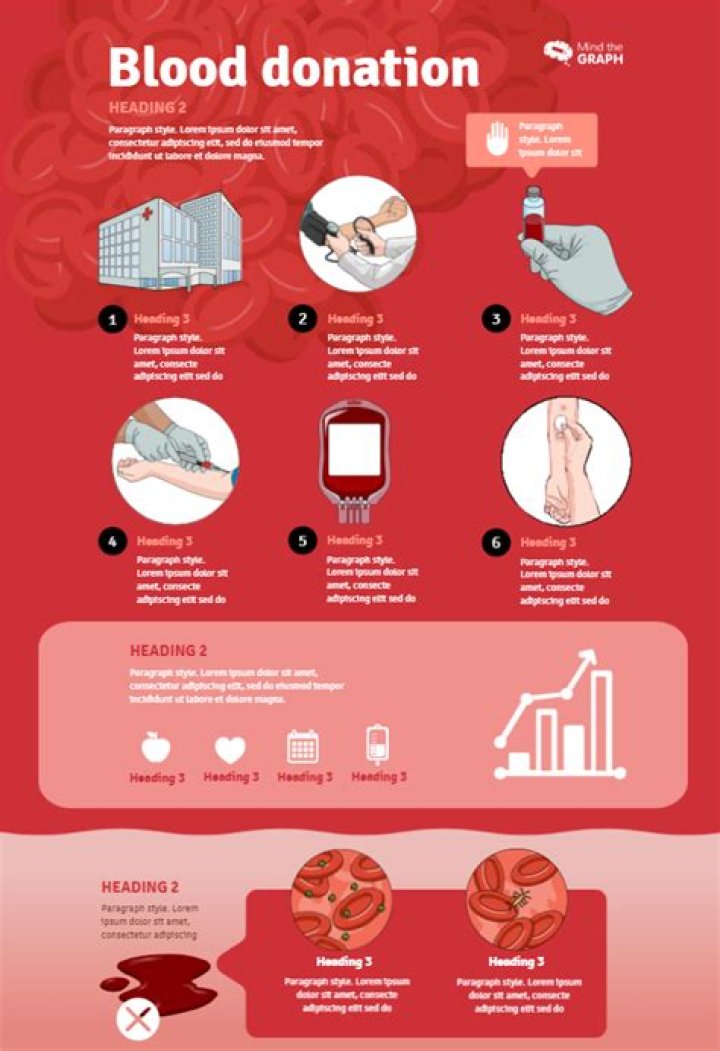
How is an atomic spectra produced
Atomic emission spectra are produced when excited electrons return to the ground state. When electrons return to a lower energy level, they emit energy in the form of light.
How is the atomic emission spectrum of an element produced quizlet?
The energy released when an electron jumps from the excited state to the ground state is equal to the difference between the two levels. An emission line spectrum is produced by low-density hot gases. Energy is absorbed when an electron jumps from a lower energy level to high energy level.
What is the atomic spectra of an element?
Atomic spectra are defined as. The spectrum of the electromagnetic radiation emitted or absorbed by an electron during transitions between different energy levels within an atom. When an electron gets excited from one energy level to another, it either emits or absorbs light of a specific wavelength.
How are spectral lines produced quizlet?
When an electron falls from higher to lower energy level, it emits a photon of energy that is equal to the difference in energy between two levels. Hence, only photons of specific energies which correspond to specific wavelengths are emitted, giving rise to an emission line spectrum.Who explained atomic spectra?
1913: Danish physicist Niels Bohr (1885–1962) (Figure 12) presents his theory of the atom, which explains the Rydberg formula of simple spectra.
What is an atomic emission spectrum quizlet?
definition of atomic emission spectrum. the set of frequencies of electromagnetic waves given off by atoms of an element, consists of a series of fine lines of individual colors.
What causes the unique atomic spectra produced for each element?
When atoms are excited they emit light of certain wavelengths which correspond to different colors. Each element produces a unique set of spectral lines. … Since no two elements emit the same spectral lines, elements can be identified by their line spectrum.
How does a spectroscope produce a continuous spectrum quizlet?
Continuous spectra is where the sun and heated solids emit radiation that contain all frequencies, and line spectra are produced by excited atoms in the gas phase, and contain only certain frequencies.How the different types of spectra are generated?
