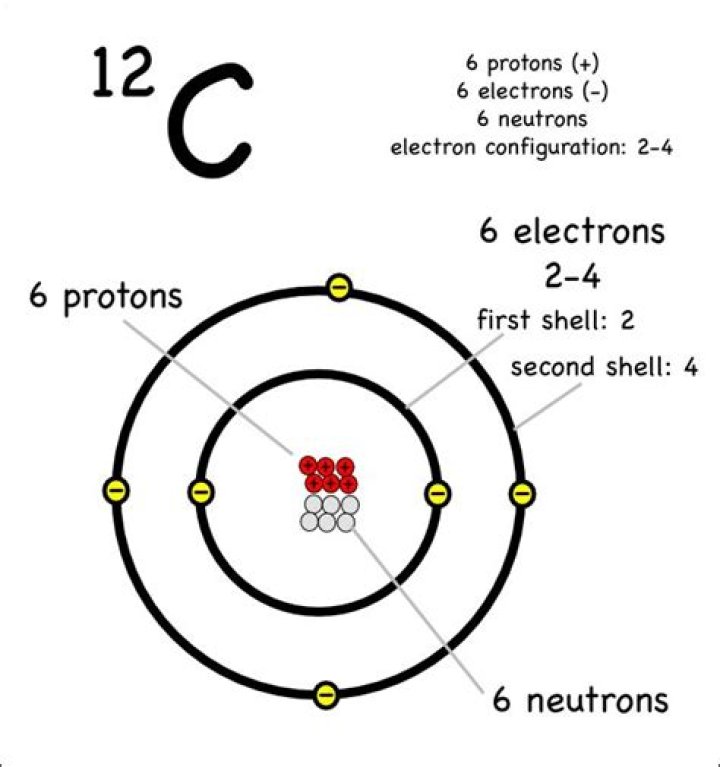
How heavy is a carbon 12 atom
Average Atomic Mass On the periodic table the mass of carbon is reported as 12.011 amu.
How many grams does 1 carbon atom weigh?
We know that a mole is defined as a collection of 6.022×1023 atoms. So, a mole carbon contains 6.022×1023 atoms of carbon. Therefore, we can say that the mass of 6.022×1023 atoms of carbon is 12 grams. Hence the mass of one carbon atom is 1.992×10−23grams and correct answer is option A.
How big is an atom of carbon?
Carbon is in group IV A of the periodic table with atomic number 6, an atomic weight of 12.011, and a density of 2.26. It melts at 3727 C. The electronic configuration of C is (1s)2(2s)2(2p)2, and the atomic radius is 0.0914 nm.
How heavy is an atom?
No atom has a mass of 1 gram. 1 mole of hydrogen atoms (about 6.02*10^23 atoms) has a mass of 1.008 grams. Hydrogen has an average atomic mass of 1.008 unified atomic mass units (amu). The commonly stated reason that atomic weights are not whole numbers is that they are weighted averages of several isotopes.Which atom has the smallest atomic weight?
The lightest chemical element is Hydrogen and the heaviest is Hassium.
Why is the atomic mass of carbon not exactly 12?
Simply because the atomic mass is defined as 1/12 of the mass of 12C. Others isotopes of carbon (13C mostly, with an abundance of 1.1% approximately) account for an average atomic mass slightly above 12.
How many atoms are in 1g of carbon?
Therefore 1g Carbon will contain (6.0225X10^23)/12 atoms of Carbon.
What is difference between 12g of carbon and 12u of carbon?
Difference between 12 gram of carbon and 12 unified mass of carbon: 12 gram of carbon means a mole of carbon while 12 unified mass of carbon means that the molar mass of carbon is 12g and that 12g of carbon make up one mole. It means that 1 mole or 12 u of carbon contain 6.023×1023 carbon atoms.What is the mass of 4 atoms of carbon?
1 mole of C atoms has a mass of 12.011 g. So the molar mass of C = 12.011 g/mol. So multiply that by 4. 4 moles of carbon atoms has a mass of 48.044 g.
Do all atoms weigh the same?REMEMBER: Atoms of the same chemical element do not always have the same mass because, although the number of protons in the nucleus is the same for all atoms of the same element, the number of neutrons is not. Most elements as they occur naturally on earth are mixtures of several isotopes.
