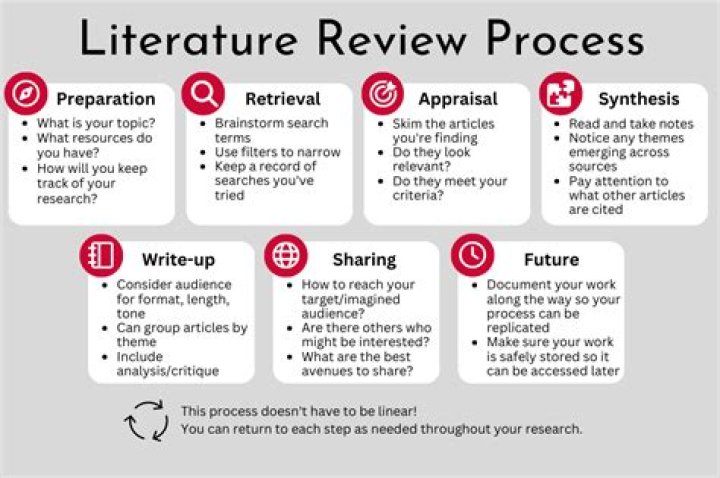
How do you find the spectral line

A spectral line is a dark or bright line in an otherwise uniform and continuous spectrum, resulting from an excess or deficiency of photons in a narrow frequency range, compared with the nearby frequencies.
What is spectral line?
A spectral line is a dark or bright line in an otherwise uniform and continuous spectrum, resulting from an excess or deficiency of photons in a narrow frequency range, compared with the nearby frequencies.
How do spectral lines work?
Spectral lines are produced by transitions of electrons within atoms or ions. As the electrons move closer to or farther from the nucleus of an atom (or of an ion), energy in the form of light (or other radiation) is emitted or absorbed.…
How do you find the spectral line width?
The spectral width caused by phase noise is commonly referred to as spectral linewidth, which is proportional to the rate of spontaneous emission and inversely proportional to the photon density: Δ ω ∝ R sp / ( 2 P ) .What is a line spectrum in physics?
Line spectra is a phenomenon which occurs when excited atoms emit light of certain wavelengths which correspond to different colours. The emitted light can be observed as a series of coloured lines with dark spaces in between. These series of coloured lines are called line or atomic spectra.
What is the line spectrum in chemistry?
1. A spectrum that consists of narrow, dark, parallel lines on a brightly colored background, produced from a hot light source surrounded by cooler gases that absorb and thus remove certain wavelengths: used to determine the chemical composition of a gas, star, etc. noun. 6.
What is an example of a line spectrum?
An example of a line spectrum is an exit sign. An example of a continuous spectrum is a clock radio.
What is linewidth in spectroscopy?
The linewidth (or line width) of a laser, e.g. a single-frequency laser, is the width (typically the full width at half-maximum, FWHM) of its optical spectrum. More precisely, it is the width of the power spectral density of the emitted electric field in terms of frequency, wavenumber or wavelength.What is linewidth in Matlab?
Line width is used to adjust (increase) the width of any object. Line width operation mostly executes inside the plot operation. Plot operation is used to plot the input and output in a graphical way. We can increase the width of an object to any extent. By default, the line width size is ‘1’ in Matlab.
What is spectral line broadening?line broadening, in spectroscopy, the spreading across a greater wavelength, or frequency range, of absorption lines (dark) or emission lines (bright) in the radiation received from some object.
Article first time published onWhat is the wavelength of the spectral line in the spectrum of hydrogen?
nλ, air (nm)∞364.6Source:
Why do elements have a number of spectral lines?
Its because, elements have electrons in orbitals. In addition there will be infinite number of empty orbitals. So when transitions occur in the atoms of an element, they absorb/release energy in the form of spectral lines.
How is spectrum formed?
When white light passes through a glass prism, it spreads out into a band of different colours called the spectrum of light. … So, the rays of each colour emerge along different paths and appear distinct. Hence, we get a spectrum of seven different colours.
