How do you make isopentyl acetate
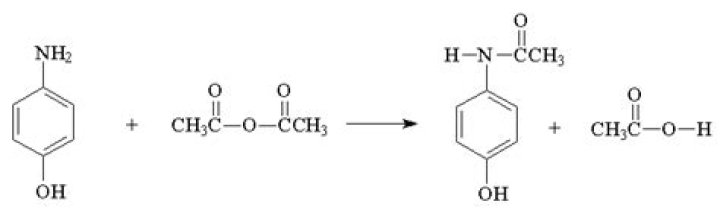
1) Mix isopentyl alcohol (5.4 mL, via burette) and glacial acetic acid (8.5 mL, via graduated cylinder) in a round-bottom flask. Carefully add 20 drops of conc. H2SO4 to the mixture. 2) Put in a few boiling chips and assemble the reflux apparatus (Figure 1).
How is isoamyl acetate formed?
Isoamyl acetate is produced from a reaction between amyl alcohol and acetyl coenzyme A catalyzed by the enzyme isoamyl alcohol acetyl transfer- ase11.
How do you synthesize banana oil?
Add several boiling chips and distill using a sand bath, electric mantel or flame. Cool the receiver flask in an ice bath. Collect the fraction that distills between 135°C and 143°C. The clear colorless product has an intense odor of bananas; it should be obtained in 80-90% yield.
Can the synthesis of isoamyl acetate be carried out by simple distillation?
A simple distillation is performed to collect the isoamyl acetate. The flask is heated in using a heating manifold. The isopentyl acetate will boil and condense and will be collected. This distillation will significantly purify the material, but not if there is alcohol or water present.How do you make isobutyl acetate?
A common method for preparing isobutyl acetate is Fischer esterification, where precursors isobutyl alcohol and acetic acid are heated in the presence of a strong acid. Isobutyl acetate has three isomers: n-butyl acetate, tert-butyl acetate, and sec-butyl acetate, which are also common solvents.
How is isoamyl alcohol prepared commercially?
Isoamyl alcohol can be synthesized by condensation of isobutene and formaldehyde which produces isoprenol and hydrogenation. It is a colourless liquid of density 0.8247 g/cm3 (0 °C), boiling at 131.6 °C, slightly soluble in water, and easily dissolved in organic solvents.
Why is sodium bicarbonate used to extract isopentyl acetate?
Introduction: The purpose of this experiment is to prepare isopentyl acetate by direct esterification of acetic acid with isopentyl alcohol. The mixture is extracted with sodium bicarbonate because doing so will cause any unreacted acid to react with the sodium bicarbonate to become neutralized. …
What is isoamyl acetate in?
Isoamyl acetate is used for the preparation of many synthetic flavours from apple to piña colada and even coffee. Flavour production is the main area of application for Isoamyl acetate. … Isoamyl acetate is also used in cosmetics, primarily in nail varnish application and removal because of its good solvency.What functional group is in isoamyl acetate?
Isopentyl acetate belongs to the family of Carboxylic Acid Esters. These are carboxylic acid derivatives in which the carbo atom from the carbonyl group is atached to an alkyl or oaryl moiety through an oxygen atom (forming an ester group).
How do you isolate isoamyl acetate?Isolation of Isoamyl Acetate Slowly add 1.0 mL of an aqueous 5% sodium bicarbonate solution to the organic layer. Stir the contents of the vial, and once the bubbling slows, cap the vial and shake gently. Remember to vent while shaking. Separate the aqueous layer from the organic layer.
Article first time published onIs isoamyl acetate soluble in water?
SYNONYMSAmyl acetate, isoamyl ethanoateCHARACTERISTICSIDENTIFICATIONSolubilitySlightly soluble in water, insoluble in glycerol, practically insoluble in propylene glycol, soluble in ethanol, diethyl ether, ethyl acetate, most fixed oils and mineral oilsRefractive index: 1.400-1.404
What is isopropyl acetate used for?
Isopropyl Acetate is a colorless liquid with a fruity odor. It is used as a solvent for cellulose, plastics, oils and fats, and in printing inks and perfume.
Can I make banana oil?
Step 1 – Cut the banana peels into pieces and place them in a plastic bag. Step 2 – Beat, crush and mash the banana peels until they are softened. Step 3 – Move banana peels to a glass jar and add the almond oil (olive oil also works). Step 4 – Seal the jar and put it in a sunny place for 2-3 days.