Article first time published onWhat is the heaviest element found in nature?
The heaviest naturally stable element is uranium, but over the years physicists have used accelerators to synthesize larger, heavier elements.
How many atoms are in a fingernail?
This means a row of 108 (or 100,000,000) atoms would stretch a centimeter, about the size of your fingernail. Atoms of different elements are different sizes, but 10-10 m can be thought of as a rough value for any atom.
What is the largest atom possible?
Thus, helium is the smallest element, and francium is the largest.
What is the RAM of oxygen?
ElementRelative atomic massCarbon (C)12Oxygen (O)16Magnesium (Mg)24Chlorine (Cl)35.5
How long do man made elements last?
The mechanism for the creation of a synthetic element is to force additional protons onto the nucleus of an element with an atomic number lower than 95. All synthetic elements are unstable, but they decay at widely varying rates: their half-lives range from a few hundred microseconds to millions of years.
Why is helium the smallest atom?
Explanation: Helium has the smallest atomic radius. This is due to trends in the periodic table, and the effective nuclear charge that holds the valence electrons close to the nucleus. Atomic radius decreases as you move across a period from left to right and decreases as you move up a group from bottom to top.
How many atoms are there in 6g of carbon?
Answer: Answer: Since 12 grams of carbon contains 1 mole of its atoms, 6 grams of it contains half a mole, that is, 3.01 x 10^23 atoms of it.
How much atoms are in a mole?
The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12. 12.00 g C-12 = 1 mol C-12 atoms = 6.022 × 1023 atoms • The number of particles in 1 mole is called Avogadro’s Number (6.0221421 x 1023).
What is the mass of 1 carbon?
Elementmass of 1 atom (amu)mass of 100 atoms (amu)C12.011,201W183.918,390
Why was carbon-12 chosen as the standard?
Carbon-12 is the standard while measuring the atomic masses. Because no other nuclides other than carbon-12 have exactly whole-number masses in this scale.
What has the atomic number 13?
Atomic NumberSymbolName10NeNeon11NaSodium12MgMagnesium13AlAluminum
What is the mass of 1 mole of carbon atoms?
Exactly 12 grams of pure carbon-12 powder is known as one mole. The number of atoms of carbon-12 present in this one mole sample is 6.022 136 7 x 1023. This number is known as Avogadro’s number.
What is the mass of 25 moles of carbon atoms?
Answer and Explanation: Therefore, a 0.25 mole of carbon has a mass of 3.00 g.
What is the mass of 6.02 x10 23 atoms of carbon?
Example: Carbon (atomic mass = 12.01) – 1 mole of Carbon weighs 12.01 grams. This means that 6.022×1023 carbon atoms (or molecules) weights 12.01 grams.
How many grams does an atom of hydrogen weigh class 11?
We know that the weight of a hydrogen atom is 1 gram which is also its atomic weight and also its atomic number. Moreover, we know that 1 molecule is made up of 6.023×1023 atoms. Hence, 1 atom will have 1 gram of hydrogen. Then, 6.023×1023 atoms will have 16.023×1023 grams of hydrogen.
How many grams does an atom hydrogen weigh?
The weight of 1 atom of hydrogen is calculated as 1.66058 × 10-24 grams.
How many grams does an atom of hydrogen?
The mass of a hydrogen atom is 1.6736 × 10–24 g.
What is the g mol of helium?
The molar mass of of helium is 4.002602 g/mol . This means that one mole of helium has a mass of 4.002602 g .
What weighs most in an atom?
Over 99.9 percent of an atom’s mass resides in the nucleus. The protons and neutrons in the center of the atom are about 2,000 times heavier than the electrons orbiting around it. Because the electrons are so light by comparison, they represent only a tiny fraction of a percent of the atom’s total weight.
How many atoms are in the human body?
It is hard to grasp just how small the atoms that make up your body are until you take a look at the sheer number of them. An adult is made up of around 7,000,000,000,000,000,000,000,000,000 (7 octillion) atoms.
What does Amu stand for?
abbreviation. atomic mass unit (def.