How can disulfide bonds be broken
Disulfide bonds can be broken by addition of reducing agents. The most common agents for this purpose are ß-mercaptoethanol (BME) or dithiothritol (DTT).
Can disulfide bonds be broken by heat?
Many extracellular globular proteins have evolved to possess disulphide bonds in their native conformations, which aids in thermodynamic stabilisation. However, disulphide bond breakage by heating leads to irreversible protein denaturation through disulphide-thiol exchange reactions.
How can disulfide bonds be cleaved?
Disulfide bonds can be cleaved at an alkaline pH by treating a protein with excess of a reagent disulfide in the presence of catalytic amounts of thiol. … Disulfide bonds cleaved by the reaction can be re-formed by exposing the mixed disulfide of the protein to catalytic amounts of thiol.
How can disulfide bonds be broken in hair?
Human hair is made mostly of a fibrous protein called keratin. … In healthy hair, these bonds—known as disulfide bridges—are secure and unbroken. But daily life takes its toll: Too much exposure to sunlight and products such as bleaches, straighteners, and dyes breaks the disulfide bridges, making hair brittle.How can disulfide bonds be reduced?
Disulfide-reducing reagents are routinely used in biochemical manipulations for (i) reducing the native disulfide bonds in proteins and (ii) maintaining the essential thiol groups in proteins by preventing their oxidation to the disulfide state. Dithiothreitol (DTT) is the most popular disulfide-reducing reagent.
How do you break disulfide bonds in hair naturally?
The disulphide bonds cannot be broken apart by oxidizing agents, which are acids, but can be broken apart by strong reducing agents, which are bases. Alkaline solutions, therefore, are applied to hair to break apart the disulphide bonds. The hair is then held straight and acidic solutions are applied to it.
At what temp do disulfide bonds break?
Some disulfides decompose at lower temperatures than those containing small hydrocarbon radicals. For example, dibenzyl disulfide decomposes around 200°C to generate sulfur, stilbene, and a mixture of other compounds [2].
Can broken disulfide bonds cause protein loss in hair?
The chemicals and/or thermal tools being used react with the disulfide bonds breaking them and causing protein loss, leaving behind damaged hair. When damage occurs, it affects the disulfide bonds throughout the entire hair fiber.How do hair bonds break?
Disulphide bonds are broken by the hair being heated by a hairdryer, curling tongs or straighteners, and by the application of chemicals such as those used in hair relaxants or bleaching.
What bonds are in the hair?The keratin and bonds in your hair determine its texture and shape and give each hair around a third of its overall strength. There are three types of bond that link the protein chains together: disulphide bonds (which are strong); and hydrogen bonds and salt bonds (which are weaker).
Article first time published onHow do disulfide bonds form?
2.58. Correct disulfide bonds are usually reformed during the renaturation step. Although air provides a suitable environment for disulfide bond formation, usually a redox system containing reduced and oxidized forms is added to the refolding buffer.
How do disulfide bonds affect protein structure?
Disulfide bonds play a critical stabilizing role in many protein structures by forming cross-links between different regions of polypeptide chains.
What are disulfide bonds between?
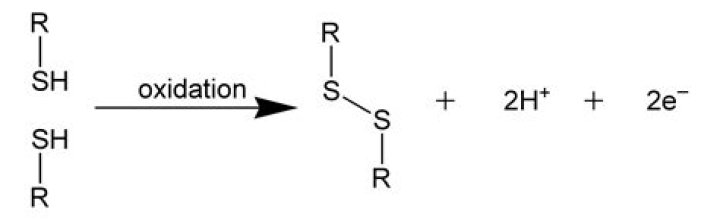
A disulfide bond is a covalent bond between two sulfur atoms (–S–S–) formed by the coupling of two thiol (–SH) groups. Cysteine, one of 20 protein amino acids, has a –SH group in its side chain, and can easily be dimereized to cystine in aqueous solution by forming a disulfide bond.
How do you break disulfide bonds in proteins?
When biochemists want to break disulfide bonds within or between molecules, they add dithiothreitol to their buffer solutions.
What reagent is used to reduce disulfide bonds?
Dithiothreitol and 2-mercaptoethanol (ME) are the most commonly used reagents for the reduction of disulfide bonds.
