How does particle size affect a molecules transport across a cell membrane
Surface area and thickness of the plasma membrane: Increased surface area increases the rate of diffusion, whereas a thicker membrane reduces it. Distance travelled: The greater the distance that a substance must travel, the slower the rate of diffusion. This places an upper limitation on cell size.
How does molecule size affect cell transport?
If the size of a cell increases it will be more difficult (all other things being equal) for diffusion to transport materials out of the cell. The reason for this is because of the change in the surface to area ratio of the cell.
How do large molecules cross the cell membrane?
It is possible for large molecules to enter a cell by a process called endocytosis, where a small piece of the cell membrane wraps around the particle and is brought into the cell. If the particle is solid, endocytosis is also called phagocytosis. If fluid droplets are taken in, the processes is called pinocytosis.
How does the size of a molecule affect permeability across a cell membrane?
Since smaller molecules generally penetrate more easily through the cell membrane, the diffusivity (D) is higher for smaller molecules. … For molecules of equal size, the one with greater solubility in lipids will pass more quickly into the cell. For molecules of equal solubility, smaller ones penetrate faster.How does particle size affect movement?
Mass of Particle: Heavier particles will move more slowly and so will have a slower rate of diffusion. Smaller particles on the other hand will diffuse faster because they can move faster.
How does increasing cell size affect diffusion?
Explanation: When the cell increases in size, the volume increases faster than the surface area, because volume is cubed where surface area is squared. When there is more volume and less surface area, diffusion takes longer and is less effective.
How does the size of a molecule affect its diffusion?
We know that higher the molecular mass bigger is the size of the molecule. The expression for the Graham’s law of diffusion shows that the rate of diffusion is inversely proportional to the molecular mass i.e. the rate of diffusion is inversely proportional to the size of the molecule.
Are large molecules permeable?
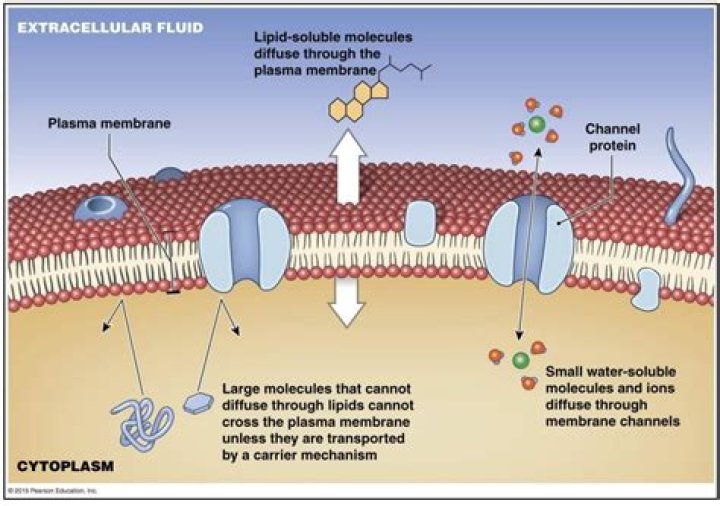
The plasma membrane is selectively permeable; hydrophobic molecules and small polar molecules can diffuse through the lipid layer, but ions and large polar molecules cannot.How does molecular weight affect cell permeability?
The molecular weight affected HPC leaching, pore morphology and film permeability; increasing the molecular weight resulted in decreasing permeability. … Thus, the molecular weight influences the microstructural features of pores, which are crucial for mass transport in EC/HPC films.
Why can only small molecules cross membrane?The cell membrane’s main trait is its selective permeability, which means that it allows some substances to cross it easily, but not others. Small molecules that are nonpolar (have no charge) can cross the membrane easily through diffusion, but ions (charged molecules) and larger molecules typically cannot.
Article first time published onWhat happens if the molecule is too big to fit through the plasma membrane?
Membrane proteins The cell membrane is selectively permeable . … Larger molecules such as glucose require a specific transport protein to facilitate their movement across the cell membrane. Very large molecules such as proteins are too big to move through the cell membrane which is said to be impermeable to them.
How are small molecules transported across membrane?
The simplest mechanism by which molecules can cross the plasma membrane is passive diffusion. During passive diffusion, a molecule simply dissolves in the phospholipid bilayer, diffuses across it, and then dissolves in the aqueous solution at the other side of the membrane.
Why are cells so small and how is this small size beneficial for transport of substances within and between cells?
A large cell has a large volume, or contents. … The large surface area to volume ratio of small cells makes the transport of substances into and out of cells extremely efficient. Another reason for the small size of cells is that control of cellular processes is easier in a small cell than in a large cell.
