How do you make cyclohexanone
The synthesis of cyclohexanone is simple. First, sodium hypochlorite and acetic acid are reacted to yield hypochlorous acid. Second, hypochlorous acid is added to cyclohexanol to synthesize cyclohexanone via Chapman-Stevens oxidation reaction.
How is cyclohexanone formed?
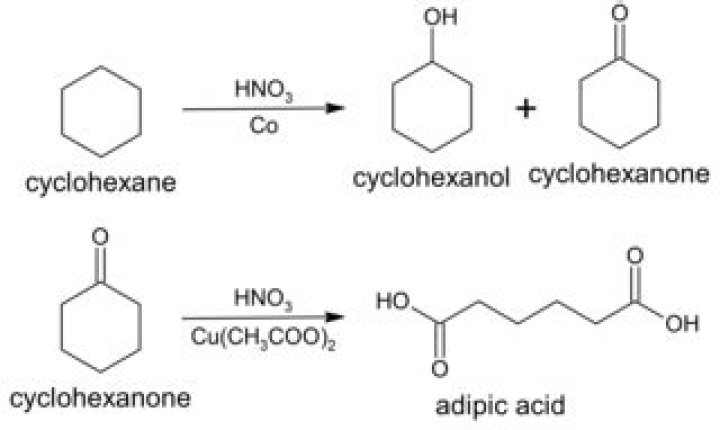
Production. Cyclohexanone is produced by the oxidation of cyclohexane in air, typically using cobalt catalysts: C6H12 + O2 → (CH2)5CO + H2O. This process co-forms cyclohexanol, and this mixture, called “KA Oil” for ketone-alcohol oil, is the main feedstock for the production of adipic acid.
How do you convert Cyclohexan to cyclohexanone?
The formation of radical from hydrogen peroxide as oxidant was important step for start the oxidation of cyclohexane. Catalyst H4[α-SiW12O40]/TiO2 can convert cyclohexane to cyclohexanol and cyclohexanone under mild conditions at 2 h reaction time, 70 °C, 0.038 g of catalyst, and 3 mL hydrogen peroxide as oxidant.
How do you make cyclohexanone from cyclohexanol?
Combine the ethyl acetate extracts with the cyclohexanone layer. Dry with anhydrous magnesium sulfate (5 min) and filter into a pre-weighed dry 50 mL RB flask. Distil off the ethyl acetate, b.p. 77 C, using a distillation apparatus set up on a steam bath. This leaves the cyclohexanone (b.p. 153 – 156 C) in the flask.What reagent is needed for cyclohexanol?
Cyclohexanol is secondary alcohol . Hence , cyclohexanol can be converted to cyclohexanone using PCC.
Is cyclohexanone a ketone or aldehyde?
Cyclohexanone is a cyclic ketone that consists of cyclohexane bearing a single oxo substituent.
Is cyclohexanone a solvent?
The remainder is used as a chemical intermediate in other processes, as an additive or as a high-boiling, slow-drying solvent. Cyclohexanone is used as a solvent in insecticides, wood stains, paint and varnish removers, spot removers, cellulosics, and natural and synthetic resins and lacquers.
How do you convert cyclohexanol to cyclohexene?
One molecule of cyclohexanol should produce one molecule of cyclohexene. One mole (mol) of cyclohexanol should produce one mole of cyclohexene. If 2.05 g of cyclohexanol is used (use the actual amount used in your experiment) convert this to moles by dividing by the molecular weight of cyclohexanol (MW = 100.2 g/mol).How do you oxidize cyclohexanol?
Primary alcohols may be oxidized to aldehydes, and then to carboxylic acids. Secondary alcohols may be oxidized to ketones, while tertiary alcohols will not oxidize. This exercise will oxidize cyclohexanol to cyclohexanone using and acidic solution of sodium hypochlorite.
How is cyclohexanol prepared from phenol?Phenol is reacted with hydrogen in contact with a nickel catalyst, wherein the concentration of hydrogen is controlled, and/or wherein controlled amounts of water are included in the reaction mixture, to produce cyclohexanol, or preferably cyclohexanol plus cyclohexanone.
Article first time published onWhat is the product of dehydration of cyclohexanol?
In this experiment, cyclohexanol is dehydrated by aqueous sulfuric acid to produce cyclohexene as the sole product [equation (1)], and no rearrangement is possible in this reaction.
What is the best reagent to convert cyclohexanol to cyclohexene?
Concentrated phosphoric acid is a good dehydrating agent which converts an alcohol to an alkene by promoting the reaction but not consuming it while other options given are all nucleophiles, which will convert alcohols to alkyl halides. So, the correct answer is “Option C”.
