How can CaCO3 be reduced in water
The usual treatment for calcium carbonate precipitation is to inject acid to acidify the water, lowering the pH to 7 or less. Common acids used for this purpose include sulfuric acid, muriatic acid, and hydrochloric acid. Other acids such as citric acid and nitric acid can be used, but they are more expensive.
How do you reduce CaCO3?
The general recommendation to prevent CaCO3 precipitation is to lower the water pH by acid injection. Using chemical can be very expensive. Also, chemicals are harmful for environment and health. Magnetic process as a physical method can be alternative to the chemicals in order to decrease precipitation of CaCO3.
What happens to CaCO3 in water?
Calcium carbonate reacts with water that is saturated with carbon dioxide to form the soluble calcium bicarbonate. This reaction is important in the erosion of carbonate rock, forming caverns, and leads to hard water in many regions.
How do you remove carbonate from water?
softening through carbonate removal. When the water has a high TH accompanied by a significant M-alk., the water can be softened by using lime to remove the carbonate.Is caco3 soluble in water?
Calcium carbonate appears as white, odorless powder or colorless crystals. Practically insoluble in water.
How do you reduce alkalinity in water?
Raw water alkalinity may be reduced using several different methods: Reverse Osmosis – Membrane filtration has become the popular option for boiler water treatment. With appropriate pretreatment, nearly all carbon dioxide can be eliminated from RO-treated feedwater.
How do Ca2+ and Mg2+ get into water?
Common cations found in hard water include Ca2+ and Mg2+. These ions enter a water supply by leaching from minerals within an aquifer.
How does CaCO3 neutralize acid?
When calcium carbonate (a solid typically found in rocks such as limestone) reacts with acidic free hydrogen (H+) ions in seawater, the solid calcium carbonate dissolves, forming free calcium (Ca2+) ions and free bicarbonate (HCO3–) ions .How can we reduce the hardness of water?
The most common way to remove hardness from drinking water is to install a water softener, which replaces the calcium and magnesium ions with sodium ions. For every milligram of hardness that is removed, 0.46 milligrams of sodium will be added to the water.
Why is CaCO3 insoluble in water?The calcium carbonate is a strong ionic compound. It has a very high electrostatic force of attraction; because of this the atoms are very tightly held together. These attractions are so strong that it becomes difficult to break the bonds apart. … And so calcium carbonate is insoluble in water.
Article first time published onWhat is CaCO3 in water?
Hardness is primarily caused by the presence of calcium and magnesium in the water. … In general, hardness values of less than 75 mg/L as calcium carbonate (CaCO3) represent soft water, and values above 150 mg/L CaCO3 represent hard water.
How does CaCO3 dissociate?
Calcium carbonate is considered a soft, nonabrasive filler. Its natural color is white, however, it can be easily colored. It is chemically stable up to 800 °C and above this temperature it dissociates into calcium oxide and carbon dioxide.
Would CaCO3 be more soluble in acid or water?
If the solvent is water, calcium carbonate is a bit soluble in water, about 10 or 20 parts per million. The solubility will increase if water is more acidic. In the case of other solvents, the case is similar with solubility increasing in presence of some acid in the solvent.
What acid will easily dissolve CaCO3?
Calcium carbonate will dissolve quickly in a strong acid, like hydrochloric, even if it is dilute and therefore not so dangerous.
How do you remove carbonate hardness?
Lime is used to remove carbonate hardness and soda ash is suggested for removal of noncarbonated hardness.
Does CaCO3 cause hardness?
According to the solubility rules, the carbonates are insoluble so, calcium carbonate is insoluble in water. It does not dissociate in water and does not produce the calcium ions which is the cause of hardness.
How can I make 1000 ppm hard water?
Water Hardness Standard, 1000 ppm Ca2+. To prepare this standard, add 3.67 grams of reagent-grade calcium chloride (CaCl2·2H2O) to a 1 liter volumetric flask about half full of distilled water. Swirl the flask to dissolve the solid and fill to the mark with distilled water.
What is alkalinity reduction?
It is important to understand the meaning of the parameter alkalinity then one considers how to reduce it: Alkalinity is the sum of dissolved species that can react to neutralize strong acid and it is defined by the analytic procedure to titrate a water sample with acid until the pH is reduced to below the pKa of …
How does alkalinity affect nitrification?
