How do acids and bases dissociate
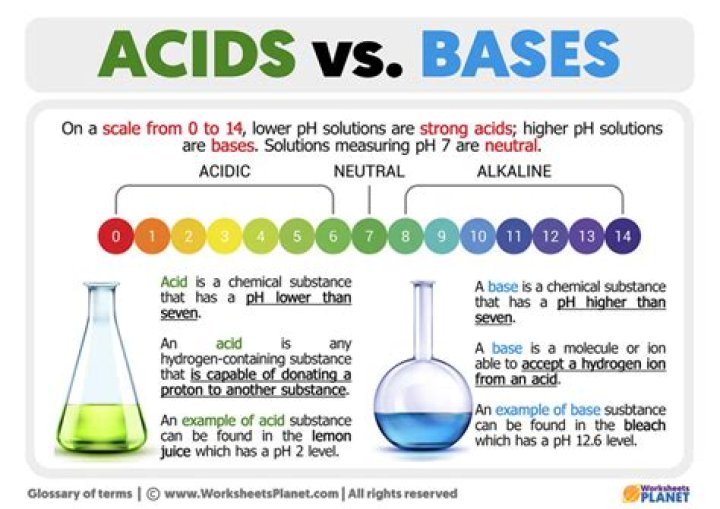
Acids dissociate into H+ and an anion, bases dissociate into OH- and a cation, and salts dissociate into a cation (that is not H+) and an anion (that is not OH-). Figure 2.4. 1 (a) In aqueous (watery) solution, an acid dissociates into hydrogen ions (H+) and anions.
Why do acids and bases dissociate in water?
Acids and Bases Atoms can gain or lose electrons in order to form ions in a process called ionization (compounds formed in this way are called ionic compounds). When ionic compounds dissolve in water, their ions separate from one another in a process called dissociation.
Do strong acids and bases dissociate?
Strong acids/bases dissociate completely whereas weak acids/bases dissociate partially.
What does it mean for an acid or base to dissociate?
Nov 17, 2015. A strong acid or base will completely disassociate, meaning that the acid will form two ions, H+ and its conjugate base. Strong acids completely disassociate because their conjugate base is weaker than water.Why do acids dissociate?
The stronger an acid is, the more easily it loses a proton, H+. Losing a proton means dissociation. Two key factors that contribute to the ease of deprotonation are the polarity of the H—A bond and the size of atom A, which determines the strength of the H—A bond.
How do acids and bases react with water?
Hence, acids and bases mix in water to dissociate and form hydronium and hydroxide ions respectively. Additional information: Acids and bases react together to form salts. This is called a neutralization reaction, and can yield various important salts, like sodium chloride, as HCl+NaOH→NaCl+H2O .
What's the reaction between an acid and a base?
The reaction of an acid with a base is called a neutralization reaction. The products of this reaction are a salt and water.
How do you find dissociation?
– There is a way to calculate the percentage of dissociation of a compound, we have to divide the mass of the dissociated ions with total mass of the dissociated and not dissociated compound and multiply with 100 to get in percentage.What it means to dissociate?
Dissociation is a mental process of disconnecting from one’s thoughts, feelings, memories or sense of identity. The dissociative disorders that need professional treatment include dissociative amnesia, dissociative fugue, depersonalisation disorder and dissociative identity disorder.
What does dissociate mean in chemistry?dissociation, in chemistry, the breaking up of a compound into simpler constituents that are usually capable of recombining under other conditions. … The idea of ionic dissociation is used to explain electrical conductivity and many other properties of electrolytic solutions.
Article first time published onDo acids dissociate?
Acids dissociate into H+ and an anion, bases dissociate into OH- and a cation, and salts dissociate into a cation (that is not H+) and an anion (that is not OH-). Figure 2.4.1 (a) In aqueous (watery) solution, an acid dissociates into hydrogen ions (H+) and anions.
Why do strong acids dissociate completely?
That is the definition: A strong acid is an acid that completely dissociates in water. … For all practical purposes, HCl is completely dissociated in solution. Strong acids have a large dissociation constant, so they dissociate completely in water.
Why weak acids do not dissociate completely?
Since, weak acids have very small dissociation constant (‘k’ value ) , so they do not donate all of its hydrogen ion (H+). That’s why , they do not dissociate completely.
How does acid dissociate?
