How do you find the Bronsted acid
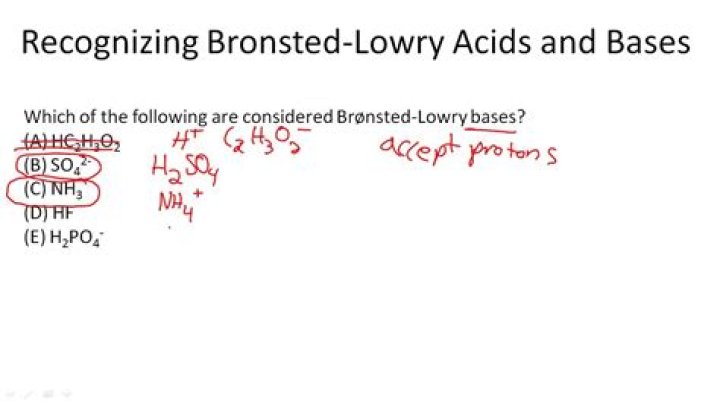
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).
How do you identify a brønsted-Lowry acid and base?
In the Brønsted–Lowry definition of acids and bases, an acid is a proton (H⁺) donor, and a base is a proton acceptor. When a Brønsted–Lowry acid loses a proton, a conjugate base is formed. Similarly, when a Brønsted–Lowry base gains a proton, a conjugate acid is formed.
Which is the Brønsted-Lowry acid?
The Brønsted-Lowry theory describes acid-base interactions in terms of proton transfer between chemical species. A Brønsted-Lowry acid is any species that can donate a proton, H+start text, H, end text, start superscript, plus, end superscript, and a base is any species that can accept a proton.
What makes a Brønsted acid?
A Brønsted-Lowry acid is any substance (molecule or ion) that can donate a hydrogen ion ( H+ ). A Brønsted-Lowry base is any species that can accept a hydrogen ion ( H+ ).What is a Brønsted-Lowry acid Example?
One of the most familiar examples of a Brønsted-Lowry acid-base reaction is between hydrochloric acid and hydroxide ion: In this reaction, a proton is transferred from HCl (the acid, or proton donor) to hydroxide ion (the base, or proton acceptor). … Chloride ion is thus the conjugate base of hydrochloric acid.
Is po4 3 a Bronsted base?
Phosphate anion, PO3−4 , is a powerful Bronsted base, and will accept protons from water to form HPO2−4 , biphosphate ion, and dihydrogenphosphate ion, H2PO−4 , along with equilibrium quantities of OH− , as is required of a Bronsted base.
How do you write a Bronsted-Lowry equation?
- Find the relative strengths of acids and bases. …
- Determine if the reaction will occur. …
- Determine whether reaction is quantitative. …
- Determine whether reactants or products are favored. …
- Determine the extent of the reaction.
Which pair is a brønsted-Lowry conjugate acid base pair?
The Brønsted–Lowry theory is an acid–base reaction theory. The fundamental concept of this theory is that when an acid and a base react with each other, the acid forms its conjugate base, and the base forms its conjugate acid by exchange of a proton. So the anwer could only be the first pair: NH3 and ammonium catione.Is HBr a Bronsted-Lowry acid?
The HBr is a Brønsted acid, and the water is a Brønsted base. In the Brønsted-Lowry theory, a conjugate base is whatever is left over after the proton has left. Thus, HBr lost a proton to become Br⁻, so Br⁻ is the conjugate base of HBr. … Thus, for example, you can easily say that the conjugate base of H₂O is OH⁻.
What is the Bronsted-Lowry definition of an acid quizlet?A Bronsted-Lowry Acid is a compound that donates a proton (H+ ion). … A Bronsted-Lowry Base is a compound that accepts a proton (H+ ion). You just studied 25 terms!
Article first time published onIs NH3 a Brønsted-Lowry acid or base?
the NH3 accepts a proton and is a Bronsted-Lowry base.
Is H+ A Brønsted-Lowry acid?
According to Brønsted-Lowry theory, the species that donates the H+ is an acid; the one that accepts the H+ is a base. … HCl donates a H+ to water and, therefore, acts as an acid. Water accepts a H+ from HCl and, therefore, acts as a base. In the reverse reaction, Cl¯ acts as a base and H3O+ acts as an acid.
