How is methyl benzoate formed
Methyl benzoate is an ester with the chemical formula C6H5COOCH3. It is formed by the condensation of methanol and benzoic acid. It is a colorless to slightly yellow liquid that is insoluble with water, but miscible with most organic solvents.
How is methyl benzoate made?
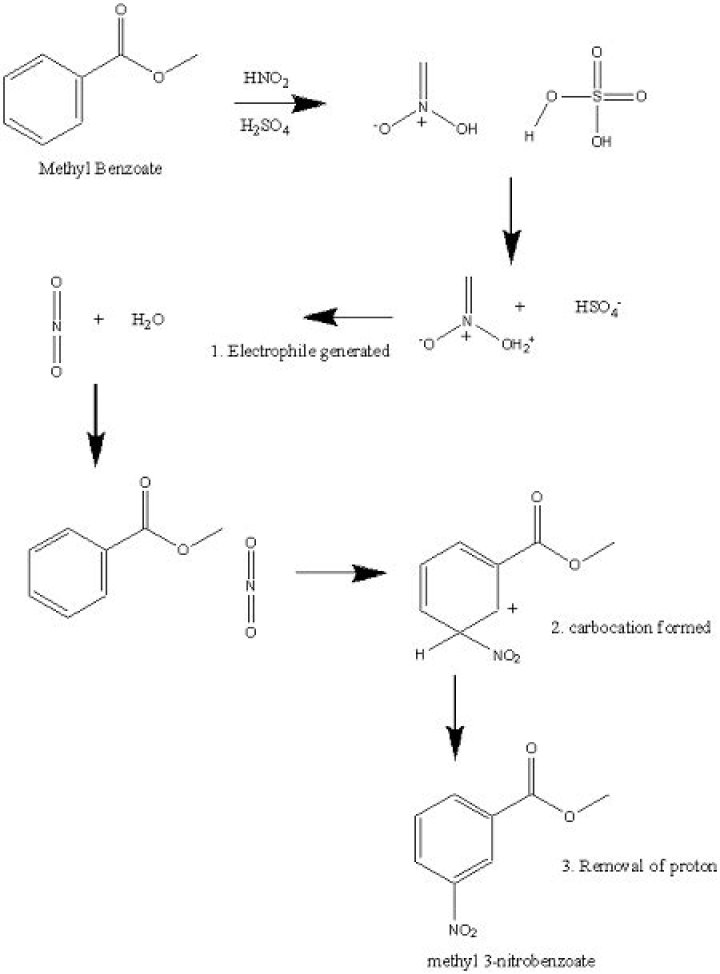
Methyl benzoate is formed by the condensation of methanol and benzoic acid, in presence of a strong acid. Methyl benzoate reacts at both the ring and the ester, depending on the substrate. Electrophiles attack the ring, illustrated by acid-catalysed nitration with nitric acid to give methyl 3-nitrobenzoate.
How do you make methyl benzoate from benzene?
(i) Methyl benzoate is the ester of benzene. It can easily be prepared after preparing benzoic acid. So, we can form benzoic acid by Friedel Craft alkylation and then form an ester, we can just oxidize the formed acid to get methyl benzoate. The process is called esterification.
How do you get methyl benzoate?
Methyl benzoate is a benzoate ester obtained by condensation of benzoic acid and methanol.What happens when methyl benzoate?
ChEBI Namemethyl benzoateDefinitionA benzoate ester obtained by condensation of benzoic acid and methanol.
What is the name of the ester formed from methanol and salicylic acid?
Methyl salicylate can be produced by esterifying salicylic acid with methanol.
What happens when methyl benzoate is hydrolyzed?
Write an equation for the hydrolysis of methyl benzoate in a potassium hydroxide solution. In basic hydrolysis, the molecule of the base splits the ester linkage. The acid portion of the ester ends up as the salt of the acid (in this case, the potassium salt).
How is benzaldehyde converted to benzophenone?
Hint: When we do the oxidation of aldehyde, it is converted into carboxylic acid. And then by decarboxylation it gets converted to aromatic hydrocarbon. Then if we treat it with benzoyl chloride, we get the desired result.How do you dispose of methyl benzoate?
Contain spillage, and then collect with an electrically protected vacuum cleaner or by wet-brushing and place in container for disposal according to local regulations (see section 13). Keep in suitable, closed containers for disposal.
How is Bromobenzene converted to benzoic acid?Answer: Bromobenzene to benzoic acid we known method is prepared Grignard reagent of bromobenzene followed by quenching in carbon dioxide and then acidification gives benzoic acid. cyanation using KCN or Zn(CN)2 followed by hydrolysis also one of the good methods.
Article first time published onHow do you convert benzene to benzaldehyde?
1. Etard reaction. 2.By methylation of benzene followed by chlorination and then reaction with aqueous KOH and then mild oxidation yields Benzaldehyde.
What is the name of the compound C8H8O2?
