How is Prussian blue prepared
Prussian blue was first synthesized about 1704 by the reaction of salts of iron in the +2 oxidation state (ferrous salts) with potassium ferrocyanide; the initial product, an insoluble white compound called Berlin white, was then oxidized to the blue pigment.
How do you make a Prussian blue solution?
- Deparaffinize and hydrate sections to distilled water.
- Mix equal parts of hydrochloric acid and potassium ferrocyanide prepared immediately before use. …
- Wash in distilled water, 3 changes.
- Counterstain with nuclear fast red for 5 minutes.
- Rinse twice in distilled water.
What type of stain is Prussian blue?
The stain is an important histochemical stain used to demonstrate the distribution and amount of iron deposits in liver tissue, often in the form of a biopsy. Perls’s procedure may be used to identify excess iron deposits such as hemosiderin deposits (hemosiderosis) and in conditions such as hereditary hemochromatosis.
How are Prussian blue and Turnbull blue compound prepared?
KFeII[FeIII(CN)6] , KFeIII[FeII(CN)6] Hint: Turnbull’s blue is produced when a ferrous salt reacts with ferricyanide. … On the other hand, the Prussian’s blue is formed when a ferric ion is treated with a ferrocyanide.Which pigments are stainable by Prussian blue reaction?
Iron is stored in hepatocytes in the form of ferritin, ferric iron bound to the protein apoferritin. Excess ferritin aggregates form hemosiderin, which is observed as gold-brown granules and can be confirmed with the Prussian blue reaction.
What is the formula of turnbulls blue?
Turnbull’s blue | C6Fe2KN6 – PubChem.
What is Prussian blue test in chemistry?
The pigment Prussian blue consists of iron cations, cyanide anions, and water. The empirical formula—minus the water of crystallization—is Fe7(CN)18. … Thus, the formula that gives a truer idea of its composition is Fe4[Fe(CN)6]3. Its formal name is iron(III) hexacyanoferrate(II).
What is formula of ferrous Ferricyanide?
Ferrous Ferricyanide. Formula: Fe3[Fe(CN)6]2.Is Prussian Blue water Soluble?
It contains ferric hexacyanoferrate(II) in a cubic lattice crystal structure. It is insoluble in water but also tends to form a colloid thus can exist in either colloidal or water-soluble form, and an insoluble form.
Is Prussian blue similar to ultramarine?Ultramarine blue does tend to be very similar to Prussian blue but you have to check the hue of your ultramarine blue as there are different shades of the color available.
Article first time published onWhy Prussian blue is deeply Coloured?
The intense blue color of Prussian blue is associated with the energy of the transfer of electrons from Fe(II) to Fe(III). Many such mixed-valence compounds absorb certain wavelengths of visible light resulting from intervalence charge transfer.
How does Prussian blue stain work?
PRINCIPLE: The reaction occurs with the treatment of sections in acid solutions of ferrocyanides. Any ferric ion (+3) in the tissue combines with the ferrocyanide and results in the formation of a bright blue pigment called ‘Prussian blue” or ferric ferrocyanide.
Where is Prussian blue found?
Prussian Blue was discovered by Diesbach and Dippel between 1704 and 1707, but most probably in 1706 in Berlin. Written evidence indicates that Prussian Blue was produced at least between 1708 and 1716 in Berlin by Diesbach and Frisch, and that it was mainly sold by Frisch.
What is Perls reaction?
In Perls’ Prussian or Berlin blue reaction (Max Perls, 1843–81), the section is treated with dilute hydrochloric acid to release ferric ions from binding proteins. These ions will react with potassium ferrocyanide to produce an insoluble blue compound.
What is the principle of Perls method?
The principle of Pearl’s Prussian blue reaction is that potassium ferrocyanide will form ferric ferrocyanide (Prussian blue) with reactive ferric salts in an acid solution. Dilute hydrochloric acid liberates loosely bound ferric iron from protein. For many years we used Gomori’s method for demonstrating ferric iron.
Is hemochromatosis stained by Prussian blue?
A Prussian blue iron stain demonstrates the blue granules of hemosiderin in hepatocytes and Kupffer cells. Hemochromatosis can be primary (the cause is probably an autosomal recessive genetic disease) or secondary (excess iron intake or absorption, liver disease, or numerous transfusions).
Is Prussian blue an organic pigment?
Prussian blue is a synthetic organic pigment of deep blue hue with greenish tint.
Who invented Prussian blue?
Discovered in the early 18th century, Prussian Blue revolutionised an art industry starved of a stable blue pigment to rival the prohibitively expensive ultramarine. The creation of Prussian blue was the result of a simple error by two German alchemists, Jacob Diesbach and Johann Konrad Dippel.