Why do larger molecules diffuse slower?
Mass of the molecules diffusing: Heavier molecules move more slowly; therefore, they diffuse more slowly. … Lower temperatures decrease the energy of the molecules, thus decreasing the rate of diffusion. Solvent density: As the density of a solvent increases, the rate of diffusion decreases.
How does the size and charge affect the rate of diffusion?
A difference in charge between two regions of fluid can affect the rate and direction of diffusion between them. For example, positively charged substances will tend to diffuse toward a region with an overall negative charge. … Thus, diffusion occurs faster at higher pressures.
What happens when the motion of particles slows?
If the motion of particles slows the particles move closer together. … As the motion of particles continues to increase, they move even farther apart. Eventually the distance between particles is so great that there are little or no attractive forces between the particles.
How does particle radius affect diffusion?
Particles with a smaller hydrodynamic radius have larger diffusion constant but weaker interaction with the sphere while larger particles have smaller diffusion constant but stronger interaction with the sphere.
How does size affect the rate at which molecules can cross the plasma membrane?
Surface area and thickness of the plasma membrane: Increased surface area increases the rate of diffusion, whereas a thicker membrane reduces it. Distance travelled: The greater the distance that a substance must travel, the slower the rate of diffusion. This places an upper limitation on cell size.
Do smaller particles diffuse faster?
Diffusion is faster at higher temperatures and for smaller particles. Particles show diffusional movement not only in cells, but also in water and other fluids. … It also shows that molecules diffuse faster at higher temperature and small molecules diffuse faster than larger ones.
What happens as cell size increases?
As a cell grows bigger, its internal volume enlarges and the cell membrane expands. Unfortunately, the volume increases more rapidly than does the surface area, and so the relative amount of surface area available to pass materials to a unit volume of the cell steadily decreases.
Why does molecular weight affect the movement of molecules through membranes?
Mass of the molecules diffusing: Heavier molecules move more slowly; therefore, they diffuse more slowly. The reverse is true for lighter molecules. Temperature: Higher temperatures increase the energy and therefore the molecules’ movement, increasing the diffusion rate.
Why was the size most effective at maximizing diffusion?
he smallest cube was the most effective at maximizing diffusion because it has the smallest volume, so the diffusion can occur all the way through the cube. … This means that more substances can diffuse through the smaller cube much faster due to having a bigger surface area that has a smaller volume.
What factors affect membrane permeability?
Posted Apr 22, 2021. The permeability of a membrane is affected by temperature, the types of solutes present and the level of cell hydration. Increasing temperature makes the membrane more unstable and very fluid. Decreasing the temperature will slow the membrane.
What are the limits on membrane permeability?
Membrane permeability is typically limited when polar surface area (PSA) exceeds 140 Å2. 13 Compounds that can alternately expose or shield polar functionality depending on the environment (“chame- leonic molecules”) represent an important opportunity to venture beyond the borders of traditional drug space.
What is the relationship between molecular radius and permeability?
As the molecular radius increases, both the diffusion coefficient and permeability decrease.
How do small hydrophobic molecules cross a membrane?
3 – Simple Diffusion Across the Cell (Plasma) Membrane: The structure of the lipid bilayer allows small, uncharged substances such as oxygen and carbon dioxide, and hydrophobic molecules such as lipids, to pass through the cell membrane, down their concentration gradient, by simple diffusion.
What decreases membrane permeability?
Higher concentrations of cholesterol, by filling in gaps between phospholipid tails, decreases permeability even for small molecules that can normally pass through the membrane easily. Cells need far more than small nonpolar molecules for their material and energy requirements.
How can small molecules become larger molecules?
Condensation polymerization refers to the joining together of smaller molecules through the loss of a small molecule, such as water, to form a larger molecule.
Why can't small molecular weight material such as ions pass through the cell membrane?
While some polar molecules connect easily with the outside of a cell, they cannot readily pass through the lipid core of the plasma membrane. Additionally, while small ions could easily slip through the spaces in the mosaic of the membrane, their charge prevents them from doing so.
How do small molecules or ions pass across the cell membrane in active transport?
Active transport is the energy-requiring process of pumping molecules and ions across membranes “uphill” – against a concentration gradient. The active transport of small molecules or ions across a cell membrane is generally carried out by transport proteins that are found in the membrane, also known as a protein pump.
What are two consequences of the extracellular accumulation of sodium ions?
3. Describe two consequences of the extracellular accumulation of sodium ions: Increased amounts of sodium ions outside the cell could create a hypertonic environment, causing intracellular water to diffuse out.