Phenylacetic acid | C8H8O2 – PubChem.
Is methyl benzoate activating or deactivating?
The nitration of methyl benzoate is an example of electrophilic substitution. The carbonyl group withdraws electron density from the ring deactivating it towards electrophilic substitution.
Is methyl benzoate soluble in methanol?
It is colorless oily liquid with strong floral and cherry aromas. It is miscible with ethyl ether, soluble in methanol, ethyl ether but insoluble in water and glycerol.
Why is methyl benzoate used instead of benzene?
Methylbenzene reacts rather faster than benzene – in nitration, the reaction is about 25 times faster. That means that you would use a lower temperature to prevent more than one nitro group being substituted – in this case, 30°C rather than 50°C.
What is the function of the acid in the preparation of methyl benzoate?
Contact your TA! Allow the solution to cool. Add 50 mL of water to a separatory funnel, then add the contents of the flask. The flask should then be washed with 40 mL of dichloromethane (CH2Cl2) and the washings transferred to the separatory funnel.
Is methyl benzoate a carboxylic acid?
Methyl benzoate belongs to the class of organic compounds known as benzoic acid esters. Methyl benzoate is an ester with the chemical formula C6H5COOCH3. It is formed by the esterification of the carboxyl group in benzoic acid with methanol.
What type of functional group is present in methyl benzoate?
What type of functional group is present in methyl benzoate? Methyl Benzoate has an aromatic double carbon- carbon bond which makes the functional group an Ester.
Why does methyl benzoate dissolve in sulfuric acid?
Concentrated sulfuric acid converts methyl benzoate into benzoic acid. Therefore, the polar benzoic acid dissolves in polar sulfuric acid.
Can methanol be produced by fermentation?
Methanol is produced during fermentation by the hydrolysis of naturally occurring pectin in the wort (Nakagawa et al. 2000; Mendonca et al. 2011). PME de-esterify pectin to low—methoxyl pectins resulting in the production of methanol (Chaiyasut et al.
How methyl alcohol is formed?
Methanol is predominantly produced by steam reforming of natural gas to create a synthesis gas, which is then fed into a reactor vessel in the presence of a catalyst to produce methanol and water vapor. Although a variety of feedstocks other than natural gas can and have been used, today’s economics favor natural gas.
Why is methanol called wood alcohol?
In industrial settings, methanol is produced synthetically by a multi-step process involving natural gas and a process called “steam reforming.” In the past, methanol was once made by the distillation of wood, which is why it is also called wood alcohol.
What happens to the methanol produced when making aspirin?
Methanol is the alcohol which is released by hydrolysis. Second, the hydroxyl group on salicylic acid will be used in ester formation to produce aspirin. … As a result, the thermodynamic equilibrium will now be for the formation of an ester instead of its hydrolysis.
How are esters formed?
Esters are formed by the condensation reaction between an alcohol and a carboxylic acid. This is known as esterification. In a condensation reaction, two molecules join and produce a larger molecule whilst eliminating a small molecule. During esterification this small molecule is water.
How does vinegar react with aspirin?
If you have an old bottle of aspirin tablets you may find they have a slight smell of vinegar, especially if they have become damp. As you have seen, when aspirin reacts with water the carboxylic acid that it forms, by hydrolysis of the ester group, is acetic acid.
What is the signal word for acetone?
H225: Highly flammable liquid and vapour. H319: Causes serious eye irritation H336: May cause drowsiness or dizziness P210: Keep away from heat/sparks/open flames/hot surfaces – No smoking. P305+351+338: IF IN EYES: Rinse cautiously with water for several minutes.
Is benzyl alcohol hazardous?
Ingestion: May cause severe gastrointestinal tract irritation with nausea, vomiting and possible burns. May be harmful if swallowed. Inhalation: Vapors cause irritation of the respiratory system. Chronic: Prolonged or repeated skin contact may cause dermatitis.
How is acetone converted to propene?
Acetone to can be converted to propene by reducing acetone with NaBH4 to form 2-propanol followed by dehydrohalogenation using alc.
How do you convert benzaldehyde to benzyl alcohol?
In the process of converting benzaldehyde to benzly alcohol, one molecule is oxidesed to sodium benzoate at the expense of other which is reduced to benzyl alcohol. This reactio is know as Cannizzaro’s reaction.
How will you convert benzophenone?
Complete step by step answer: –Benzonitrile reacts with phenyl magnesium bromide in presence of dry ether to give an imine complex which on acid hydrolysis gives benzophenone. -During the reaction benzonitrile and phenyl magnesium bromide should be taken in equimolecular proportion.
How do you convert Bromobenzene to benzyl alcohol?
Bromobenzene is first converted to a Grignard reagent by reaction with Mg/ether, and then is converted to the alcohol by reaction with formaldehyde. In a second step,benzyl alcohol is reacted with thionyl chloride to give the final product.



