How is percent by mass calculated
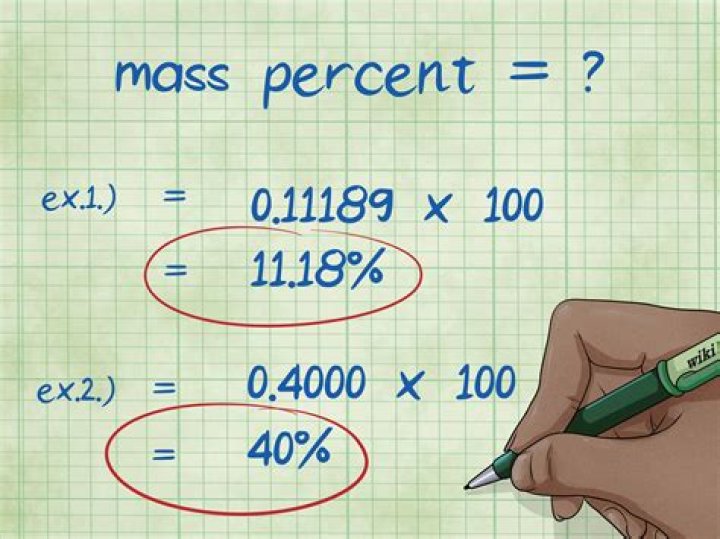
To calculate the mass percent of an element in a compound, we divide the mass of the element in 1 mole of the compound by the compound’s molar mass and multiply the result by 100.
What is the formula for percent by mass?
Steps for Finding the Mass Percentage The essential formula for mass percent of a compound is mass percent = (mass of chemical÷total mass of compound) x 100.
How can I calculate percentage?
Percentage can be calculated by dividing the value by the total value, and then multiplying the result by 100. The formula used to calculate percentage is: (value/total value)×100%.
How do you find the percent mass example?
Divide the mass of the element by the total mass of the compound and multiply by 100. This will give you the mass percent of the element. Example 1: mass percent = (2.01588/18.01528) x 100 = 0.11189 x 100 = 11.18%. Thus, the mass percent of Hydrogen atoms in a water molecule is 11.18%.How do you calculate the percent composition by mass of an element?
- Find the molar mass of all the elements in the compound in grams per mole.
- Find the molecular mass of the entire compound.
- Divide the component’s molar mass by the entire molecular mass.
- You will now have a number between 0 and 1. Multiply it by 100% to get percent composition.
How do you calculate percent by volume?
- A percent v/v solution is calculated by the following formula using the milliliter as the base measure of volume (v):
- % v/v = mL of solute/100 mL of solution.
- Example:
- X % = 5.0 mL HCl/100 mL of solution.
- X/100 = 5.0/100.
- 100X = 500.
- X = 5.0% % v/v.
How do u calculate mass?
Mass is always constant for a body. One way to calculate mass: Mass = volume × density. Weight is the measure of the gravitational force acting on a mass.
What percentage is $7 of $35?
Now we can see that our fraction is 20/100, which means that 7/35 as a percentage is 20%.What is mass percent of the solution?
Mass Percent , which is the grams of solute per 100 g of solution. Sometimes you may want to make up a particular mass of solution of a given percent by mass and need to calculate what mass of the solvent to use. For example, you need to make 3000 g of a 5% solution of sodium chloride.
How do you find the percent composition by mass of an isotope?As a percent, the equation would be: (x) + (100-x) = 100, where the 100 designates the total percent in nature. If you set the equation as a decimal, this means the abundance would be equal to 1. The equation would then become: x + (1 – x) = 1. Note that this equation is limited to two isotopes.
Article first time published onHow do you convert GPL to percentage?
The easier thing is to convert to a mass/volume percentage. A 1% m/v solution has 1 gram of solute per 100 mL of solution, or 10 grams per liter. Using our example of 100 mg/L again, There are ten 100-mL units in a liter, so 100 mg/L = 10 mg/100 mL = 0.01 g/100 mL = 0.01% m/v.
How do you calculate percentages online on a calculator?
