How do you determine Ka from acid
The Ka value is found by looking at the equilibrium constant for the dissociation of the acid. The higher the Ka, the more the acid dissociates. Thus, strong acids must dissociate more in water. In contrast, a weak acid is less likely to ionize and release a hydrogen ion, thus resulting in a less acidic solution.
What is the formula for finding ka?
Ka=([H+][A−]HA) where [H+],[A−]&[HA] are molar concentrations of hydronium ion, conjugate base and weak acid at equilibrium.
How do you calculate Ka from pKa?
To create a more manageable number, chemists define the pKa value as the negative logarithm of the Ka value: pKa = -log Ka. If you already know the pKa value for an acid and you need the Ka value, you find it by taking the antilog. In practice, this means raising both sides of the equality to exponents of 10.
How do you find KA from initial concentration?
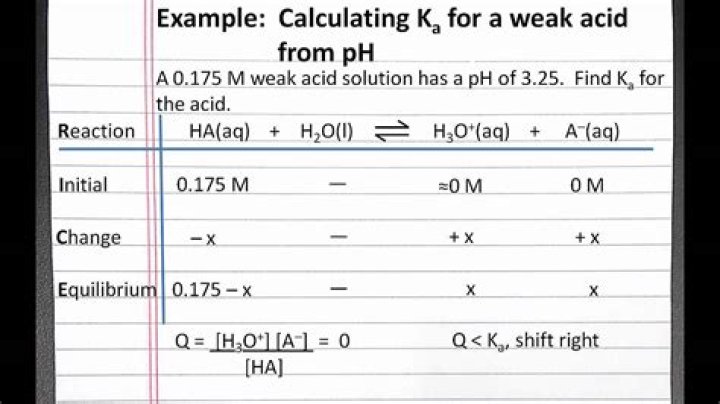
Set up an ICE table for the chemical reaction. Solve for the concentration of H3O+ using the equation for pH: [H3O+]=10−pH. Use the concentration of H3O+ to solve for the concentrations of the other products and reactants. Plug all concentrations into the equation for Ka and solve.How do you find Ka given pH and molarity?
- You start by using the pH of the solution to determine the concentration of the hydronium ions, H3O+ .
- By definition, the acid dissociation constant, Ka , will be equal to.
- Ka=[H3O+]⋅[A−][HA]
How do you determine the Ka of a weak acid or the KB of a weak base?
Explanation: Typically, a weak acid is titrated by a strong base, or a weak base is titrated by a strong acid. In order to measure Ka (or more usually −log10Ka=pKa ). So do the measurement, i.e. the titration of weak acid by a strong base, and measure the pH as you add increments of the titrant.
Is Ka directly proportional to pH?
The acidity of a compound can be given in terms of Ka value which is the acid dissociation constant. The Ka value is directly proportional to the acidity of a molecule.
What is KA and pKa in chemistry?
Ka is the acid dissociation constant. pKa is simply the -log of this constant. Similarly, Kb is the base dissociation constant, while pKb is the -log of the constant.How do you find KA and KB values?
Solve the equation for Kb by dividing the Kw by the Ka. You then obtain the equation Kb = Kw / Ka. Put the values from the problem into the equation. For example, for the chloride ion, Kb = 1.0 x 10^-14 / 1.0 x 10^6.
Can Ka be positive?pKa=-log10(Ka). The log10 of a number greater than 1 is positive. log10 of a number less than 1 is negative. Since you already have a minus sign in your formula for pKa, strong acids with a Ka (much) greater than one will have a negative pKa and weak acids for which the Ka is less than one will have a positive pKa.
Article first time published onWhen the value of Ka increases the acid strength?
The larger the Ka, the stronger the acid and the higher the H+ concentration at equilibrium. Like all equilibrium constants, acid–base ionization constants are actually measured in terms of the activities of H+ or OH−, thus making them unitless. The values of Ka for a number of common acids are given in Table 16.4. 1.



