How do you calculate average rate
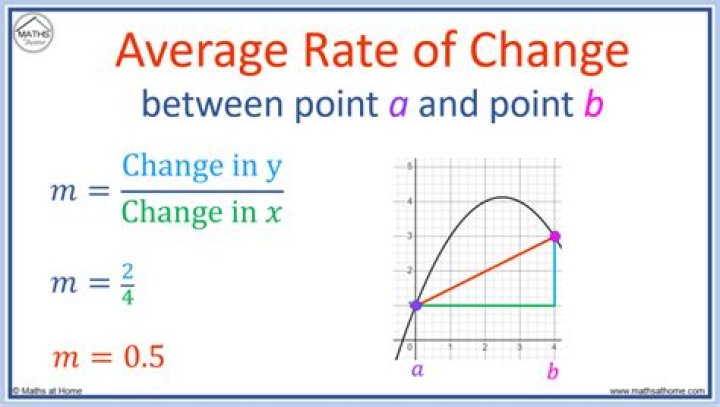
The average rate of change formula is used to find the slope of a graphed function. To find the average rate of change, divide the change in y-values by the change in x-values.
What is an average rate?
Average Rate — a single rate applying to property at more than one location that is a weighted average of the individual rates applicable to each location.
What is average rate of reaction How is it determined?
The average rate of reaction is an average rate, obtained by taking the change in concentration over a time period. The rate at which chemical substances react varies greatly. Usually, a reaction rate involves the change in the concentration of a substance over a given period of time.
How do you calculate the overall average rate of reaction?
You measure the rate by determining the concentration of a component at various times. We insert a minus sign to make the rate a positive number. The overall rate of reaction should be the same whichever component we measure. So, we divide the rate of each component by its coefficient in the chemical equation.Which unit is correct for the rate of reaction?
Reaction rates are usually expressed as the concentration of reactant consumed or the concentration of product formed per unit time. The units are thus moles per liter per unit time, written as M/s, M/min, or M/h.
What is the average rate of the reaction between 0 and 10 s?
Time (s)[C4H8] (M)01.000100.913200.835300.763
What is average rate in accounting?
Definition: The Average Rate of Return or ARR, measures the profitability of the investments on the basis of the information taken from the financial statements rather than the cash flows. It is also called as Accounting Rate of Return.
What is meaning of rate of reaction?
The rate of reaction is the speed at which reactants are converted into products and it highly depends on the type and nature of the reaction. This proportional is increased in the concentration of a product per unit time and to the decrease in the concentration of a reactant per unit time.What is average rate in chemistry?
The average rate is the change in concentration over a selected period of time. … It is determined by finding the slope of the tangent to the concentration vs time curve at that time. • The initial rate is the instantaneous rate at the very beginning of a reaction.
What happens during the rate of reaction?The rate of a reaction is the speed at which a chemical reaction happens. If a reaction has a low rate, that means the molecules combine at a slower speed than a reaction with a high rate. … The reaction will happen faster which means the rate of that reaction will increase.
Article first time published onHow do you calculate average rate of return and net present value?
NPV = F / [ (1 + r)^n ] where, PV = Present Value, F = Future payment (cash flow), r = Discount rate, n = the number of periods in the future and Internal Rate of Return (IRR. In other words, it is the expected compound annual rate of return that will be earned on a project or investment.).
How do you calculate average annual revenue?
Your annual revenue is the amount of money your company earns from sales over a year; it does not include costs and expenses. To calculate your annual revenue, you multiply the quantity of each product you sold by its sale price, and then add each product’s annual sales to determine your gross annual revenue.
How do you calculate average net income?
Average Net Income means Cumulative Net Income divided by the number of Fiscal Years included in the relevant Calculation Period.
What is the instantaneous rate of the reaction at T 800 S?
OneClass: What is the instantaneous rate of the reaction at t=800s? The answer is rate= 6.8×10^-5 m/s…
Is the slowest step the rate determining step?
The slowest step in a reaction mechanism is known as the rate-determining step. The rate-determining step limits the overall rate and therefore determines the rate law for the overall reaction.
What is the order of the reaction?
The Order of Reaction refers to the power dependence of the rate on the concentration of each reactant. … This is distinct from the molecularity (or stoichiometry) of the reaction which is the theoretical integer value of the number of molecules involved in the reaction.
What is the average rate of reaction 12 chemistry?
The average rate of the reaction is the ratio of change in concentration of reactants to the change in time. It is determined by the change in concentration of reactants or products and the time taken for the change as well.
How do you find the average rate of reaction in Class 12?
Hint: The average rate of reaction is calculated by dividing the change in concentration of the reactant or the change in concentration of the product over a specific time duration by the time interval.
How do you find initial rate?
- The average rate of reaction.
- The instantaneous rate of reaction.
- The initial rate of reaction.