How do you identify a strong base
The issue is similar with bases: a strong base is a base that is 100% ionized in solution. If it is less than 100% ionized in solution, it is a weak base
What are the characteristics of a strong base?
Strong bases are capable of deprotonating weak acids; very strong bases can deprotonate very weakly acidic C–H groups in the absence of water. Sodium hydroxide pelletsSodium hydroxide pellets, before being suspended in water to dissociate. Some common strong Arrhenius bases include: Potassium hydroxide (KOH)
What is strong base example?
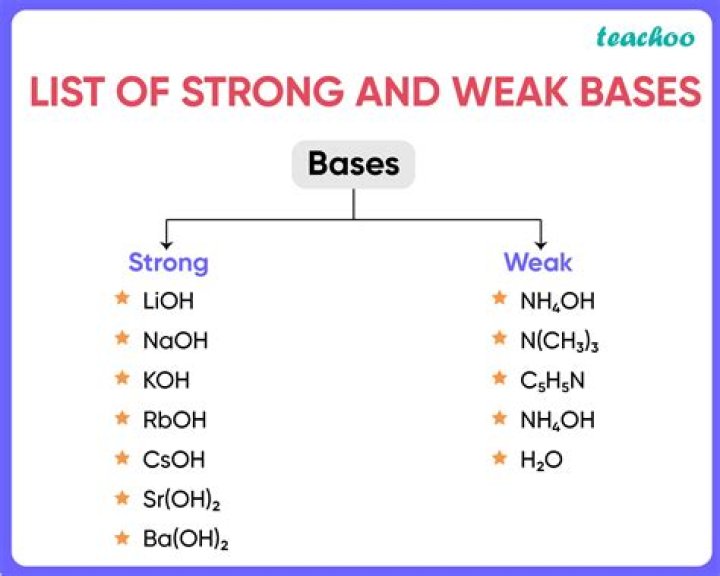
- Potassium hydroxide (KOH)
- Sodium hydroxide (NaOH)
- Barium hydroxide (Ba(OH)2)
- Caesium hydroxide (CsOH)
- Strontium hydroxide (Sr(OH)2)
- Calcium hydroxide (Ca(OH)2)
- Lithium hydroxide (LiOH)
- Rubidium hydroxide (RbOH)
How do you identify bases?
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).How does litmus paper identify a base?
How does litmus paper identify a base? A base turns red litmus blue.
How do you determine the strength of an acid?
The bond strength of an acid generally depends on the size of the ‘A’ atom: the smaller the ‘A’ atom, the stronger the H-A bond. When going down a row in the Periodic Table (see figure below), the atoms get larger so the strength of the bonds get weaker, which means the acids get stronger.
What are the characteristics of bases?
Bases taste bitter, feel slippery, and conduct electricity when dissolved in water. Indicator compounds such as litmus can be used to detect bases. Bases turn red litmus paper blue. The strength of bases is measured on the pH scale.
What are strong bases and weak bases give examples?
Sodium hydroxide, a chemical compound with the formula NaOH, is known to be a strong base. This is because sodium hydroxide undergoes almost complete ionization when it is dissolved in water. Weak bases are the basic substances that do not completely ionize in water. An example of a weak base is ammonia.What's the strongest base?
The title of strongest base in the world belongs to ortho-diethynylbenzene dianion. This superbase has the strongest proton affinity ever calculated (1843 kJ mol−1), beating out a long-standing contender known as lithium monoxide anion. Watch the video to learn more about bases and superbases!
How does litmus paper identify a base quizlet?How does litmus paper identify a base? A base turns red litmus blue. Four different solutions (I, II, III, and IV) are labeled on the pH scale below.
Article first time published onHow can you identify an acid or base without litmus paper?
In the absence of litmus paper, a reagent phenolphthalein can be used. The solution which develops pink colour with phenolphthalein is a base and the other is acid.
What color Is litmus in base?
The main use of litmus is to test whether a solution is acidic or basic, as blue litmus paper turns red under acidic conditions, and red litmus paper turns blue under basic or alkaline conditions, with the color change occurring over the pH range 4.5–8.3 at 25 °C (77 °F).
How do you remember strong bases?
- bases – barium.
- can – calcium.
- certainly – cesium.
- look – lithium hydroxide.
- pleasing – potassium hydroxide.
- really – rubidium hydroxide.
- something – sodium hydroxide.
- strong – strontium hydroxide.
Which of the given is a strong base *?