According to Arrhenius’s original molecular definition, an acid is a substance that dissociates in aqueous solution, releasing the hydrogen ion H+ (a proton): HA ⇌ A− + H+. The equilibrium constant for this dissociation reaction is known as a dissociation constant.
Why do strong bases dissociate?
Strong base showing dissociation. Strong bases fully dissociate to give ions in solution. Weak bases only partially dissociate. Here it can be seen that the ammonia causes the water molecules to break apart by removing a hydrogen ion (proton) from the water molecule leaving an excess of OH- ions in the solution.
How do bases dissociate in water?
Arrhenius argued that bases are neutral compounds that either dissociate or ionize in water to give OH- ions and a positive ion. NaOH is an Arrhenius base because it dissociates in water to give the hydroxide (OH-) and sodium (Na+) ions.
How do acids and bases react with each other explain with an activity?
acids and bases react with each other to produce neutral salt and water . this reaction is known as neutralisation reaction. HCL + NAOH>>>>>>> NACL+H2O. here NACL is neutral salt.
What is formed when acids and bases come together in neutralization reactions?
Neutralization is the reaction of an acid and a base, which forms water and a salt.
How do acid and base react with metal?
In general, acids react with metals and release hydrogen gas. When base reacts with metal it forms salt and release hydrogen gas.
What happens when an acid or a base is added to the water Why does the beaker appear warm?
When an acid or base is added to water, an exothermic reaction takes place, which releases heat. This makes the beaker warm. … This is because the reaction between acid and water produces a lot of heat.
Do acids dissolve bases?
Bases can be either water soluble or insoluble. Insoluble bases react with acids, directly dissolving in the acid as they react. … When an acid and a base are combined, water and a salt are formed. For example, when hydrochloric acid is mixed with sodium hydroxide, water and sodium chloride are formed.
Which is an example of dissociation chemistry?
Dissociation Reaction Examples The reaction in which water breaks into hydrogen and hydroxide ions is a dissociation reaction. When a molecular compound undergoes dissociation into ions, the reaction can also be called ionization. When acids undergo dissociation, they produce hydrogen ions. … HCl → H+(aq) + Cl-(aq)
Why do I dissociate during intimacy?
Dissociating while you’re having sex is more common than you might think. It can happen for many reasons, including anxiety, substance use, or past sexual trauma. Using mindfulness or grounding techniques before and during sex can help you stay present while you’re having sex.
What's the difference between dissociation and disassociation?
Disassociation and dissociation are not interchangeable, they are two different words meaning two different things. Disassociate means what op is asking about. Dissociate means to separate from oneself and disconnect from personal identity, often feeling it replaced by something else.
What Is percent dissociation of weak acids and bases?
Percent dissociation is related to the concentration of both the conjugate base and the acid’s initial concentration; it can be calculated if the pH of the solution and the pKa of the acid are known.
What percentage of the acid is dissociated?
Sample Dissociation Calculation A weak acid, HA, has a pKa of 4.756. If the solution pH is 3.85, what percentage of the acid is dissociated? Percent dissociation is therefore given by 1.413 x 10-4 ÷ 0.0011375 = 0.1242 = 12.42%.
How does dissociation affect conductivity?
Dissociation influences electrical conductivity. The greater the percentage of dissociation for ionic compounds or for acids and bases, the stronger the conductivity.
What is the dissociation of HCl?
1. HCl is a strong acid, HCl → H+ + Cl-, therefore it dissociates (comes apart into + and – ions in water) completely. pH 3 is a low pH (< 7), therefore this is an acidic solution.
Why do strong acids have weak conjugate bases?
Therefore, a strong Lewis acid forms its conjugate Lewis base easily. Because the Lewis base forms easily, it does not strongly want to donate its electron pair (that it just received) to get a proton. That makes it a weak (Lewis) base. … That makes it a weak (Bronsted-Lowry) base.
What is the difference in how strong and weak acids dissociate in water?
According to the way that acid molecules dissociate in water, there are two types of acids as strong acids and weak acids. The main difference between strong and weak acids is that strong acids dissociate completely in aqueous solutions whereas weak acids partially dissociate in aqueous solutions.
Why do weak bases only partially dissociate?
Weak acids like HF and weak bases like NH3 only partly ionize in water due to being weak electrolytes.



