How do you make acyl chloride
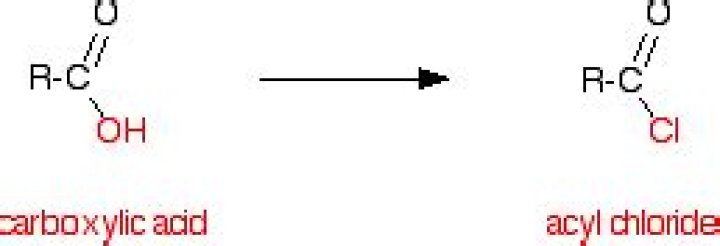
Replacing the -OH group using phosphorus(V) chloride, PCl5.Replacing the -OH group using Phosphorous(III) chloride, PCl3.Replacing the -OH group using sulfur dichloride oxide (thionyl chloride)
How is Ethanamide made?
To make ethanamide from ethanoyl chloride, you normally add the ethanoyl chloride to a concentrated solution of ammonia in water. There is a very violent reaction producing lots of white smoke – a mixture of solid ammonium chloride and ethanamide. Some of the mixture remains dissolved in water as a colorless solution.
What are carboxylic acids?
carboxylic acid, any of a class of organic compounds in which a carbon (C) atom is bonded to an oxygen (O) atom by a double bond and to a hydroxyl group (―OH) by a single bond. A fourth bond links the carbon atom to a hydrogen (H) atom or to some other univalent combining group.
What is thionyl chloride used for?
Thionyl chloride is mainly used in the industrial production of organochlorine compounds, which are often intermediates in pharmaceuticals and agrichemicals.What are acyl chlorides used for?
Acyl chlorides are used to prepare acid anhydrides, amides and esters, by reacting acid chlorides with: a salt of a carboxylic acid, an amine, or an alcohol, respectively.
How is Amino ethene obtained from Ethanamide?
Explanation: Ethanamide will be obtained from aminoethane by reduction using Clemmensen reduction. CH3-C=O. … Hcl ———————> CH3-CH2 -NH2.
Do acyl chloride react with alcohol?
Acyl chlorides, prepared by the reaction of carboxylic acids with thionyl chloride, react readily with alcohols to give ester. Pyridine is used as a base to neutralize the HCl that forms in the reaction. The reaction occurs in two steps.
Is thionyl chloride toxic?
* Thionyl Chloride is a CORROSIVE CHEMICAL and contact can severely irritate and burn the skin and eyes with possible eye damage. * Exposure to Thionyl Chloride can irritate the nose and throat. * Breathing Thionyl Chloride can irritate the lungs causing coughing and/or shortness of breath.How will you prepare Ethanamide from Ethanoic acid?
The ammonium salt is formed by adding solid ammonium carbonate to an excess of the acid. For example, ammonium ethanoate is made by adding ammonium carbonate to an excess of ethanoic acid. When the reaction is complete, the mixture is heated and the ammonium salt dehydrates producing ethanamide.
Is SOCl2 hazardous?Highly corrosive and toxic. Long-term inhalation of low concentrations or short-term inhalation of high concentrations has adverse health effects. COLOURLESS-TO-YELLOW OR REDDISH FUMING LIQUID WITH PUNGENT ODOUR. Colorless to yellow to reddish liquid with a pungent odor like sulfur dioxide.
Article first time published onDoes thionyl chloride react with alcohol?
Mechanism of the Reaction of Alcohols with Thionyl Chloride First, a nucleophilic oxygen atom of the alcohol displaces a chloride ion from thionyl chloride to form a protonated alkyl chlorosulfite intermediate.
What is a carboxylic acid give an example?
Carboxylic acidpKaAcetic acid (CH3CO2H)4.76Benzoic acid (C6H5CO2H)4.2Formic acid (HCOOH)3.75Chloroacetic acid (CH2ClCO2H)2.86
Where are carboxylic acids found?
Many carboxylic acids occur naturally in plants and animals. Citrus fruits such as oranges and lemons contain citric acid. Citric acid is a large carboxylic acid with three ionizable hydrogen atoms. It is found in citrus fruits and gives them their sour or tart flavor.