What is the limiting reagent in synthesis of isopentyl acetate?
In the experiment, the acetic acid was in excess and the isopentyl alcohol was the limiting reagent, therefore, the reaction depended on the amount of isopentyl alcohol available.
How do you synthesize isobutyl formate?
NamesChEBICHEBI:173348ChemSpider10492ECHA InfoCard100.008.017EC Number208-818-1
What is isobutyl acetate used for?
Isobutyl Acetate is a clear, colorless liquid with a pleasant, fruity odor. It is used as a solvent and in perfumes and artificial fruit-flavoring agents.
Is isopentyl acetate more polar than isopentyl alcohol?
Retention time is determined principally by polarity. For example, isopentyl alcohol is more polar than isopentyl acetate, and is more strongly attracted to polar sites on the stationary phase. it elutes after isopentyl acetate even though it has a lower BP.
How many grams are there in 1.00 mL of isopentyl acetate?
The volume of isopentyl alcohol taken is 1.00 mL. The mass of isopentyl acetate obtained is 1.00 g.
How are alcohols manufactured commercially?
How are alcohols manufactured commercially? Ans: Alcohol can be manufactured commercially by fermentation of starch or sugar. In the fermentation process complex, organic compounds are broken down into alcohol by biological enzymes like invertase, zymase, etc.
How will you synthesize methyl alcohol from ethyl alcohol?
You need to first convert ethanol into acetic acid through oxidation followed by treatment with ammonia which will give ethanamide. The Hoffman bromamide degradation of ethanamide will yield methyl amine which can be transformed into methanol by treating with nitrous acid.
How can alcohol be prepared commercially?
There are two main processes for the manufacture of ethanol: the fermentation of carbohydrates (the method used for alcoholic beverages) and the hydration of ethylene. Fermentation involves the transformation of carbohydrates to ethanol by growing yeast cells.
What is the correct empirical formula for isopentyl acetate?
Molecular FormulaC7H14O2Molar Mass130.185 g/molDensity0.879g/cm3Melting Point-78℃Boling Point142.1°C at 760 mmHg
Where is isoamyl acetate found naturally?
Isoamyl acetate is found naturally in the banana plant. In addition to its role as a flavoring or aroma agent in foods and perfumes, isoamyl acetate is released by a honey bee’s sting where it serves as a pheromone to attract other bees and provoke them to sting ( PMID: 13870346).
How many single bonds does isoamyl acetate have?
Chemical Structure Description The ISOAMYL ACETATE molecule contains a total of 22 bond(s) There are 8 non-H bond(s), 1 multiple bond(s), 4 rotatable bond(s), 1 double bond(s) and 1 ester(s) (aliphatic).
What causes isoamyl acetate in beer?
Isoamyl Acetate, a key ester (combination of an acid and an alcohol) present in all beers. … As with other esters, it is produced by yeast during fermentation and has a major flavor impact in certain beer styles, particularly Bavarian-style wheat beers. In general it contributes to the fruity qualities of beer.
What does benzyl acetate smell like?
Powerful but thin, sweet floral fresh, fresh and light, fruity odor reminiscent of Jasmin, Gardenia, Muguet, Lily and other flowers.
What is the purpose of extracting with 25 mL of water?
What is the purpose of extracting with 25 mL of water? To remove any excess acids/bases or salts that may still be in the organic layer. CONCLUSION Isopentyl acetate was prepared from an esterification reaction of acetic acid and isopentyl alcohol.
What is another name for isoamyl acetate?
Synonyms. ISOAMYL ACETATE. 3-Methylbutyl acetate. Isopentyl acetate. 123-92-2.
Is isoamyl acetate expected to be a yellow liquid?
Isoamyl Acetate is a strongly fruity-smelling liquid and has been identified in many fruit aromas. It is the main component of banana aroma and is, therefore, also used in banana flavors. All isomers of amyl acetate are highly flammable, colorless to yellow, watery liquids.
What does amyl acetate smell like?
n-Amyl Acetate is a colorless liquid with a banana-like odor. It is widely used as a solvent and in the manufacture of artificial fruit-flavoring agents.
Why should you not smell banana oil?
* Breathing Isoamyl Acetate can irritate the nose, throat and lungs. * Exposure to high concentrations of Isoamyl Acetate can cause headache, drowsiness, dizziness, lightheadedness, fatigue, and may cause you to pass out.