During nitrification, 7.14 mg of alkalinity as CaCO3 is destroyed for every milligram of ammonium ions oxidized. Lack of carbonate alkalinity will stop nitrification. In addition, nitrification is pH-sensitive and rates of nitrification will decline significantly at pH values below 6.8.
What if water has high alkalinity?
What if my water is acidic or alkaline? If you are concerned about the taste of your well water, you can install a treatment system to neutralize the acidity or alkalinity of your private well. A neutralizer is a treatment system that can reduce the acidity or alkalinity of your well water.
How do you reduce total hardness?
- Drain and replace some water. Before you replace the water, test your water source. …
- Use pool flocculant. If you can’t or don’t want to drain any water from the pool, try using pool floc to clump the excess calcium. …
- Try muriatic acid.
How can we reduce the hardness of water in aquaculture?
Excessively hard water can be treated by ion exchange to remove calcium and magnesium. This technique usually is too expensive for treating waters used in aquaculture, however.
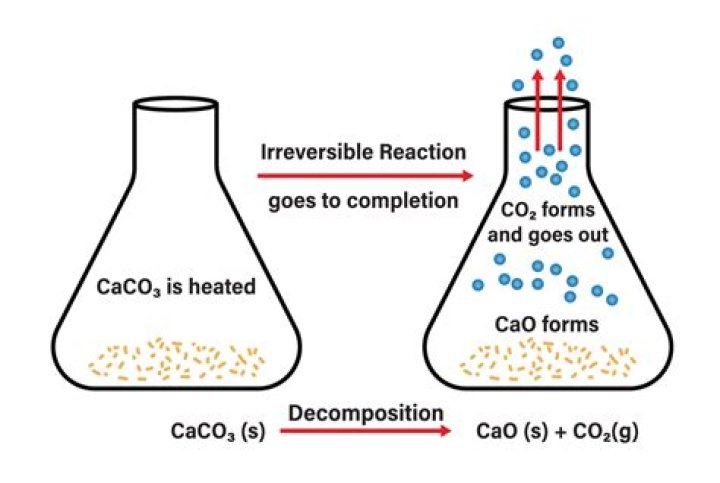
What happens when CaCO3 is heated?
If we heat calcium carbonate to the temperature of 1200 K, it decomposes to form calcium oxide and evolve carbon dioxide gas.
How does limestone affect water pH?
Limestone is calcareous (contains calcium) and is known for its ability to both harden the water and increase the pH. … Your local tap water may also be naturally hard (containing calcium and magnesium) and alkaline (containing carbonate), which adds further to the high pH.
How is limestone used in water treatment?
Production. Lime softening is a water treatment process that uses calcium hydroxide, or limewater, to soften water by removing calcium and magnesium ions. In this process, hydrated lime is added to the water to raise its pH level and precipitate the ions that cause hardness.
What is CaCO3 most soluble in?
Solubility. Calcium carbonate has a very low solubility in pure water (15 mg/L at 25°C), but in rainwater saturated with carbon dioxide, its solubility increases due to the formation of more soluble calcium bicarbonate.
Why does nacl dissolve and CaCO3 not?
That’s partly due to the fact that the ions in sodium chloride, Na+ and Cl-, have lower charges than the ions in calcium carbonate, Ca2+ and CO32-. The higher the charges on the ions, the stronger their electrostatic attraction for each other, and the harder it is for the water to pull them apart.
What happens to calcite in water?
Hello Radek, Water that is not acid will not hurt calcite unless you leave it in the water for weeks. Solubility depends on the concentration of the common ions ( ions of the elements in the mineral) in the water, the lower the concentration in the water the more easily or faster it will dissolve.
Is CaCO3 a limestone?
Limestone is a sedimentary rock made of calcium carbonate (CaCO3), usually in the form of calcite or aragonite. It may contain considerable amounts of magnesium carbonate (dolomite) as well. However, minor constituents of clay, iron carbonate, feldspar, pyrite, and quartz is also commonly present.
What is the product of caco3 and h2o?
Calcium carbonate react with carbon dioxide and water to produce calcium hydrogen carbonate.
Does HNO3 dissociate in water?
Water, being a polar solvent, makes HNO3 ionise or dissociate or simply break down into Hydronium ions, i.e,H+ or H3O+ ions and nitrate NO3- ions..that’s all. Nitric acid does NOT react with water, water disassociates the molecules into those ions.