How do you convert phenol to cyclohexene?
To convert (b) Phenol to Cyclohexane: Phenol is treated with hydrogen gas in presence of Palladium to give Cyclohexanone. This intermediate product is further treated with hydrazine in presence of hydroxide ions to give Cyclohexane. And the Phenol can be converted into Cyclohexane.
Which of the following reagents would be to convert cyclohexanol to Chlorocyclohexane?
Complete step-by-step answer:When cyclohexanol is reacted with thionyl chloride, it results in the formation of chlorocyclohexane.
Which is the product formed when cyclohexanone?
When cyclohexanone undergoes aldol condensation in the presence of a base, it will produce a β− hydroxy ketone.
What is cyclohexanone for?
Cyclohexanone is used as a solvent for lacquers, paints, resins, degreasers, spot removers, polymers, copolymers, waxes, crude rubber, cellulose acetate, the manufacturing of herbicides and anihistamines.
How do you neutralize cyclohexanone?
Dissolve the solid with water, neutralize the solution with an aqueous solution of sodium hydroxide (5% solution), and extract with dichloromethane.
Why is cyclohexanone a liquid?
Cyclohexane is a liquid at room temperature because its molecules do not have enough energy to escape into the vapour phase. … These are weak forces, but there are enough of them so that at room temperature the molecules are attracted to each other strongly enough to make cyclohexane a liquid.
What is cyclohexanone derivative?
Cyclohexanones can be also transformed to ε-caprolactones under the action of recombinant baker’s yeast <1998JA3541> and lipases <1998J(P1)2625>, the latter process being an autocatalytic Baeyer–Villiger oxidation with urea–hydrogen peroxide as the primary oxidant.
Is cyclohexanone a primary alcohol?
Cyclohexanol is a secondary alcohol because the −OH group is attached to a secondary carbon if the ring.
How does bleach oxidize an alcohol?
The Reaction and Conditions In general, an alcohol reacts with sodium hypochlorite in the presence of acetic acid to give a ketone as the final product. … When cyclohexanol is exposed to sodium hypochlorite (NaOCl) and acetic acid, an oxidation reaction takes place that gives cyclohexanone as the product.
How do ketones turn into alcohol?
A secondary alcohol can be oxidised into a ketone using acidified potassium dichromate and heating under reflux.
Can ketones be oxidized?
Oxidation of Ketones Because ketones do not have hydrogen atom attached to their carbonyl, they are resistant to oxidation. Only very strong oxidizing agents such as potassium manganate(VII) (potassium permanganate) solution oxidize ketones.
How do you make an alkene from an alcohol?
The dehydration reaction of alcohols to generate alkene proceeds by heating the alcohols in the presence of a strong acid, such as sulfuric or phosphoric acid, at high temperatures.
How do you calculate theoretical yield of cyclohexene?
Convert this number of moles of cyclohexene to grams of cyclohexene by multiplying by the MW of cyclohexene (82.1 g/mol). In other words, 2.05 g of cyclohexanol should produce 1.68 g of cyclohexene. This is the best-case yield also known as the theoretical yield.
How will you obtain cyclohexane from phenol?
Introduction. Cyclohexanone is mainly obtained by cyclohexane oxidation in the liquid phase. In this process, cyclohexanol is generated in quantities almost equal to that of cyclohexanone. Therefore, catalytic dehydrogenation of cyclohexanol in the gas phase to obtain cyclohexanone must be conducted.
How do you turn benzene into cyclohexane?
Benzene undergo catalytic hydrogenation in presence of platinum to give cyclohexane.
Which is the best method to prepare cyclohexene from cyclohexanol?
The best method to prepare cyclohexene from cyclohexanol is by conc. H3PO4 because in given options dehydrating agent is conc. H3PO4.
Why is cyclohexanol to cyclohexene e1?
If cyclohexanol is heated with a catalytic amount of phosphoric acid, elimination of water (dehydration) results in cyclohexene as the product. The role of the phosphoric acid is to protonate the alcohol (‘step a’ below), making it a viable leaving group.
What should you do if you spill sulfuric acid on your hand quizlet?
What should you do if you spill sulfuric acid on your hand? Wash it with cold water and apply 0.6 M sodium bicarbonate solution.
Is cyclohexanol toxic?
Cyclohexanol appears as a colorless liquid with a camphor-like odor. Soluble in most organic liquids. Flash point 154°F. May be toxic by inhalation or skin exposure.