct electricity.