How does a Buchner flask work
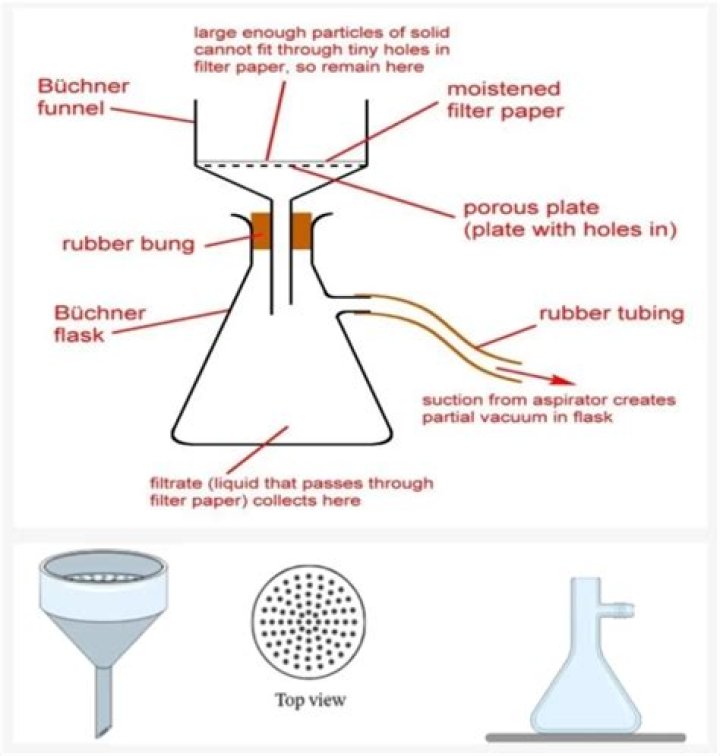
The Büchner funnel holds the sample isolated from the suction by a layer of filter paper. During filtration, the filtrate enters and is held by the flask while the residue remains on the filter paper in the funnel.
How does a Buchner filter work?
A Buchner Funnel is a fritted/perforated funnel used in commercial laboratories for the vacuum-assisted filtration and separation of liquid substances. Buchner Funnels allow you to filter liquids by pulling them through qualitative filter paper and perforations in the top of the funnel using a vacuum pump.
When should you clamp a Buchner flask?
Any solvent which boils at about 125 degrees or lower will boil off under the reduced pressure in the vacuum flask. To perform a vacuum filtration, clamp the flask securely to a ring stand.
What is the function of Buchner funnel?
What Is a Buchner Funnel Used For? Buchner funnels are used in the laboratory for vacuum-assisted liquid filtering. In organic chemistry, these funnels are used to collect recrystallized compounds because they help remove moisture from the final product.How does suction filtration work?
Suction filtration is a chemistry laboratory technique which allows for a greater rate of filtration. Whereas in normal filtration gravity provides the force which draws the liquid through the filter paper, in suction filtration a pressure gradient performs this function.
How do you clean a Buchner filter?
Carefully add 15ml 30% hydrogen peroxide, or 1/3 the volume of sulfuric acid used, to the Büchner funnel and stand back! Let the reaction proceed for about a minute and then apply a quick vacuum (just enough to pull the solution through the sinter into the flask and the sinter is dry.) Monitor the reaction.
What sits between a Büchner funnel and the filter flask in a vacuum filtration?
When a funnel is placed on top of the filter flask, a neoprene adapter (the black or gray “rubbery thing”) or rubber stopper is used to ensure a good seal between the filter flask and the funnel. When the filtration is finished, the tubing from the filter flask should be removed before the vacuum is turned off.
Why is it important to ensure that all the ice has dissolved before using the Buchner funnel?
first thing we will do here is to dissolve this compound in border will miss it, the conductors to see if the solution carries and electrical current. If solution is conducting, then we can determine whether the solution is a strong or weak electrolyte.When and why is Buchner funnel used in crystallization procedure?
Vacuum filtration is used to collect crystals Vacuum filtration can be a fast process because the vacuum helps to rapidly pull the solution through the filter. When collecting crystals, a buchner funnel is used to collect the crystals while the solution is pulled out by the vacuum.
Why should you break the suction in the filter flask?break the suction in the filter flask before turning off the faucet, because the back pressure will cause water to be drawn from the water aspirator into the filter flask and that would be bad.
Article first time published onWhat is the use of distilling flask?
It is primarily used for distillation, the process of separating a mixture of liquids with different boiling points through evaporation and condensation.
How is vacuum created in the sidearm flask Buchner funnel contraption?
When setting vacuum filtration, the first step is fitting the Buchner funnel to a suction flask. After that, we can connect the side arm of the flask to our vacuum pump which is of high quality, advanced design, and high efficiency. … The force of vacuum will draw the liquid through the solid slowly.
Why does fluting a filter paper aid filtration?
“Fluted “ filter paper (LEFT) is used when you wish to separate a liquid and a solid, keeping the liquid and discarding the solid. This arrangement of folds in the filter paper will allow the liquid to pass through it very quickly and give you a lot of surface area on which to collect the solid “impurity”.