Is no3 a Bronsted-Lowry acid or base?
Furthermore, because nitric acid, HNO3, loses a proton, H+1, to generate its conjugate in the reaction that is shown above, this reactant can be classified as a Brønsted-Lowry acid, and, consequently, the nitrate ion, NO3–1, is the conjugate base of this acid.
Is ch3cooh a Bronsted base?
Here, acetic acid acts as a Brønsted-Lowry acid, donating a proton to water, which acts as the Brønsted-Lowry base. The products include the acetate ion, which is the conjugate base formed in the reaction, as well as hydronium ion, which is the conjugate acid formed. Here, ammonia is the Brønsted-Lowry base.
What is an example of a Bronsted-Lowry base?
Ammonia is the Bronsted-Lowry base because it is the ‘proton acceptor’ – it accepts a hydrogen atom from water. On the other hand, water is the Bronsted-Lowry acid because it is the ‘proton donor’. … The conjugate base is the hydroxide ion (OH-) because this is the substance produced when H2O donated the proton.
Is NH3 a conjugate acid or base?
NH3 and NH4+ are a conjugate acid-base pair.
What is the Brønsted-Lowry definition of an acid Edgenuity?
What is the Bronsted-Lowry definition of an acid? a compound that donates protons. You just studied 14 terms!
What is the Brønsted-Lowry definition of an acid a compound that donates protons a compound that accepts protons?
An acid is a substance that donates protons (in the Brønsted-Lowry definition) or accepts a pair of valence electrons to form a bond (in the Lewis definition). A base is a substance that can accept protons or donate a pair of valence electrons to form a bond. Bases can be thought of as the chemical opposite of acids.
Which statement best describes a Brønsted-Lowry base?
Which statement best describes a Bronsted-Lowry base? It must accept protons to form a conjugate acid.
How is NH3 a Bronsted base?
Explanation: A Brønsted-Lowry base is a substance that behaves as a proton acceptor. The NH3 is accepting a proton from the HCl , so it is behaving as a Brønsted-Lowry base.
Why is NH3 a Bronsted-Lowry acid?
Because the water molecule donates a hydrogen ion to the ammonia, it is the Brønsted-Lowry acid, while the ammonia molecule—which accepts the hydrogen ion—is the Brønsted-Lowry base. Thus, ammonia acts as a base in both the Arrhenius sense and the Brønsted-Lowry sense.
Is h3o Bronsted-Lowry acid?
Yes, it is an Arrhenius acid. It will dissociate in water to form H+…even though it is hydronium already. Essentially, it acts as a proton donor…which makes is a Bronsted-Lowry acid too.
Is h2so4 Bronsted-Lowry acid?
Any system where hydrogen ions are transferred can be considered according to the Brønsted Lowry definition of acids and bases. … In this reaction sulfuric acid is behaving as a Brønsted-Lowry acid and the nitric acid is a Brønsted-Lowry base.
Are all Bronsted acids Lewis acids?
All Bronsted-Lowry acids are Lewis acids, but the reverse is not true. The chemist Gilbert Lewis, who invented the idea of Lewis acids and bases, actually commented on this. Lewis stated that Bronsted-Lowry acids only include species that have a hydrogen, but this is like saying that oxidizers must contain oxygen.
Is co32 a Bronsted acid or base?
[CO3](2-) is carbonate ion. It can accept a proton (H+) to form bicarbonate ion, [HCO3](-), making carbonate ion a Bronsted-Lowry base.
Is CH3 3N a Brønsted-Lowry base?
b) H2O(l) is the B-L acid; (CH3)3N(aq) is the B-L base.
Is CH3 3N a Bronsted base?
By definition, a Lewis Base is an electron pair donor, whereas a Bronsted base is a proton acceptor. Methylamine, H3CNH2 , dimethylamine, (H3C)2NH , and trimethylamine, (H3C)3N are all Bronsted bases and Lewis bases by virtue of the lone electron pair on the nitrogen centre.
Is Hcoo a Bronsted base?
HCOOH is a Brønsted-Lowry monoprotic acid, it donates a proton to form the conjugate base HCOO- (formate ion, or, methanoate ion).