Which element has the most spectral lines?
Mercury: the strongest line, at 546 nm, gives mercury a greenish color. Fig. 2. When heated in a electric discharge tube, each element produces a unique pattern of spectral `lines’.
What is the number of spectral lines produced when an electron?
The number of spectral lines that are possible when electrons in the 7th shell in different hydrogen atoms return to the second shell is 15.
What is line spectra or atomic spectra?
When atoms are excited they emit light of certain wavelengths which correspond to different colors. The emitted light can be observed as a series of colored lines with dark spaces in between; this series of colored lines is called a line or atomic spectra. Each element produces a unique set of spectral lines.
How many spectral lines are there in the hydrogen spectrum?
The electron energy level diagram for the hydrogen atom. He found that the four visible spectral lines corresponded to transitions from higher energy levels down to the second energy level (n = 2).
What is origin of line spectrum?
What is the origin of the Atomic Line spectrum ? The line emission line spectrum results from electrons dropping from higher energy level to lower energy levels. Each time an electron drops, a proton of light is released whose energy correspond to the difference in energy between the two levels.
What is continuous and line spectrum?
Continuous spectrum is a spectrum having all the wavelengths within a given limit whereas line spectrum is a spectrum having some lines of wavelengths within a given limit. Thus, continuous spectrum and line spectrum differ from each other according to the presence or absence of lines in the spectrum.
How did Niels Bohr explain spectral lines?
Niels Bohr explained the line spectrum of the hydrogen atom by assuming that the electron moved in circular orbits and that orbits with only certain radii were allowed.
How do you write Xlabel in MATLAB?
Reissuing the xlabel command replaces the old label with the new label. xlabel( target , txt ) adds the label to the specified target object. xlabel(___, Name,Value ) modifies the label appearance using one or more name-value pair arguments. For example, ‘FontSize’,12 sets the font size to 12 points.
How do you insert a LineWidth in MATLAB?
- To plot two lines with different line widths, you can use either of these approaches.
- Return the two “Line” objects as an output argument from the “plot” function and then set the “LineWidth” property for each.
- Use the “hold on” command to plot the two lines separately.
How do you name axis in MATLAB?
- title(‘Line Plot of Sine and Cosine Between -2\pi and 2\pi’)
- xlabel(‘-2\pi < x < 2\pi’) ylabel(‘Sine and Cosine Values’)
- legend({‘y = sin(x)’,’y = cos(x)’},’Location’,’southwest’)
- k = sin(pi/2); title([‘sin(\pi/2) = ‘ num2str(k)])
Why are spectral lines broadened?
Electromagnetic radiation emitted at a particular point in space can be reabsorbed as it travels through space. This absorption depends on wavelength. The line is broadened because the photons at the line center have a greater reabsorption probability than the photons at the line wings.
What is the spectral width of ILD?
ILDs are well suited for application in medium- to high-bandwidth fiber-optic communication systems. Compared to LEDs, injection lasers offer the advantage of narrower spectral width (<3 nm), larger modulation bandwidth (>500 MHz), and greater launched power (1 mW).
What are the factors on which the line width of a spectral line depends?
- Widths of Spectral Lines (continued)
- Pressure Broadening. This cause of line broadening may be regarded as being due to random collisions between atoms, which disturb internal interactions and shorten the lifetime. …
- Doppler Broadening. …
- Absorption of Radiation and Absorption Coefficients.
Why are spectral lines so thin?
Only photons with energies exactly equal to the energy difference between two energy levels can be emitted or absorbed. … The result is a natural spread of photon energies around the spectral line. The longer an excited state exists (Δt), the narrower the line width so that metastable states can have very narrow lines.
How many spectral lines are there for lithium?
Lithium spectra The three atomic emission spectra for Lithium can be shown on graph bellow: NIST Atomic Spectra Database [1], gfall.