How do you synthesize Acetanilide
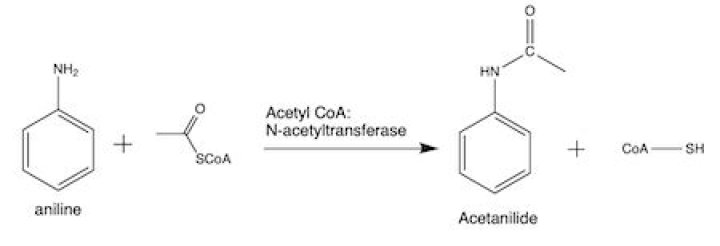
Acetanilide is synthesized from aniline by acetylating it with acetic anhydride in presence of glacial acetic acid
How do you synthesis acetanilide?
Acetanilide is prepared from aniline when it reacts with acetic anhydride/glacial acetic acid in the presence of zinc dust. A mixture of aniline, glacial acetic acid, acetic anhydride and zinc dust is refluxed under anhydrous condition and then poured the mixture into ice cold water to get acetic anhydride precipitate.
What happens when you heat acetanilide?
The solubilities of most solids in solution increase as the temperature of the solution increases. … But the solubility of acetanilide in ethanol increases to about 80g/100mL at 50 ºC. This means that if we heated up the same acetanilide-ethanol suspension to 50 ºC, all of the acetanilide would dissolve.
Where does acetanilide come from?
Acetanilide is commonly synthesized by reacting aniline with either acetyl chloride or acetic anhydride, with acetic anhydride being preferred over acetyl chloride due to corrosiveness and toxicity concerns.What is the theoretical melting point of acetanilide?
Physical-chemical properties of acetanilide are as follows: melting point 113.7 ºC, boiling point 304 ºC at 760 mmHg, water solubility 4 g/L at 20 ºC, Log Pow 1.16 at 23 ºC.
Which reagent is used for nitration of acetanilide?
Theory: The organic compound p-nitroacetanilide is prepared from acetanilide through nitration. When acetanilide is treated with nitrating mixture that is a mixture of nitric acid and sulphuric acid p-nitroacetanilide is formed.
Which type of reaction is involved in synthesis of acetanilide?
Aniline reacts with acetic anhydride to form Acetanilide by nucleophilic substitution reaction and the reaction is called acetylation.
Why is Acetanilide not used as a drug anymore?
In the body acetanilide is mostly converted to acetaminophen, which has replaced acetanilide in therapy because it is less likely to induce blood disorders.Why was Acetanilide discontinued?
Acetanilide was used in the 19th century to treat fever and headaches but was discontinued because of toxic side effects.
Why is Acetanilide not used as a drug?Unfortunately, Acetanilide exhibited an unacceptable profile of toxic effects, the most alarming being cyanosis due to methemoglobinemia. … Acetanilide is no longer used as a drug in its own right, although its primary metabolite, paracetamol, has been widely succesful.
Article first time published onWhat is the best solvent for acetanilide?
Water is the best solvent to crystallize acetanilide because water is the cheapest solvent and its solubility in water at 100°C is ten times its solubility at 25°C. So dissolve acetanilide in boiling water and cool the solution to room temperature when acetanilide crystallizes out.
What product is formed in the bromination of acetanilide?
Bromination of acetanilide gives para brominated acetanilide mainly, because amino group of acetanilide is protected by acetyl group. Reaction: Mechanism: Bromination is an electrophilic substitution reaction on an aromatic ring.
How do you purify acetanilide?
When possible and practical, solids are purified via recrystallization or sublimation and liquids via distillation. In this experiment, students will purify crude acetanilide from water (Scheme 1). The purity of the crude and recrystallized acetanilide will be assessed by melting point.
How is acetanilide metabolized?
The route of metabolism of acetanilide in man was shown to be as follows: a minor fraction of the drug deacetylates to form aniline; this compound was shown to be the precursor of the substance which oxidizes hemoglobin to methemoglobin; the major fraction of the drug is oxidized to N-acetyl p-aminophenol; this …
Does water react with acetanilide?
