How do you find an isotope number
Subtract the atomic number (the number of protons) from the rounded atomic weight. This gives you the number of neutrons in the most common isotope. Use the interactive periodic table at The Berkeley Laboratory Isotopes Project to find what other isotopes of that element exist.
What is a isotope number?
Definition of isotopic number : the number of neutrons minus the number of protons in an atomic nucleus.
What is isotope and example?
Isotopes can be defined as the variants of chemical elements that possess the same number of protons and electrons, but a different number of neutrons. … For example, carbon-14, carbon-13, and carbon-12 are all isotopes of carbon.
What is isotopic formula?
Isotopic number is defined as “the number of neutrons minus the number of protons in an atomic nucleus.” Number of protons = atomic number (given by the letter Z) Let us use N for the number of neutrons Then isotopic number = N – Z A = mass number, which is equal to number of neutrons + number of protons A = N + Z A – …How do you find the isotope symbol?
To write the symbol for an isotope, place the atomic number as a subscript and the mass number (protons plus neutrons) as a superscript to the left of the atomic symbol. The symbols for the two naturally occurring isotopes of chlorine are written as follows: 3517Cl and 3717Cl.
How do you find isotopic abundance?
The equation can be set up as a percent or as a decimal. As a percent, the equation would be: (x) + (100-x) = 100, where the 100 designates the total percent in nature. If you set the equation as a decimal, this means the abundance would be equal to 1. The equation would then become: x + (1 – x) = 1.
How do you calculate isotopic composition?
Mr(Cl) =(75.78 × 35 + 24.22 × 37)= 35.48100
What are isotopes class 9th?
Isotopes: Isotopes are atoms with the same number of protons but that have a different number of neutrons. Since the atomic number is equal to the number of protons and the atomic mass is the sum of protons and neutrons, isotopes are elements with the same atomic number but different mass numbers.What are 3 examples of isotopes?
For example, carbon-12, carbon-13, and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13, and 14, respectively. The atomic number of carbon is 6, which means that every carbon atom has 6 protons so that the neutron numbers of these isotopes are 6, 7, and 8 respectively.
What is an isotope symbol?Isotope Notation Isotopes can also be defined in standard, or “AZE”, notation where A is the mass number, Z is the atomic number, and E is the element symbol. The mass number “A” is indicated with a superscript to the left of the chemical symbol “E” while the atomic number “Z” is indicated with a subscript.
Article first time published onHow do you find the isotopic fraction?
The isotopic fractionation, ε, is defined as: ε = α − 1. This is usually a small number, hence is multiplied by 103 and is then expressed in per mil (‰). The isotopic fractionation is temperature dependent (Urey, 1947).
How do you calculate isotopes of carbon?
To find the average atomic mass, you take a certain number of atoms, find the total mass of each isotope, and then divide the total mass of all the atoms by the total number of atoms. Assume that you have, say, 10 000 atoms of carbon. Then you have 9893 atoms of 12C and 107 atoms of 13C .
What is an isotopic mixture?
Isotopic mixtures, i.e., mixtures of isotopomers (isotopomers are species that differ solely in isotopic. content, e.g., C. 6. H.
How do you find a second isotope?
However, if you have access to a periodic table, you can determine the mass number of the second isotope by examining its atomic mass. So, the key to this problem is the fact that gallium has two isotopes. This means that the percent abundances of these two isotopes must amount to 100% .
How do you find the Amu of the isotope?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
How do you find isotopic mass?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
What is meant by isotopic abundance?
the relative number of atoms of the different isotopes of one chemical element. A precise measurement of the isotopic abundance is of great importance in determining the atomic masses of elements. …
How do you calculate atomic weight?
The atomic weight of any atom can be found by multiplying the abundance of an isotope of an element by the atomic mass of the element and then adding the results together. This equation can be used with elements with two or more isotopes: Carbon-12: 0.9889 x 12.0000 = 11.8668.
Does every element have an isotope?
