How does Liming reduce acidity in soils
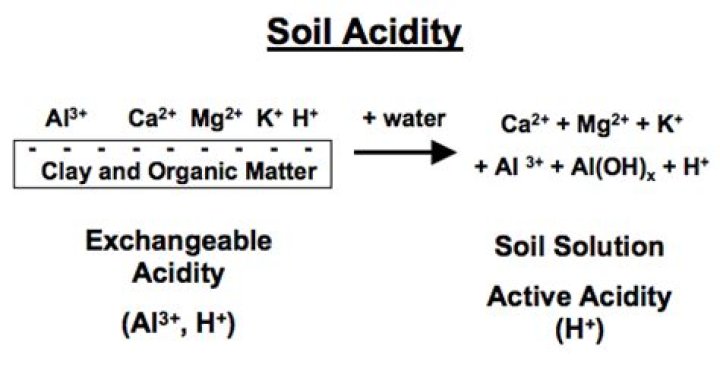
“Soil acidity” is the term used to express the quantity of hydrogen (H) and aluminum (Al) cations (positively charged ions) in soils. … Lime will neutralize this acidity by dissolving, whereupon it releases a base into the soil solution that reacts with the acidic components, hydrogen and aluminum.
How does lime affect soil acidity?
Lime is a soil amendment made from ground limestone rock, which naturally contains calcium carbonate and magnesium carbonate. When lime is added to soil, these compounds work to increase the soil’s pH, making soil less acidic and more alkaline.
Does lime raise or lower acidity?
Adding lime (Figure 1) increases soil pH (reduces acidity), adds calcium (Ca) and/or magnesium (Mg), and reduces the solubility of Al and Mn in the soil.
Why liming is important in acidic soil?
Farmers can improve the soil quality of acid soils by liming to adjust pH to the levels needed by the crop to be grown. Benefits of liming include increased nutrient availability, improved soil structure, and increased rates of infiltration.What is the purpose of liming in soil?
Liming improves soil pH, Ca, and Mg contents and reduces Al concentrations in the soil solution. In addition, liming improves beneficial microbe populations in the soil. Furthermore, liming improves P concentration in the soil solution by reducing P immobilization by Fe and Al in acid soils (Fageria and Baligar, 2008).
Does lime reduce nitrogen in soil?
Lime increases the soil pH and plant nutrient uptake is directly tied to soil pH. (See graph below) Most of the major nutrients including nitrogen, phosphorus, potassium, magnesium and calcium are more available to the plant as the soil pH rises (5.8 to 6.5).
How does limestone affect acidity?
As acid rain falls to the earth’s surface, limestone rocks and limestone components in soil will react with the rain, neutralize the acid and dissolve. As the limestone dissolves, the rocks will wear away, becoming pitted with rounded edges.
How does lime raise soil pH?
You can apply lime with a lime spreader, using a wheelbarrow to transport the liming substance and a trowel to spade it into the spreader as you proceed. Rototill the lime into the top 6 inches of the soil, either over a general area in which you want to raise the pH level or into the hole in which a plant will go.What is liming and its importance?
Liming is the application (to soil) of calcium- and magnesium-rich materials in various forms, including marl, chalk, limestone, burnt lime or hydrated lime. In acid soils, these materials react as a base and neutralize soil acidity. … Structure liming can reduce losses of clay and nutrients from soil aggregates.
Is Limestone good for acidity?The value of limestone is in its ability to neutralize soil acidity. Limestone’s properties differ considerably, and these differences influence the limestone’s ability to neutralize soil acidity.
Article first time published onCan lime neutralize acids?
Lime. Lime is one of the most common alkalis used for neutralizing acids. Lime reacts readily with all types of acids, both inorganic and organic and weak or strong.
Why is lime used to treat soil?
Lime can be used to treat soils in order to improve their workability and load-bearing characteristics in a number of situations. … Lime can substantially increase the stability, impermeability, and load-bearing capacity of the subgrade. Both quicklime and hydrated lime may be used for this purpose.
What is the purpose of using lime?
Lime is the versatile mineral. Various forms of lime are used in environmental, metallurgical, construction, and chemical/industrial applications, and more. The fastest growing use of lime is in environmental applications, where lime is used to comply with air, drinking water, wastewater, and solid waste regulations.
Why does limestone raise pH?
If your rocks are actually limestone, they are the cause for the pH elevation in your aquarium water. Limestone is calcareous (contains calcium) and is known for its ability to both harden the water and increase the pH.
How much lime is needed to change the pH of soil?
You can amend the soil with lime to sweeten the soil for your crops. It will take about 4 tablespoons of lime per 1-square-foot to raise the pH level two points.
What is lime used for in agriculture?
Agricultural lime helps lower the soil’s acidity levels by rendering it more pH neutral. By applying lime to the soil when it becomes too acidic, farmers can ensure they are helping improve their crop output. If crops can’t properly grow, they can’t produce, which impacts the agriculture business and its bottom line.
Is lime for soil acidic or alkaline?
The universal advice is to add lime to the soil ie liming your soil. Lime is alkaline and it will neutralize the acidity of the soil and make it more neutral. Adding lime certainly works – but there is a catch!
What is the economic importance of lime?
The bottom line Limes are high in vitamin C and antioxidants — both of which may offer health benefits. Eating limes or drinking the juice may improve immunity, reduce heart disease risk factors, prevent kidney stones, aid iron absorption, and promote healthy skin. Avoid limes if you’re allergic to citrus fruit.
What is a liming material?
Liming materials are defined as “all materials and all calcium and magnesium products in the oxide, hydrate, carbonate, silicate form or combinations thereof and intended for use in the correction of soil acidity…”.
What happens when lime reacts with acid?
The lime that goes into solution immediately ionizes into Ca++, Mg++ and OH- where these ions unite with the corresponding acid ions of opposite charge, forming calcium or calcium-magnesium salts and water as the reaction products.
Why acidic soil is treated with quick lime?
Acidic soil needs to be neutralise. Quick line when reacts with water/moisture in surrounding produces slaked lime or Ca(OH)2 which is basic and can neutralise the soil for better crops.
What effect does lime have on clay soil?
The addition of lime can raise the soil pH to excessively high levels, reducing the availability of plant nutrients and leading to poor plant growth. Advertisements for gypsum often claim the addition of gypsum will help loosen heavy, clay soils and improve soil drainage.
Is lime good for garden soil?
It has been used safely in agriculture for over a thousand years to change the soil pH, making it easier for plants to take up minerals and nutrients from the soil. Lime also promotes the spread of new, good bacteria in your garden by supplying critical nutrients like phosphorus and zinc in your soil.
Are limes acidic?
Acidity. Both lemons and limes are high in citric acid. This means that they are acidic compared with many other foods.
What is in the lime that neutralizes soil acidity?
According to chemical definition, lime is calcium oxide (CaO). In agriculture, lime is usually defined as calcium or calcium-magnesium containing compounds capable of reducing harmful effects of an acid soil by neutralizing soil acidity and raising the soil pH.
Why does clay soil need more lime?
Soils with a high clay and organic matter content (greater reserve acidity) will require greater amounts of limestone to neutralize acidity than a sandy soil lower in clay content and organic matter (lower reserve acidity) given that each soil has the same pH to start with.
Does agricultural lime increase pH?
Adding lime or other materials can raise soil pH to the ideal range for crop production, create an environment for a healthy function of microbes, and increase the levels of calcium or magnesium ions.