How do you do Hesss law problems
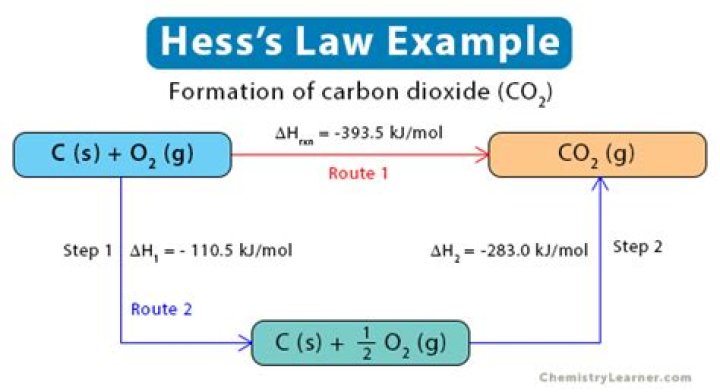
By Hess’s law, the net change in enthalpy of the overall reaction is equal to the sum of the changes in enthalpy for each intermediate transformation: ΔH = ΔH1+ΔH2+ΔH3.
How do you calculate Hess's law?
By Hess’s law, the net change in enthalpy of the overall reaction is equal to the sum of the changes in enthalpy for each intermediate transformation: ΔH = ΔH1+ΔH2+ΔH3.
What is Hess law example?
Hess’s Law Example. Experiments were performed to determine the heat of combustion of methanol, formaldehyde, and hydrogen. Using this data determine the DHrxn for the conversion of methanol to formaldehyde and hydrogen. Rearrange the equations so that they add up to the reaction of interest.
How do you add equations to Hess's law?
2CO(g) + C(s) —> C3O2(g)ΔH° = +127.3 kJCO(g)ΔH f o = −110.5 kJWhat data is needed to use Hess's law?
In order to use Hess’ Law one would have to utilize data such as enthalpy changes and the individual thermochemical equations.
What is Hess law and its application?
Hess’ law allows the enthalpy change (ΔH) for a reaction to be calculated even when it cannot be measured directly. This is accomplished by performing basic algebraic operations based on the chemical equations of reactions using previously determined values for the enthalpies of formation.
What is Hess law class 11?
The Hess’s law states that the total enthalpy change during a complete chemical reaction is the same regardless of the path taken by the chemical reaction. … Hess’s law can be seen as an application of the principle of conservation of energy.
Can you subtract in Hess law?
operationenergy changerearrangementhas no effect on the energy changereversing the equationchanges the sign of the energy changeWhat is the main application of Hess's law?
Applications of Hess’s Law: Thermochemical equations can be added subtracted or multiplied like ordinary algebraic equations. Hess’s law is useful to calculate heats of many reactions which do not take place directly. It is useful to find out heats of extremely slow reaction.
What is Hess law of heat summation discuss its application?Hess’s law of heat summation states that if two or more thermochemical equations can be added together to give a final equation, then the heats of reaction can also be added to give a heat of reaction for the final equation. … However, enthalpy changes for combustion reactions are relatively easy to measure.
Article first time published onWhat is Hess law of constant heat summation explain with example?
Hess’s law, also called Hess’s law of constant heat summation or Hess’s law of heat summation, rule first enunciated by Germain Henri Hess, a Swiss-born Russian chemist, in 1840, stating that the heat absorbed or evolved (or the change in enthalpy) in any chemical reaction is a fixed quantity and is independent of the …
How do you work out enthalpy problems?
Once you have m, the mass of your reactants, s, the specific heat of your product, and ∆T, the temperature change from your reaction, you are prepared to find the enthalpy of reaction. Simply plug your values into the formula ∆H = m x s x ∆T and multiply to solve. Your answer will be in the unit of energy Joules (J).
How do you know if Hess's law is validated?
If a process written as the sum of several stepwise processes, the enthalpy change of the total process equals the sum of the enthalpy changes of the various steps. Hess’s law is valid because enthalpy is a state function.
Which chapter is Hess law?
Chapter 15.4: Hess’s Law.
What is Hess law BYJU's?
Hess’ law also known as Hess’s law of constant heat summation states, “at constant temperature, heat energy changes (enthalpy – ΔHrec) accompanying a chemical reaction will remain constant, irrespective of the way the reactants react to form product”.
How does Hess's law follows the first law of thermodynamics?
Hess’s law is based on the first law of thermodynamics which says that energy cannot be created or destroyed but can be converted from one form to another. Hess’s Law of Heat Summation states that the enthalpy change for a reaction is the same whether it occurs by one step or by any (hypothetical) series of steps.
How is Hess's law useful in real life?
Hess’s law of constant heat summation Car companies must see how much energy the car engine uses or produces when it burns gasoline. The industry, generally, can measure how much energy each process releases when it is performed, so that they can make effective energy choices.
What is Hess law write down its two applications?
(1) To calculate the heat of formation, combustion, neutralisation, ionization, etc. (2) To calculate enthalpies of reactants and products. (3) To determine the bond enthalpies. (4) To determine the lattice energies of the crystalline solids.
What is an enthalpy level diagram?
An enthalpy diagram plots information about a chemical reaction such as the starting energy level, how much energy needs to be added to activate the reaction, and the ending energy. An enthalpy diagram is graphed with the enthalpy on the y-axis and the time, or reaction progress, on the x-axis.
How do you manipulate chemical reactions?
Reactions may be manipulated by changing the temperature, kinetic energy, surface area, or concentration of reactants or products. Chemical reactions in a state of equilibrium can be manipulated to shift from products to reactants or vice versa.