How does salt melt ice chemically
Salt melts ice essentially because adding salt lowers the freezing point of the water. … Ice typically is coated with a thin film of liquid water, which is all it takes. Pure water freezes at 32°F (0°C). Water with salt (or any other substance in it) will freeze at some lower temperature.
Is using salt to melt ice a chemical change?
The melting of ice is a physical change when it occurs naturally. But when you speed up the process by using a reactant, such as salt, it becomes a chemical reaction.
How does salt make ice melt faster?
Because salt particles make it harder for water particles to freeze back onto the ice, the ice that is in contact with dissolved salt melts faster. When the saltwater flows over the surface it melts the ice on its way, creating channels, like rivers, over the surface of the ice ball.
How does ice melt work chemistry?
An ice melter must dissolve into a solution quickly to be effective. Since calcium chloride and magnesium chloride are liquids in their natural states they return to a liquid quickly. Then when these chemicals come in contact with water a reaction is caused that gives off heat to melt ice and snow.What is salt and why does it melt ice?
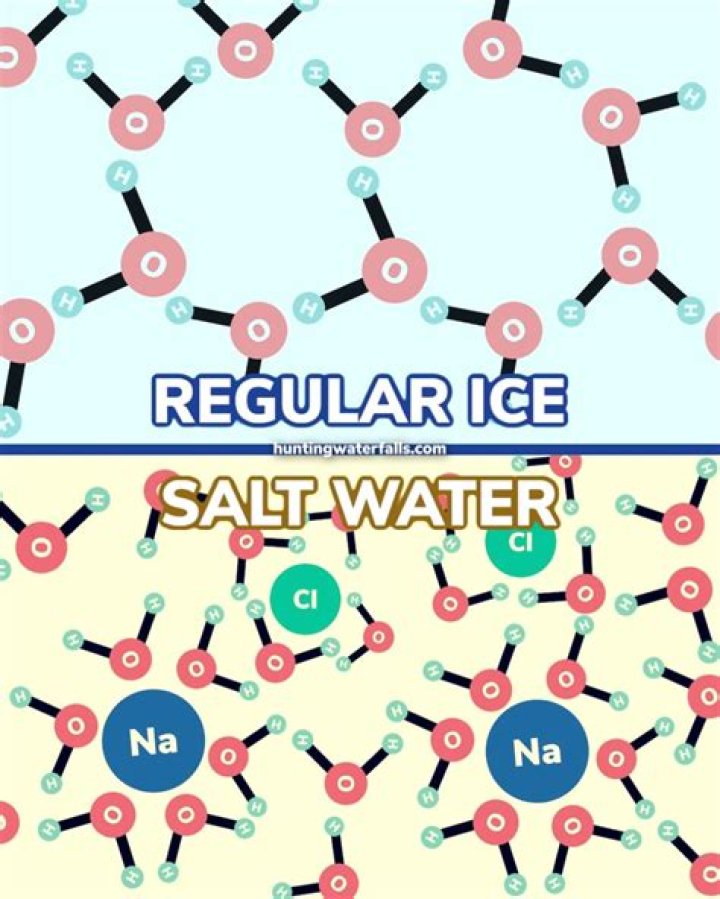
When added to ice, salt first dissolves in the film of liquid water that is always present on the surface, thereby lowering its freezing point below the ices temperature. Ice in contact with salty water therefore melts, creating more liquid water, which dissolves more salt, thereby causing more ice to melt, and so on.
Why do they sprinkle salt on ice?
Road salt works by lowering the freezing point of water via a process called freezing point depression. The freezing point of the water is lowered once the salt is added, so it the salt makes it more difficult for water to freeze.
What is the purpose of using salt on ice during winter storms chemistry?
So if there’s snow, sleet or freezing rain and the ground is 32 F or colder, solid ice will form on streets and sidewalks. If the water is mixed with salt, though, the freezing temperature of the solution is lower than 32 F. The salt impedes the ability of the water molecules to form solid ice crystals.
Why does ice melt slower in salt water?
Assuming equal temperature, ice melts faster in salt water because salt water has a lower freezing point than fresh water, so the ice cube will have to absorb less heat in order to melt in salt water than in fresh water.How salt melts ice and prevents freezing?
Salt molecules block water molecules from packing together when temperature is lowered. It then prevents them from becoming ice. More water molecules leave the solid phase than the ones entering the solid phase. Freezing point depression occurs when the freezing point of the liquid is lowered by addition of solute.
Why does ice melt turn to liquid?Ice melts when heat energy causes the molecules to move faster, breaking the hydrogen bonds between molecules to form liquid water.
Article first time published onWhat type of mixture is created by putting salt on ice?
1Sensor-CASSY524 010 or 524 0131CASSY Lab 2524 2201Chemistry box or NiCr-Ni adapter S524 067(3)1Temperature sensors NiCr-Ni, type K529 676
How does sand melt ice?
