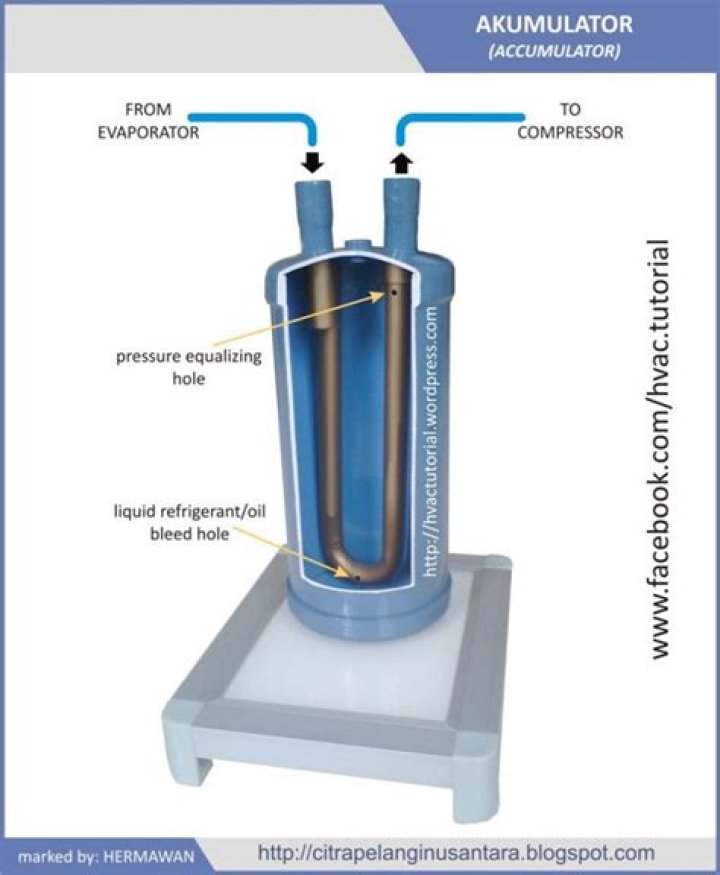
How can pressure be increased

Pressure can be increased by either increasing the force or by decreasing the area or can oppositely be decreased by either decreasing the force or increasing the area.
What happens when you increase the pressure of a gas?
Boyle found that when the pressure of gas at a constant temperature is increased, the volume of the gas decreases. … this relationship between pressure and volume is called Boyle’s law. So, at constant temperature, the answer to your answer is: the volume decreases in the same ratio as the ratio of pressure increases.
What causes pressure in a gas?
Gas pressure is caused when gas particles hit the walls of their container. The more often the particles hit the walls, and the faster they are moving when they do this, the higher the pressure. This is why the pressure in a tyre or balloon goes up when more air is pumped in.
How can we increase the gas pressure inside a sealed container?
- You could add more molecules. …
- You could increase the temperature. …
- You could decrease the size of the container, this would force the molecules into a smaller area and therefore increase the number of collisions that take place.
How do you increase pressure without changing volume?
Yes, if you increase the temperature of a isolated system then the gas inside it expands and that expansion causes the pressure on the walls to increase if volume kept constant.
How can you increase the pressure of a gas assuming that temperature remains constant?
Gay Lussac’s Law – states that the pressure of a given amount of gas held at constant volume is directly proportional to the Kelvin temperature. If you heat a gas you give the molecules more energy so they move faster. This means more impacts on the walls of the container and an increase in the pressure.
Can natural gas pressure be increased?
You cannot increase that pressure by removing unneeded gas pipe. Instead, you can try having the gas company adjust or replace your regulator. If you’re measuring the pressure while gas is flowing, you might have lines that are too small, long or bendy.
Why does the pressure inside a container of gas increase if more gas is added to the container?
Why does the pressure inside a container of gas increase if more gas is added to the container? There is an increase in the number of collisions between particles and the walls of the container. … The pressure will increase.How do you increase air pressure in a container?
First, simply adding molecules to any particular container will increase the pressure. A larger number of molecules in any particular container will increase the number of collisions with the container’s boundary which is observed as an increase in pressure.
What two factors affect gas pressure?Overview. The pressure of an enclosed gas is dependent on the number of gas particles, the volume of the container, and the temperature of the gas. Increasing the number of gas particles in a container will increase the pressure.