How carboxylic acids are formed?
The hydrolysis of nitriles, which are organic molecules containing a cyano group, leads to carboxylic acid formation. These hydrolysis reactions can take place in either acidic or basic solutions. The mechanism for these reactions involves the formation of an amide followed by hydrolysis of the amide to the acid.
Are acyl chlorides toxic?
ICSC 0210 – ACETYL CHLORIDE. Highly flammable. Many reactions may cause fire or explosion. Gives off irritating or toxic fumes (or gases) in a fire.
Why are acyl chlorides so reactive?
Acyl chlorides are the most reactive carboxylic acid derivatives. The electronegative chlorine atom pulls electrons toward it in the C-Cl bond, which makes the carbonyl carbon more electrophilic. This makes nucleophilic attack easier. Also, the Cl- is an excellent leaving group, so that step is also fast.
What do acyl chlorides react with?
Acid chlorides react with carboxylic acids to form anhydrides. … Acid chlorides react with ammonia, 1o amines and 2o amines to form amides.
What is the best leaving group in a nucleophilic acyl substitution?
20.10 Conversion of Carboxylic Acids into Acyl Halides First, the competing reaction of nucleophiles with the acidic proton of carboxylic acids is eliminated. 2. Second, a chloride ion is a better leaving group than a hydroxide ion.
What reacts with water to produce misty fumes?
The reaction with water Adding an acyl chloride to water produces the corresponding carboxylic acid together with steamy acidic fumes of hydrogen chloride. The reaction is usually extremely vigorous at room temperature.
How does acyl chloride turn into alcohol?
If you are interested in another alcohol, you can replace the CH3CH2 group by any other alkyl group. Ethanoyl chloride reacts instantly with cold ethanol. There is a very exothermic reaction in which a steamy acidic gas is given off (hydrogen chloride). Ethyl ethanoate (an ester) is formed.
How will you convert acetamide to methylamine?
Acetamide changes into methylamine by hoffman bromamide reaction.
Which exist as a Zwitter ion?
All Amino acids exist as Zwitter ions.
How is ethylamine prepared from acetamide?
Ethylamine is produced when acetamide is reduced using LiAlH4 or sodium and alcohol. Acetamide is reduced using sodium and absolute alcohol or LiAlH4 in ether or hydrogen in the presence of a nickel catalyst to produce ethylamine.
What are secondary amides?
Secondary amide (2o amide): An amide in which the nitrogen atom is directly bonded to two carbon atoms: the carbonyl group carbon plus one other carbon.
Is acetamide polar or nonpolar?
Information on this page: Normal alkane RI, non-polar column, temperature ramp.
How do you get amide from Amine?
Compounds that have a nitrogen atom bonded to one side of a carbonyl group are classified as amides. Amines are a basic functional group. Amines and carboxylic acids can combine in a condensation reaction to form amides.
How are amides produced?
In an analogous reaction, an amide can be prepared through the reaction of a carboxylic acid and an amine using a coupling agent such as DCC. Simple amides can be prepared by reacting an acid anhydride with an amine. Lastly, amides can be formed through the direct reaction of a carboxylic acid and an amine.
How do amides differ from amines?
Amines and amides are two types of compounds found in the field of organic chemistry. … The main difference between amine and amide is the presence of a carbonyl group in their structure; amines have no carbonyl groups attached to the nitrogen atom whereas amides have a carbonyl group attached to a nitrogen atom.
Are amines and amides the same?
Compounds containing a nitrogen atom bonded in a hydrocarbon framework are classified as amines. Compounds that have a nitrogen atom bonded to one side of a carbonyl group are classified as amides.
Why does thionyl chloride and water explode?
Emits dense corrosive fumes in moist air. Violently reacts with water to liberate hydrochloric acid and sulfur dioxide [NFPA 10th ed. … Thionyl chloride reacts vigorously with water to generate gaseous HCl and SO2.
How do you neutralize SOCl2?
Add Sodium Bicarbonate to neutralize excess thionyl chloride.