How sand works to melt the ice. Sand works by improving traction over ice and reduces slippery conditions on roads. Sand applied over ice may sometimes cause friction which does help in melting the ice. It operates by simply creating a firmer grip over the icy surfaces.
How does salt and ice make ice cream?
Salt provides the solution. Similar to sugar, salt affects how water freezes and effectively lowers the freezing/melting point of water. Creating a saltwater slush and packing this around our ice cream base allows us to cool the base enough so that it starts to thicken and freeze before the ice melts completely.
Why does rock salt make ice colder?
On the surface of every ice cube, an exchange process goes on between water in the liquid and solid states. The presence of salt in the water lowers the equilibrium point of this exchange by lowering the freezing point of the liquid water.
Why is salt put on icy roads and sidewalks in the winter?
Why does salt melt ice and snow? Salt causes a phenomenon called freezing point depression, which means it lowers the freezing point of water. Water normally freezes at 32 degrees Fahrenheit, but when you add salt, that threshold can drop as low as 20 degrees Fahrenheit.
Why does salt not melt when heated?
Why It Matters Only a little bit of energy is required to loosen these intermolecular interactions enough to allow the formation of a free-flowing liquid, so its melting point is relatively low. The ions in a salt crystal are held together by strong ionic bonds.
Why do the majority of our oceans never freeze?
Salt is the key to understanding our experiment’s results! Here’s why: The more salt in the water, the lower the temperature has to be for the water to freeze. This is why the ocean doesn’t freeze: There’s too much salt in it.
Is salting highways chemical or physical?
Today an estimated 20 million tons of salt is scattered on U.S. roads annually—about 123 pounds for every American. The rock salt used on roadways is chemically much like regular table salt, and is mined from large underground deposits that formed after prehistoric oceans evaporated.
At what temperature does salt stop melting ice?
Rock salt is a staple for most winter maintenance deicing programs, but at what temperature does it become ineffective? Salt will “work,” i.e. it will melt ice, all the way down to its eutectic temperature of -6 0F.
Does freezing salt water remove the salt?
Q: can you separate salt from water by freezing? A: Yes, because when ice and liquid water are in equilibrium, the concentration of salt in the liquid is much higher than in the ice.
What type of additive slows the melting of ice?
Adding salt to the ice/water mix causes a temperature drop that slows the melting rate and increases the freezing rate [3]. The net result is that the ice melts more and more slowly after the initial addition of salt.
What substance makes ice melt slower?
Insulation. General insulation of the ice causes it to melt slower. Wrapping it in wool, Styrofoam or wood contains the cold air emitting from the ice, keeping the temperature of the ice low. Putting the ice in a vacuum, such as a vacuum-insulated Thermos bottle, also prevents the ice from melting quickly.
Why does ice melt faster on metal?
Energy can be transferred (move) from the surroundings to the ice by conduction through the metal or plastic. Metal is a better conductor than plastic, so energy is transferred more quickly through the metal. This is why we saw the ice on the metal block melt more quickly.
What chemical is liquid ice?
The most common liquid deicers are simple sodium chloride brines. That means they are composed of a proportion of water and salt—the very same salt that goes into rock salt. Liquid deicers work differently than rock salt, not due to their chemical composition, but due to the way they’re applied.
Does ice melt harm concrete?
It is not recommend that ice melter be used on damaged concrete. Damaged concrete will absorb the water [melted ice] more readily since its ‘seal’ has been broken and therefore can be more susceptible to damage. It is best to reseal or treat your damaged concrete as mentioned earlier, before using any ice melter on it.
Does ice melt expire?
Most ice melt products do not expire! This is because salt products contain chloride which is a mineral that never goes bad. … Opened bags of chloride ice melt will still work but may require a greater amount of product – making them less cost-effective. There are also other types of ice melt such as rock salt.
What melts ice the fastest sand or salt?
Out of the three substances tested, salt melted ice the fastest, followed by sugar, then sand, and lastly control.
How does sugar melt ice?
Sugar lowers the freezing point of water by binding with the water molecules and creating more space between them. This helps them overcome the electrostatic forces that bind them into a solid structure. The same is true for any substance that dissolves in water.
Is sand safe to melt ice on concrete?
When to Use Sand on Icy Driveways Whereas rock salt melts ice, sand does not. … It can capably create traction on ice at any temperature, whereas rock salt is not effective in extreme cold. But sand is only effective if it is on the surface of the ice. If it gets buried under snow, it needs to be reapplied.
What is the chemical formula of salt?
To most people, salt refers to table salt, which is sodium chloride. Sodium chloride forms from the ionic bonding of sodium ions and chloride ions. There is one sodium cation (Na+) for every chloride anion (Cl–), so the chemical formula is NaCl (Fig.
Does salt water boil faster?
When salt is added, it makes it harder for the water molecules to escape from the pot and enter the gas phase, which happens when water boils, Giddings said. This gives salt water a higher boiling point, she said.