To calculate a percentage of a percentage, convert both percentages to fractions of 100, or to decimals, and multiply them. For example, 50% of 40% is calculated; (50/100) x (40/100) = 0.50 x 0.40 = 0.20 = 20/100 = 20%.
What is mass percentage Class 12?
Class 12 Chemistry Solutions. Mass percentage. Mass percentage. Mass percentage can be defined as the ratio of Mass of the component of the solution and total mass of the solution multiplied by 100%.
How do you calculate the mass of an element in a given mass of a compound?
Now, mass of an element in a given mass of compound = (atomic mass of the element × number of atoms of the element in one molecule of the compound) × given mass of compound / molar mass of the compound.
How do you calculate mass from Newtons?
Plug the numbers you want to convert to mass into the new equation, m = F/a. As an example, we will use an object with a force of 10 N and an acceleration of 2 meters per second squared. Divide the newtons by the rate of acceleration, which will give you the mass of the object.
What is mass maths?
A measure of how much matter is in an object. … Mass is commonly measured by how much something weighs. But weight is caused by gravity, so your weight on the Moon is less than here on Earth, while the mass stays the same.
What is the formula of percent by mass and percent by volume?
Percent by mass (m/m) is the mass of solute divided by the total mass of the solution, multiplied by 100 %. What is the percent by mass of a solution that contains 26.5 g of glucose in 500 g of solution? Percent by volume (v/v) is the volume of solute divided by the total volume of the solution, multiplied by 100 %.
What percent is 20 out of 60?
What is this? Now we can see that our fraction is 33.333333333333/100, which means that 20/60 as a percentage is 33.3333%.
What number is 30% of 200?
30 percent of 200 is 60.
What percentage is 23 out of 38?
Now we can see that our fraction is 60.526315789474/100, which means that 23/38 as a percentage is 60.5263%.
How do you calculate percent abundance of chlorine?
- 34.96 × x + 36.95 × y = 35.43.
- 34.96 × x + 36.95 × (1 – x) = 35.43 (since, y = 1 – x)
- Percentage abundance of Cl-35 = 0.7595 × 100 = 75%
- Percentage abundance of Cl-37 = 0.2405 × 100 = 24.05%
How do we calculate relative atomic mass?
To work out the relative atomic mass of an element, all you need to do is multiply each isotopic mass by its relative abundance, add all the values together and divide by 100.
How do you calculate grams to percentage?
For example, if the recommended daily amount is 45 grams and I eat 1.5 grams, then that converts to a percentage like this: 1.5 ÷ 45 = 0.0333 = 3.33 %.
How do you convert mg/kg to percent?
- percent to mg/kg = 10000 mg/kg.
- percent to mg/kg = 20000 mg/kg.
- percent to mg/kg = 30000 mg/kg.
- percent to mg/kg = 40000 mg/kg.
- percent to mg/kg = 50000 mg/kg.
- percent to mg/kg = 60000 mg/kg.
- percent to mg/kg = 70000 mg/kg.
- percent to mg/kg = 80000 mg/kg.
How do you convert mg dL to percent?
How to Convert Milligram/Deciliter to Percent (mg/dL to %) By using our Milligram/Deciliter to Percent conversion tool, you know that one Milligram/Deciliter is equivalent to 0.001 Percent. Hence, to convert Milligram/Deciliter to Percent, we just need to multiply the number by 0.001.
How do you find the percentage of multiple percentages?
To find the average percentage of the two percentages in this example, you need to first divide the sum of the two percentage numbers by the sum of the two sample sizes. So, 95 divided by 350 equals 0.27. You then multiply this decimal by 100 to get the average percentage. So, 0.27 multiplied by 100 equals 27 or 27%.
How do you find the percent of a whole number?
- Determine the whole or total amount of what you want to find a percentage for. …
- Divide the number that you wish to determine the percentage for. …
- Multiply the value from step two by 100.
How do you find the percentage of mass in Class 11?
The mass percentage is calculated as the mass of a component divided by the total mass of the mixture, multiplied by 100.
What is meant by mass percentage of solution Class 9?
The solution composition can be described in mass percentage which shows the mass of solute present in a given mass of solution. … For a solution, the mass per cent is described as the grams of solute per grams of solution, multiplied by 100 to get the percentage.