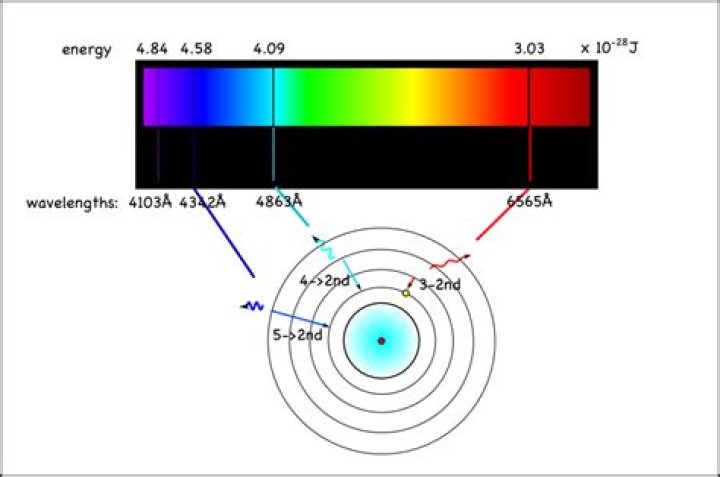
Balmer lineWavelength (nm)Hγ434.1Hδ410.2Hε397.0
What are continuous emission and absorption spectra how are they produced quizlet?A continuous spectrum is produced by a hot, high-density light source. An emission spectrum is produced by a hot, low-density light source. An absorption spectrum is produced by a hot, high-density light source shining through a cool, low-density medium. You just studied 16 terms!
Article first time published onWhat are some examples of atomic spectra?
White light viewed through a prism and a rainbow are examples of continuous spectra. Atomic emission spectra were more proof of the quantized nature of light and led to a new model of the atom based on quantum theory.
How can spectra be used to find the composition of a gas?
When astronomers look at an object’s spectrum, they can determine its composition based on these wavelengths. … This is absorption, and each element’s electrons absorb light at specific wavelengths (i.e., energies) related to the difference between energy levels in that atom.
Why are unique atomic spectra produced by different elements quizlet?
The emission spectra are unique to each element because all atoms of one element are identical, and each atom has a specific number of valence electrons, with a specific level of energy present. That energy holds the valence electrons in orbit within a specific energy level.
How can atomic spectra be used to identify elements?
In emission spectra, bright lines will show up corresponding to the difference between energy levels of the elements where in an absorption spectrum, the lines will be dark since every element has unique energy levels, the spectra can help identify elements in a sample.
How did Bohr explain the hydrogen spectrum using his new theory?
Niels Bohr explained the line spectrum of the hydrogen atom by assuming that the electron moved in circular orbits and that orbits with only certain radii were allowed.
What would an atomic spectrum look like if the electrons energy levels in an atom were not quantized?
If atomic energy levels were not quantized, the light would be white (all energies).
What is the name of the visible emission spectrum produced by hydrogen?
The electron energy level diagram for the hydrogen atom. He found that the four visible spectral lines corresponded to transitions from higher energy levels down to the second energy level (n = 2). This is called the Balmer series.
What is happening inside the atom that causes the line emission spectra to be produced?
An emission line is produced by an atom in a “excited” energy state—the electron is not in as low an energy orbit as possible. Remember rule #3! In order to go to a lower energy orbit, the electron must lose energy of a certain specific amount.
How does a spectroscope produce a continuous spectrum?
These lines are formed when the cool gas absorbs certain wavelengths (colors) from the original beam of light. … A luminous solid or liquid, or a sufficiently dense gas, emits light of all wavelengths and so produces a continuous spectrum of radiation (Figure 2.11).
What is a continuous spectrum quizlet?
Continuous spectrum. A spectrum that exhibits all the wavelengths of visible light.
How does line spectra differ from continuous spectra?
The key difference between continuous spectrum and line spectrum is that the continuous spectrum contains all the wavelengths in a given range whereas the line spectrum contains only a few wavelengths.
How can a continuous spectrum be normally produced quizlet?
continuous spectrum is emitted by a “blackbody.” It consists of light of all wavelengths, but the amount of light emitted at each wavelength varies and depends on the temperature of the blackbody. An absorption spectrum is created when the light from a blackbody shines through a cooler gas.
How absorption spectrum is formed?
An absorption spectrum occurs when light passes through a cold, dilute gas and atoms in the gas absorb at characteristic frequencies; since the re-emitted light is unlikely to be emitted in the same direction as the absorbed photon, this gives rise to dark lines (absence of light) in the spectrum.
What conditions produce an emission line spectrum quizlet?
The emission line spectrum is produced by electrons jumping up in energy level, while the absorption line spectrum is produced by electrons jumping down in energy level. The only way to decide is to make a graph of the intensity of the light at every wavelength, and then analyze the graph carefully.
What is the tool used to separate the components of light and atomic spectra?
A spectrometer (/spɛkˈtrɒmɪtər/) is a scientific instrument used to separate and measure spectral components of a physical phenomenon. … A mass spectrometer measures the spectrum of the masses of the atoms or molecules present in a gas. The first spectrometers were used to split light into an array of separate colors.
How can we explain the distinct lines that appear in an atomic spectrum quizlet?
Every element emits a unique line spectrum. When the light emitted is separated into wavelengths, a spectrum is produced. Both emission and absorption spectrum produce lines in the same place. Certain gases can only absorb and emit specific wavelengths of electromagnetic radiation.
What is a continuous spectrum and how is it produced?
Continuous spectra (also called thermal or blackbody spectra) arise from dense gases or solid objects which radiate heat. They emit radiation over a broad range of wavelengths, thus the spectra appear smooth and continuous.