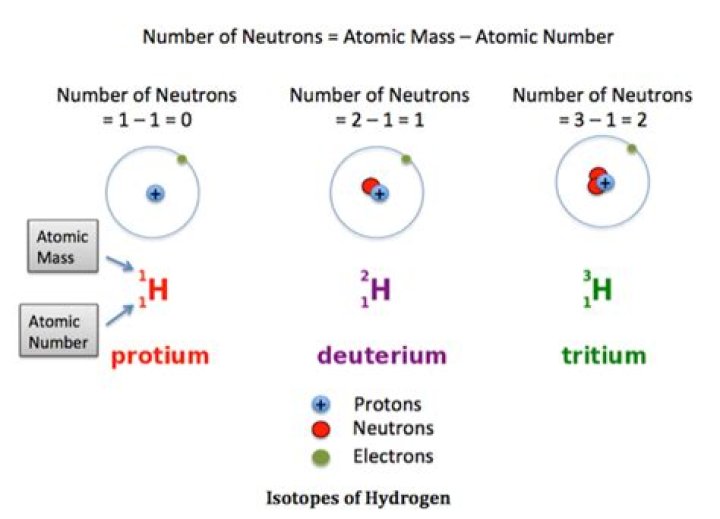
All elements have isotopes. There are two main types of isotopes: stable and unstable (radioactive). … Some elements can only exist in an unstable form (for example, uranium). Hydrogen is the only element whose isotopes have unique names: deuterium for hydrogen with one neutron and tritium for hydrogen with two neutrons.
What is an isotope that is used in everyday life?
Radioactive isotopes have many useful applications. In medicine, for example, cobalt-60 is extensively employed as a radiation source to arrest the development of cancer. Other radioactive isotopes are used as tracers for diagnostic purposes as well as in research on metabolic processes.
What are the common examples of isotopes?
Isotope Examples Carbon 12 and Carbon 14 are both isotopes of carbon, one with 6 neutrons and one with 8 neutrons (both with 6 protons). Carbon-12 is a stable isotope, while carbon-14 is a radioactive isotope (radioisotope). Uranium-235 and uranium-238 occur naturally in the Earth’s crust. Both have long half-lives.
What is an isotope in simple terms?
An isotope is one of two or more species of atoms of a chemical element with the same atomic number and position in the periodic table and nearly identical chemical behavior but with different atomic masses and physical properties. Every chemical element has one or more isotopes.
What are isotopes GCSE?
Atoms of the same element must have the same number of protons , but they can have different numbers of neutrons . Atoms of the same element with different numbers of neutrons are called isotopes .
How do you find isotopes in Class 9?
Elements having same atomic number but different atomic masses are known as Isotopes. Carbon, 136Carbon, 146Carbon, Hydrogen-1, Deuterium–2, Tritium-3 are three isotopes of hydrogen.
What are isotopes Class 11?
Isotopes are the atoms of an element which have the same number of protons but different numbers of neutrons. In other words, you can say that the isotopes have the same atomic number, as the number of protons remain the same, but they have different atomic masses due to the different number of neutrons.
What is isotope and isobar with example?
Isotopes:- Same atomic number but different mass number. Example– C−12,C−14. Isobars:- Isobars are atoms of different elements having same mass number. These have equal number of nucleons but different number of protons, neutrons and electrons.
What is Bohr diagram?
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have.
How do you find the amount of electrons in an isotope?
To find the number of electrons, add the opposite of the charge imbalance to the number of protons. For example, if an isotope has a -3 charge, as with phosphorus (atomic number 15), then the number of electrons is three greater than the number of protons.
What are isotopes Khan Academy?
Isotopes are atoms of the same element with different numbers of neutrons. … The average atomic mass of an element is calculated by taking the weighted average mass of the element’s naturally occurring isotopes. Created by Sal Khan.
How do you write atomic number in Word?
Use these shortcuts to change the format to subscript or superscript: Ctrl and the = key for subscript. Ctrl and the + key (that is, Ctrl and Shift and =) for superscript.
What is the isotope notation for carbon-14?
Hyphen NotationNuclear NotationCarbon-12 or C-12 (6 neutrons)126CCarbon-13 or C-13 (7 neutrons)136CCarbon-14 or C-14 (8 neutrons)146C