Acetanilide has a much higher solubility in hot water than in cold water. … A successful recrystallization requires that the compound be soluble at the solvent’s boiling point and insoluble at low temperature.
What is the literature value for the melting point of acetanilide provide your reference?
113-115 °C (lit.)
Is acetanilide electron withdrawing or donating?
Amide group in acetanilide is donating group to benzene, and the amide group in N-methylbenzamide is electron withdrawing group.
What is the pH of acetanilide?
Acetanilide for synthesis (Merck) containing more than 99% of the pure compound was used for the preparation of the initial solution (pH = 7.0).
What metabolite of acetanilide has replaced it as an over the counter pain killing drug?
Thus, Acetanilide has largely been replaced by less toxic drugs, in particular acetaminophen (i.e. paracetamol), which is a metabolite of Acetanilide and whose use Axelrod and Brodie suggested in the same study (8).
What is the role of glacial acetic acid in the preparation of P Bromoacetanilide from acetanilide?
Glacial acetic acid is a polar protic solvent that can dissolve acetanilide. In acetic acid, bromine is ionized to bromonium ion to a lesser extent as…
How do you calculate theoretical yield of acetanilide?
- 10 g of aniline shall yield acetanilide = 135.16 / 93 × 10 = 14.5 g.
- Then, Percentage Practical yield = Practical yield / Theoretical yield × 100.
- = 12 / 14.5 × 100 = 82.75 %
Why acid chloride is added in a lot during preparation of acetanilide?
Answer: Preparation of acetanilide from aniline and acetyl chloride : The nucleophilic substitution of aromatic amines with acid chloride results in the formation of N-substituted amides. … Zinc is added to prevent the oxidation of aniline during the reaction. It reduces the coloured impurities present in the solution.
Which product is produced in major quantity on nitration of acetanilide?
Acetanilide also exhibits reagent-dependent orientation in nitration; nitric and sulfuric acids yield paranitroacetanilide as major product, while with nitric acid – acetic anhydride, ortho- nitration is favored (10).
What is the aim of nitration of acetanilide?
In this electrophilic aromatic substitution reaction, the acetamido group (−NHCOCH3) directs the nitronium ion (+NO2) to the ortho and para positions of the aromatic ring. 6 Thus, nitration of acetanilide principally produces ortho- and para-nitroacetanilides, with the para compound being the major product.
How can you separate the products ortho and para in the nitration of acetanilide?
Ortho nitro acetanilide is separated from para nitro acetanilide by dissolving the solution in ethanol. o-nitroacetanilide is more soluble in ethanol as compared to water.
What makes acetanilide soluble in water?
The reason for why water is able to dissolve a number is substances, is due to this polarity that enables water to separate polar solute molecules. Waters polarity makes gives it the property of being a good solvent. … When the water was boiled at a temperature below the acetanilide melting point.
Is acetanilide soluble in hot water?
Acetanilide has a much higher solubility in hot water than in cold water. … A successful recrystallization requires that the compound be soluble at the solvent’s boiling point and insoluble at low temperature.
How is the solubility of acetanilide?
NamesBoiling point304 °C (579 °F; 577 K)Solubility in water<0.56 g/100 mL (25 °C)SolubilitySoluble in ethanol, diethyl ether, acetone, benzenelog P1.16 (23 °C)
What is the biotransformation of Acetanilide?
Acetanilide like phenylbutazone is paraoxidized by the liver endoplasmic reticulum as a primary biotransformation step. … Plasma clearance values and half lives of acetanilide were measured in this group of patients and compared with those of a group of 30 healthy volunteer controls.
How is Acetanilide poisoning treated?
Solvent wash all contaminated surfaces with 60-70% ethanol followed by washing with a soap and water solution. Do not reenter the contaminated area until the Safety Officer (or other responsible person) has verified that the area has been properly cleaned.
What type of organic functional group does Acetanilide contains?
Structure & Chemical Formula Acetanilide is an organic chemical compound (meaning it’s composed of carbon and hydrogen mostly) that is classified as an amide in terms of its functional group. This means that it has the carbonyl group (carbon-oxygen double bond) bonded directly to a nitrogen atom.