What should you do if you see crystals or cloudiness in the bottom of the filter flask during vacuum filtration?
The proper way to wash the crystals is to SHUT OFF the vacuum, add a minimum amount of cold solvent so that the crystals are barely sitting in solvent for about 5 Page 4 seconds (the solvent will not drip through quickly) and then apply the vacuum. The solvent will be sucked into the filtrate.
How do you do filtration?
- Flute filter paper if necessary. …
- Place filter paper in the funnel. …
- Wet the filter paper using a small amount of the liquid that is the solvent of the mixture being filtered.
- After the filtrate has been collected, pass a small amount of the wash liquid through the filter paper to wash the residue.
What are the advantages of Buchner filtration?
The main advantage in using this type of filtration is that it proceeds much more quickly (several orders of magnitude) than simply allowing the liquid to drain through the filter medium via the force of gravity.
How do you secure your filter flask?
It is best to use a ring clamp to secure the filtration funnel, although the funnel could also be simply placed atop the flask. If not using a ring clamp, it is recommended to place a bent paper clip between the flask and funnel to allow for displaced air to escape the bottom flask as liquid drains (Figure 1.81c+d).
Can a side arm flask be used for vacuum filtration?
The larger side-arm flasks (125 mL or 250 mL) work best for vacuum filtering, and the smaller ones (25 mL or 50 mL) work best for removing solvent under reduced pressure.
What is glass funnel?
A separatory funnel, , separation funnel, separating funnel, or colloquially funnel, is a laboratory glassware used in liquid-to -liquid abstraction to dispersed components of liquid mixture into 2 immiscible solvent of different thicknesses such as Oil and Water.
What are the difference between Gooch crucible and porcelain crucible?
Gooch crucible is made of porcelain, whereas sintered glass crucible is made of Pyrex glass. The key difference between Gooch crucible and sintered glass crucible is that Gooch crucibles can withstand very high temperatures while sintered glass crucibles cannot withstand temperatures above 400 Celsius degrees.
What is separating funnel used for?
A separatory funnel (sep funnel) is used to separate immiscible liquids. When two immiscible liquids are placed in a separatory funnel, two layers are seen. The denser solvent will be the bottom layer.
Why should the washing solvent be cold in the suction filtration?
Once the suction filtration process is complete the collected crystals should be washed with a little more ice–cold solvent to remove final soluble impurities which would otherwise be left on the surface of the crystals.
What is the difference between precipitate and filtrate?
In the laboratory these precipitates are generally separated from the solutions by filtering them out on a paper filter. The liquid that passes through the filter paper is the filtrate; the solid precipitate remaining on the filter paper is the residue. Filtering a Product.
How filtration is done by the laboratory?
Filtration is an interesting sterilization method in laboratories. It’s the only method that uses force to separate rather than to kill. When you filter a liquid or gas, it passes through a pore, which stops, or filters out, the passage of larger particles.
How do you separate a precipitate from water?
This is called precipitation. The insoluble solid, called the precipitate, initially forms a suspension, meaning that it is well dispersed in solution. The precipitate typically agglomerates, and then is separated from the liquid by sedimentation, centrifugation, or filtration.
What would happen if the hot saturated solution was filtered by vacuum filtration using a Buchner funnel?
If a hot saturated solution were filtered by vacuum filtration using a Buchner funnel, it would cool as it comes in contact with the Büchner funnel. At lower temperatures, the solubility of the solution decreases. So, solute begins to precipitate.
What is Gooch crucible describe its benefits?
A Gooch crucible, named after Frank Austin Gooch, is a filtration device for laboratory use (and was also called a Gooch filter). It is convenient for collecting a precipitate directly within the vessel in which it is to be dried, possibly ashed, and finally weighed in gravimetric analysis.
Why the resulting crystals can only be rinsed with a little amount of distilled water?
Recrystallization: … When we synthesize a compound in solid form, some impurities are typically present in it, so to remove the impurities, we recrystallize it under specific conditions to minimize the amount of impurities retained in the final product.
Why is it important to recrystallize the solid before taking its melting point?
Compounds, which are less soluble, will crystallize first. … Regardless of crystallization method, the purity of the solid can be verified by taking the melting point. A good (suitable) recrystallization solvent will dissolve a large amount of the impure compound at temperatures near the boiling point of the solvent.
Why must the flask and funnel be heated before the hot solution is filtered?
Place the funnel and filter paper assembly in the beaker so that the rising vapors from the boiling solvent can heat the funnel and filter paper. Having the set up heated before filtration will prevent crystals from forming on the paper and in the funnel (see Figure 1 below).
Why is it important to break the vacuum seal before turning off the vacuum?
Before closing the tap, it is necessary to “break the vacuum” (letting in the air in through any area in the apparatus, by removing the funnel for example), otherwise water goes up the apparatus from the aspirator. The vacuum flask prevents the water from going up the Büchner flask.