Article first time published onWhat is gas pressure?
Gas pressure is the name given to the force exerted by gas particles colliding with the wall of their container. Pressure is force exerted over an area. Gas pressure is the force exerted by a gas on a specific area.
What uses gas pressure?
An example of an application that uses gas pressure for transportation is the engine of a car. Gasoline or diesel fuel is added to air and compressed in the engine. The fuel burns, heating up the gas and producing pressure to push on the pistons of the engine.
How do you increase the pressure of a gas pipe?
Increasing temperature adds energy to the gas molecules, increasing their motion and, again, increasing collisions. Decrease the volume of the gas. This is the “V” in the equation. By their very nature, gases can be compressed, so if the same gas can be put into a smaller container, it will exert a higher pressure.
Does heat increase pressure?
As the temperature increases, the particles move faster, and therefore have greater speeds, so greater momentum and therefore greater force when they collide with the walls, so the pressure increases.
How do you increase the pressure on a natural gas regulator?
- With a small amount of gas flowing through the regulator: Rotate the gas pressure regulator adjustment screw clockwise to raise the outlet pressure.
- Rotate the adjustment screw counter-clockwise to lower the outlet pressure.
What does low gas pressure mean?
A low-pressure system means that the home is supplied with a gas pressure around 7 in. There are inches of water column. When all the gas appliances are on, the pressure drop must be less than a half an inch.
What should the gas pressure be in my home?
The home pressure of natural gas is between 0,25 to 60 psi depending on the amount of houses served by the same line. The gas pipelines can reach 1500 psi due to long distances for transportation.
How do you find pressure when temperature increases?
- First convert the temperatures into kelvin.
- T 1 = 27 + 273 = 300 K.
- T 2 = 42 + 273 = 315 K.
- Now calculate the new pressure.
- T 1 = 300 K.
- T 2 = 315 K.
- P 1 = 1.25 × 10 5 Pa.
What is the effect on the pressure of a gas if its temperature is increased at constant volume?
What is the effect on the pressure of a gas if its temperature is increased at constant volume ? The pressure of the gas increases.
Why does the pressure of a gas sample increase when the temperature is increased assume the volume remains constant?
The volume of the gas increases as the temperature increases. As temperature increases, the molecules of the gas have more kinetic energy. They strike the surface of the container with more force. If the container can expand, then the volume increases until the pressure returns to its original value.
How would the pressure of the gas in an air tank change if more gas particles are added?
With more particles there will be more collisions and so a greater pressure. … Because the area of the container has increased, there will be fewer of these collisions per unit area and the pressure will decrease. Volume is inversely proportional to pressure, if the number of particles and the temperature are constant.
What happens to the gas pressure if the gas is released from a small container to a large container?
Pressure is also affected by the volume of the container. If the volume of a container is decreased, the gas molecules have less space in which to move around. As a result, they will strike the walls of the container more often, and the pressure increases. … The pressure of the gas doubles.
What are the 3 main factors that influence pressure?
- Temperature.
- Altitude or Elevation.
- Moisture ow water vapour.
Which of the following changes would increase the pressure of a gas in a closed container?
Changing the temperature affects pressure in a closed container. Raise the temperature, and the pressure increases. This occurs due to the increased movement of the gas molecules. Double the temperature, and you double the pressure.
What factors influence pressure?
- Temperature: As the temperature increases, air expands because of which its density decreases which results in low pressure. …
- Height from Sea Level: …
- Moisture in Air (Humidity): …
- Gravitation of Earth: …
- Rotation of Earth:
How do you find gas pressure?
If volume and temperature are held constant, the ideal gas equation can be rearranged to show that the pressure of a sample of gas is directly proportional to the number of moles of gas present: P=n(RTV)=n×const.
What is gas pressure example?
Explanation: Cars have tires. These tires (in British English tyres) have to have air pressure. Inflatable beds can be given as another example as well as baloons.
Does gas pressure increase with temperature?
The pressure law states that for a constant volume of gas in a sealed container the temperature of the gas is directly proportional to its pressure. … This means that they have more collisions with each other and the sides of the container and hence the pressure is increased.
Does increasing volume increase pressure?
More collisions mean more force, so the pressure will increase. When the volume decreases, the pressure increases. This shows that the pressure of a gas is inversely proportional to its volume. This is shown by the following equation – which is often called Boyle’s law.