Sodium hydroxide is a strong base.
What are 5 characteristics of bases?
- Bitter taste (opposed to sour taste of acids)
- Slimy, or soapy feel on fingers (Slippery)
- Many bases react with acids and precipitate salts.
- Strong bases may react violently with acids. …
- Bases turn red litmus paper blue.
- Bases are substances that contain metal oxides or hydroxides.
What are 3 properties of bases?
- Bases change the colour of litmus from red to blue.
- They are bitter in taste.
- Bases lose their basicity when mixed with acids.
- Bases react with acids to form salt and water. …
- They can conduct electricity.
- Bases feel slippery or soapy.
- Some bases are great conductors of electricity.
How do bases feel?
A base tastes bitter, feels slippery, and turns red litmus paper blue. The properties of bases are often described as the “opposite” of acids. … Slippery Feel – Bases have a slippery feel. Many soaps and detergents contain bases.
What is the strength of a base?
Base strength of a species is its ability to accept H+ from another species (see, Brønsted-Lowry theory). The greater the ability of a species to accept a H+ from another species, the greater its base strength.
How do you determine the relative strength of an acid and a base?
The relative strengths of acids and bases are reflected in the magnitudes of their ionization constants; the stronger the acid or base, the larger its ionization constant. A reciprocal relation exists between the strengths of a conjugate acid-base pair: the stronger the acid, the weaker its conjugate base.
How does pH describe the strength of acids and bases?
The pH Scale. One qualitative measure of the strength of an acid or a base solution is the pH scaleA logarithmic scale that relates the concentration of the hydrogen ion in solution., which is based on the concentration of the hydronium (or hydrogen) ion in aqueous solution.
How can you tell the difference between strong and weak bases?
A weak base is one that only partially dissociates to give ions in solution. A strong base is one that fully dissociates to give ions in solution. Weak bases only partially dissociate in a solution, while the strong bases dissociate fully in a solution. Weak bases have pH 7.3 – 10, strong ones have pH 10 – 14.
What do you mean by strong acid and strong base?
Acids and bases that are completely ionized when dissolved in water are called strong acids and strong bases There are only a few strong acids and bases, and everyone should know their names and properties. These acids are often used in industry and everyday life.
What is the difference between a strong base and a weak base?
A strong base is a base that ionises or dissociates almost 100% in water to form OH− ion. … A weak base is a base that ionises or dissociates only partially in water to form OH− ion. An example of it will be ammonia. It only dissociates partially to form ammonium hydroxide.
What does it mean for a strong base to be in equilibrium?
What does it mean for a strong base to be in equilibrium? The position of equilibrium lies far to the right, with products being favored. … A strong acid is disassociated completely, producing more conjugate base.
What do strong acids and strong bases have in common?
The most common strong acids and bases, and some examples of weak acids and bases, together with definition of strong and weak acids and bases. A strong acid or a strong base completely ionizes (dissociates) in a solution. … Essentially, none of the non-ionized acid HA remains.
Which is a characteristics of a Lewis base?
Lewis Bases donate an electron pair. Lewis Bases are Nucleophilic meaning that they “attack” a positive charge with their lone pair. They utilize the highest occupied molecular orbital or HOMO (Figure 2). An atom, ion, or molecule with a lone-pair of electrons can thus be a Lewis base.
How will you distinguish an acid from base?
Acid is a kind of chemical compound that when dissolved in water gives a solution with H+ ion activity more than purified water. A base is an aqueous substance that donates electrons, accept protons or release hydroxide (OH-) ions. An acid is a proton donor. While a base is a proton acceptor.
How can you test an acid base at home?
Litmus is a natural acid-base indicator extracted from a type of lichen. If you have red and blue litmus paper, you can test different solutions for whether they are acids or bases. Blue litmus paper turns red when a solution is acidic; red litmus paper turns blue in basic solutions.
How do you know if something is a base or acid?
The use of the pH scale is one practical way of quantitatively determining how acidic something is. If the pH of a solution is less than 7, it is acidic. If the pH is 7, the solution is neutral and if the pH is greater than 7,the solution is basic.
Is white vinegar a base or acid?
Vinegar is acidic. Vinegar’s pH level varies based upon the type of vinegar it is. White distilled vinegar, the kind best suited for household cleaning, typically has a pH of around 2.5.
Is ammonia a base?
Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. … A weak base is one which doesn’t convert fully into hydroxide ions in solution.



