How do you make salt in chemistry
Add some dilute hydrochloric acid to a beaker.Add powdered zinc carbonate to some acid, one spatula at a time, stirring to mix. … Continue adding powder until it is in excess (some unreacted powder is left over).Filter the mixture in the beaker to remove the excess zinc carbonate.
How salts are formed give an example?
Normal Salt: A normal salt is a salt formed by the complete replacement of replaceable hydrogen atoms from an acid molecule by means of a metal or a group of elements acting like a metal. Examples: The compounds like KCl, NaCl, FeS04, Na2S04, FeCl2 etc are normal salts.
How soluble salts are made?
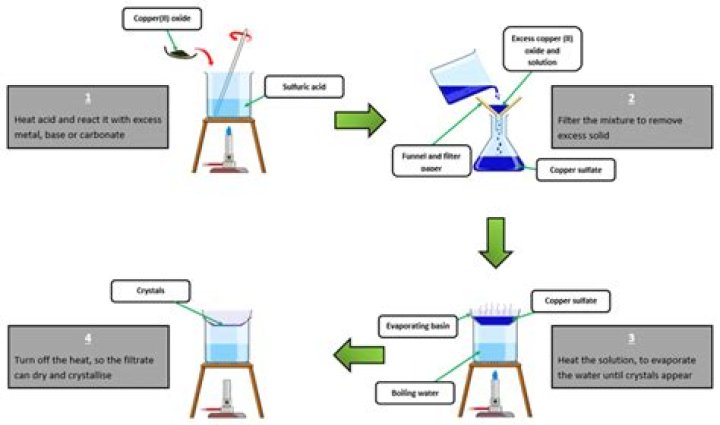
A soluble salt can be prepared by reacting an acid with a suitable insoluble reactant including: The insoluble reactant chosen depends upon the particular salt required. For example, copper does not react with dilute acids, so copper salts are made using copper oxide or copper carbonate, not copper metal.
How are acid salts prepared?
Acid salts are the converse of basic salts; they are formed in the neutralization reaction between a strong acid and a weak base. The conjugate acid of the weak base makes the salt acidic.How is salt formed explain in brief?
In chemistry, salt is produced when acids and bases are combined together in equal proportions. Whereas naturally, salt is found in sea water. The process where acids and bases neutralize each other to form salt and water is known as neutralization. The chemical formula of common salt is NaCl.
What are the 3 methods of salt preparation?
- Precipitation method. – to produce INSOLUBLE salts. – Reagents: (aqueous) + (aqueous) …
- Excess/ICA method. – to produce SOLUBLE salts. – Reagents: (aqueous; usually an acid) + (solid; excess METAL/ METAL OXIDES/ METAL HYDROXIDES/ METAL CARBONATES) …
- TITRATION method.
How many types of salts are formed?
The different types of salts are: normal salt, acid salt, basic salt and double salt. 1. Normal salt : A salt that does not contain any replaceable hydrogen atoms or hydroxyl groups is called normal salt.
How are soluble salts prepared in the laboratory?
- Making a salt from an alkali. A solution is placed in an evaporating basin and heated with a Bunsen burner.
- The amount of the solution has reduced through evaporation.
- The solution has evaporated, leaving a crystallised solute.
How would you prepare salt in the laboratory?
Salts are prepared by reacting an acid with a metal or a base, such as a metal carbonate, hydroxide or oxide. The acid provides the non-metal ion for the salt, e.g. chloride or sulphate or nitrate ions.
How are hydrochloride salts made?Hydrochloride (hydrochloride salt): A salt consisting of a chloride anion with the conjugate acid cation of a base, usually an amine. The generic structure is B-H+ Cl-. Pyridinium hydrochloride, a hydrochloride salt containing a pyridinium cation, can be formed by the reaction of pyridine with hydrogen chloride gas.
Article first time published onHow do you make salts from metals and acids?
- Acids will react with reactive metals, such as magnesium and zinc, to make a salt and hydrogen.
- acid + metal → salt + hydrogen.
- hydrochloric acid + zinc → zinc chloride + hydrogen.
How are salts formed Class 7?
A salt is formed when a metal displaces hydrogen from an acid.
What are 5 examples of salts?
