How is potassium oxide formed
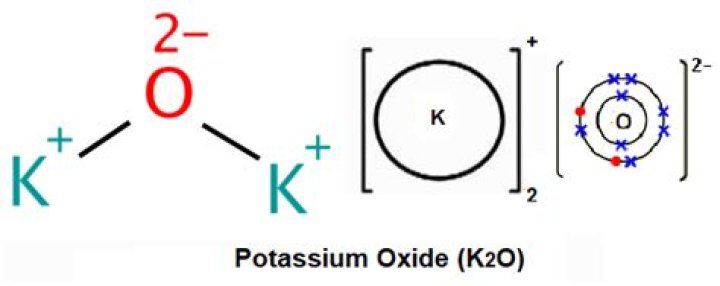
Potassium oxide is an ionic compound formed by combining potassium and oxygen. It carries the chemical formula K2O. Potassium cannot be found free because it is too reactive. It has valency +1 and combines readily with oxygen atoms forming K2O.
Is potassium oxide a metal?
Potassium oxide is a metal oxide with formula K2O. It is a potassium salt and a metal oxide.
Does potassium oxide occur naturally?
Occurrence: Potassium oxide is not found in nature. It is prepared from one of the methodologies described below. Physical properties: Potassium oxide is a yellow, odorless solid.
Is potassium oxide a peroxide?
NamesRelated potassium oxidesPotassium oxide Potassium superoxide Potassium ozonideWhat is the chemical name of Mg3N2?
Magnesium nitride | Mg3N2 | 99.6 (Ca <200 ppm) | -325 mesh (typ.
What happens when you mix potassium and oxygen?
Potassium reacts with oxygen to form K2O2 and KO2 only.
What is KCl name?
Potassium chloride, KCl, is a naturally occurring potassium salt that, aside from its use as fertilizer, is also a raw material for the production of other important potassium compounds.
Is potassium oxide a catalyst?
In this work, silica supported potassium oxide acting as a heterogeneous catalyst was developed for the dehydration of 2-picolinamide to produce 2-cyanopyridine without any additives. … The K2O/SiO2 catalyst was prepared by incipient wetness impregnation method.Where can potassium oxide found?
Potassium oxide can be found in cottonseed and tamarind, which makes potassium oxide a potential biomarker for the consumption of these food products. Potassium oxide (K2O) is an ionic compound of potassium and oxygen.
What happens when potassium peroxide reacts with water?Reacts exothermically with water (or moisture in the air) to give oxygen and a caustic solution, potassium hydroxide [NFPA 491M] .
Article first time published onWhat is the chemical name of 2H2O?
Heavy water (2H2O) The number 2 indicates that there are two atoms of hydrogen in a molecule of water. There is also one atom of oxygen but the number one is omitted from a chemical formula. Water molecule (formally known as dihydrogen monoxide) is composed of two hydrogen atoms and one oxygen atom.
What happens when potassium reacts with water?
When potassium is added to water, the metal melts and floats. It moves around very quickly on the surface of the water. The hydrogen ignites instantly. The metal is also set on fire, with sparks and a lilac flame.
Where is potassium found in nature?
Because potassium reacts so readily with water, it is not found in its elemental form in nature. Instead it is found in various minerals such as sylvite, carnallite, langbeinite, and kainite. Most minerals that contain potassium are referred to as potash.
Is sodium man made?
As such sodium is found naturally only in compounds and never as the free element. Even so it is highly abundant, accounting for around 2.6 per cent of the earths crust by weight.
What happens when you add water to magnesium nitride?
Magnesium nitride reacts with water to produce magnesium hydroxide and ammonia gas, as do many metal nitrides.
Is baking soda a compound or element?
Sodium bicarbonate (IUPAC name: sodium hydrogencarbonate), commonly known as baking soda or bicarbonate of soda, is a chemical compound with the formula NaHCO3.
What happens when magnesium is treated with dinitrogen?
The chemical formula for this reaction is Mg+N2→MgN2.
What is potassium phosphate made of?
They’re made of ions of potassium (K+) and dihydrogen phosphate (H2PO4−), hydrogen phosphate (HPO42−), and orthophosphate (PO43−), respectively.
Is potassium chloride a poison?
Side effects The typical amounts of potassium chloride found in the diet appear to be generally safe. In larger quantities, however, potassium chloride is toxic. The LD 50 of orally ingested potassium chloride is approximately 2.5 g/kg, or 190 grams (6.7 oz) for a body mass of 75 kilograms (165 lb).
Is KCl a salt acid or base?
The ions from KCl derive from a strong acid (HCl) and a strong base (KOH). Therefore, neither ion will affect the acidity of the solution, so KCl is a neutral salt.
What Colour is potassium oxide?
Potassium monoxide appears as a white-colored crystalline solid.
What type of oxide are formed by metal?
Metals tend to form basic oxides, non-metals tend to form acidic oxides, and amphoteric oxides are formed by elements near the boundary between metals and non-metals (metalloids).
Who Discovered potassium?
Potassium, 19 In 1807 Sir Humphry Davy discovered the element potassium which is why his image was included. The experiment shown at the bottom of the artwork is the reaction of potassium and water forming potassium hydroxide and hydrogen gas.
Which type of oxide is water?
An amphoteric solution is a substance that can chemically react as either acid or base. See section above on Properties of Amphoteric Oxides for more detail. Water as such is a neutral stable molecule.
What is the name of the compound made from lithium and chlorine?
PubChem CID433294StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaClLi or LiClSynonymsLITHIUM CHLORIDE 7447-41-8 Lithium chloride (LiCl) Lithiumchloride LiCl More…
What does potassium oxide and nitric acid make?
The equation for the reaction is: HNO3 + KOH KNO3 + H2O A student investigated the temperature change in this reaction.
Is potassium oxide hazardous?
contact can severely irritate and burn the skin and eyes with possible eye damage. lungs causing coughing, wheezing and/or shortness of breath. No occupational exposure limits have been established for Potassium Oxide. However, it may pose a health risk.
Is potassium oxide a good conductor of electricity?
Potassium Oxide is a highly insoluble thermally stable Potassium source suitable for glass, optic and ceramic applications. Oxide compounds are not conductive to electricity.
What is the pH value of potassium oxide?
pH = 14 – pOH = 14 – 1.48 = 12.52. Hope it helps.
What is magnesium peroxide used for?
Magnesium peroxide is a stable oxygen releasing compound, which is used in agricultural and environmental industries. It is used to reduce contaminant levels in groundwater. Magnesium peroxide is used in the bioremediation of contaminated soil and can improve the soil quality for plant growth and metabolism.
Why is na2o2 yellow?
It is yellow in colour due to the presence of unpaired electrons in the molecule. When these unpaired electrons absorb some energy, these get excited and then show colours which are in visible range. The peroxide has yellow -white to yellow colour which is due to presence of unpaired electrons.