How do you change a disulfide bond?
Breakage of Disulfide Bond in Keratin by ILs To solubilize wool, it is necessary to partially cleave the disulfide bond in keratin. The cleavage of disulfide bonds can be achieved by reduction, oxidation, sulfitolysis, or oxidative sulfitolysis with traditional methods.
Can disulfide bonds be broken by water?
Disulfide bonds are chemical side bonds. Disulfide bonds link together two sulfur atoms attached to cysteine amino acids within the polypeptide chains. … Disulfide bonds cannot be broken by water or heat.
Are disulfide bonds irreversible?
Chemical modification of disulfide bonds is commonly used for structural and functional analysis of proteins. … The process is irreversible, but entails stepwise chemical reactions.
Are disulfide bonds Ptms?
Amino AcidAbbrev.ModificationCysteineCysdisulfide-bond formation, oxidation to sulfenic, sulfinic or sulfonic acid, palmitoylation, N-acetylation (N-terminus), S-nitrosylation
How do you break hair bonds naturally?
- Combine 1 tablespoon of castor oil with 1 tablespoon of coconut oil.
- Heat the ingredients and massage into your scalp for 15 minutes.
- Leave the oil on your hair and scalp for 30 minutes post massage.
- Wash it off with cold water and a mild shampoo (sulfate-free preferably).
Does keratin break disulfide bonds?
The Young’s modulus results from α-keratin stretching, which is resisted by disulphide bonds and by hydrogen bonds between turns that stabilize the helical structure of the keratin.
What does heat do to keratin?
High temperatures can also form cracks and ruptures in the cuticle, making the hair even more vulnerable to future damage. Finally, heat can break down the keratin in your hair, which affects the strength and texture of the hair.
Can hair bonds be repaired?
Hair bonds are what give your hair strength, elasticity and shine and everything from heat styling to colouring, to brushing and pollution can cause the bonds to break. Luckily they can be repaired, with bond repair hair treatments working from the outside in to help repair and strengthen the bonds.
Can disulfide bonds be repaired?
During the perming, coloring, bleaching, and straightening processes, the chemicals break down the disulfide bonds and harden them into a new shape. … For a long time, nothing could be done to restore the broken disulfide bonds except to cut off the damaged hair.
How do you know if you have broken hair bonds?
Broken hair bonds compromise elasticity, which leads to limp, lifeless hair. To test the condition of your hair’s elasticity, isolate a single hair and gently tug on it with your thumb and forefinger. If the hair doesn’t snap back into shape, your elasticity is impaired. Split ends.
Why are disulfide bonds important in hair?
While the curliness (or straightness) of your hair depends on the shape of the follicle, it’s the disulphide bonds that keep the hair in the shape it was formed, and they can only be altered by perming or relaxing. Disulphide bonds also give your hair its elasticity and strength.
Does Olaplex fix disulfide bonds?
When disulfide bonds are broken, it results in damage. OLAPLEX restores damaged and compromised hair by repairing from the inside out with our patented single ingredient, Bis-Aminopropyl Diglycol Dimaleate. Once all the bonds are intact and in alignment, you have healthy, beautiful, shiny, touchable hair.
Why are disulfide bonds important?
Disulfide bonds function to stabilize the tertiary and/or quaternary structures of proteins and may be intra-protein (i.e., stabilizing the folding of a single polypeptide chain) or inter-protein (i.e., multi-subunit proteins such as antibodies or the A and B chains of insulin).
What are some ways that hair can be destroyed?
- You style your hair when it’s dripping wet. …
- You wash your hair in hot, hot water. …
- You’re still using 2-in-1 products. …
- You’re using the wrong hot tools. …
- You threw away the nozzle to your blow-dryer. …
- You’re not thinking about SPF protection.
When hair is permed what types of bonds are broken?
When the hair is permed (and sometimes when straightened) the disulfide bonds (the chemical side bonds) of the hair are broken through a chemical reaction called ‗reduction’. A reduction reaction involves either the removal of oxygen or the addition of hydrogen.
How are salt bonds broken?
A salt bond is another weak, cross-link side bond that can be broken by changes in pH. They are easily broken by strong alkaline or acidic solutions and account for about one-third of the hair’s overall strength.