Salts are ionic compounds which, when dissolved in water, break up completely into ions. They arise by the reaction of acids with bases, and they always contain either a metal cation or a cation derived from ammonium (NH4+). Examples of salts include NaCl, NH4F, MgCO3, and Fe2(HPO4)3.
What are three types of salts how are they formed give one example of each?
Three types of salts are neutral, acidic and basic salts. They are formed by neutralized strong acids and strong bases. Sodium chloride is a neutral salt that is produced by neutralized hydrochloric acid and sodium hydroxide. When a strong acid is neutralize with a strong base, the result is invariably a neutral salt.
How is a salt formed Class 10?
When a strong acid reacts with a weak base, the base is unable to fully neutralize the acid. Due to this, an acidic salt is formed. When a strong base reacts with a weak acid, the acid is unable to fully neutralize the base. Due to this, a basic salt is formed.
What are the two methods by which salts can be prepared?
- Reaction 1: Acid + alkali → salt + water.
- Reaction 2: Acid + metal → salt + hydrogen.
- Reaction 3: Acid + base → salt + water.
- Reaction 4: Acid + metal carbonate → salt + water + carbon dioxide.
How is seawater salt prepared?
Salt from sea water is obtained by the process of evaporation. … The sea water evaporates due to the sun’s heat and solid salt is left behind. The left salt is collected and refined to get purified salt. Hence, salt is obtained from sea water by evaporation.
Which salts can be prepared by crystallization?
Of the three salts prepared, the Na, K, and Li salts, only the Na salt is obtained with 1 molecule of water of crystallization, which is given off at 78” in vacua. The K salt, on the other hand, is distinguished by its deliquescent nature.
How are insoluble salts prepared?
To make an insoluble salt, two soluble salts need to react together in a precipitation reaction .
How do you make hydrochloride?
Hydrogen chloride gas can be produced by combining hydrogen gas and chlorine gas. Another way is to bubble chlorine, Cl2, gas into water. It will produce a mixture of hydrochloric acid and hypochlorous acid.
How is hydrochloride made?
In chemistry, a hydrochloride is an acid salt resulting, or regarded as resulting, from the reaction of hydrochloric acid with an organic base (e.g. an amine). An alternative name is chlorhydrate, which comes from French.
How do you precipitate salt?
A precipitation reaction can occur when two solutions containing different salts are mixed, and a cation/anion pair in the resulting combined solution forms an insoluble salt; this salt then precipitates out of solution.
Which salts are prepared by neutralization?
Hydrochloric acidSulfuric acidPotassium hydroxidePotassium chloridePotassium sulfateSodium hydroxideSodium chlorideSodium sulfateCalcium hydroxideCalcium chlorideCalcium sulfate
Which salts can be prepared by precipitation?
- You can prepare the insoluble salts silver bromide and silver iodide in a similar way by precipitation.
- You can make silver bromide by mixing solutions of silver nitrate and potassium bromide.
What are salts made from citric acid called?
After the mold is filtered out of the resulting solution, citric acid is isolated by precipitating it with calcium hydroxide to yield calcium citrate salt, from which citric acid is regenerated by treatment with sulfuric acid, as in the direct extraction from citrus fruit juice.
How do you make a soluble salt from an acid and an alkali?
A soluble salt can be prepared by reacting an acid with a soluble reactant . This is usually a dilute solution of an alkali such as sodium hydroxide or ammonia.
What acid is used to produce chloride salts?
Neutralising hydrochloric acid produces chloride salts.
What is a salt Class 7?
Neutralization is a process or a chemical reaction in which an acidic and basic substance is mixed with each other in order to neutralize their acidic and alkaline nature. The product that is formed after the neutralization process is called a Salt. The salt can have basic, acidic or neutral nature.
What is salt in Chemistry for Class 7?
A salt is formed when an acid and a base react. The reaction in which acids react with bases resulting in the formation of salt and water are called neutralization reactions. Types of salts: A salt can be acidic basic or neutral.
What are salts give examples Class 7?
Ans: Salts are the ionic compounds generally formed by neutralisation of an acid with base. They can be acidic, basic as well as neutral. Example acidic salts: sodium bicarbonate, basic salts: magnesium chloride, neutral salt: sodium chloride.