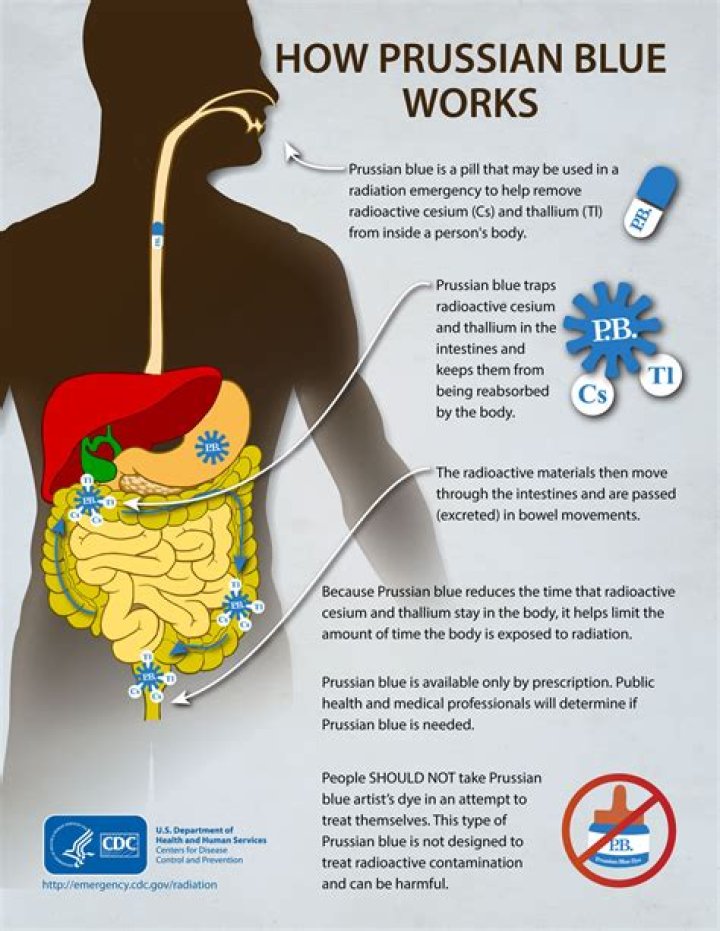
What does Prussian blue cure?
Prussian blue is used to treat radioactive cesium or thallium, or non-radioactive thallium poisoning. It works by combining with thallium and cesium in the intestines. The combination is then removed from the body through the stools.
What is Prussian blue soluble in?
Prussian blue soluble is a water soluble dye and is potentially used to detect iron in tissue sections as well as tracking mesenchymal stem cells. Solubility : Soluble in water (10 mg/ml), DMSO, and DMF.
What two colors make Prussian blue?
- Phthalo Blue (Green Shade)
- Raw Umber.
- Phthalo Green.
What happens if you touch cyanide?
Apart from causing acute poisoning, cyanide can cause reactions to the skin due to the irritant nature of cyanide and thus causing an irritant dermatitis termed as “cyanide rash”, which is characterized by itching, vesiculation and disruption of the skin as seen in our case.
Is it legal to have cyanide?
Possessing sodium cyanide is not illegal because it is used in mining to extract gold and for other industrial purposes.
Does cyanide exist on its own?
Cyanide is a rapidly acting, potentially deadly chemical that can exist in various forms. Cyanide can be a colorless gas, such as hydrogen cyanide (HCN) or cyanogen chloride (CNCl), or a crystal form such as sodium cyanide (NaCN) or potassium cyanide (KCN).
What is Colour of ferro Ferricyanide?
Treatment of ferrocyanide with ferric-containing salts gives the intensely coloured pigment Prussian blue (sometimes called ferric ferrocyanide and ferrous ferricyanide).
What is the Colour of Ferricyanide?
NamesAppearancedeep red crystals, sometimes small pellets, orange to dark red powderDensity1.89 g/cm3, solidMelting point300 °C (572 °F; 573 K)Boiling pointdecomposes
What is the difference between ferrocyanide and ferricyanide?
As nouns the difference between ferrocyanide and ferricyanide. is that ferrocyanide is (chemistry) any of various salts containing the tetravalent anion fe(cn)64-; used in making blue pigments while ferricyanide is (chemistry) any of various salts containing the trivalent anion fe(cn)63-; used in making blue pigments.
What can I substitute for Prussian blue?
Winsor Blue was created as a stable and lightfast version to replace Prussian blue.
Which is darker Prussian blue or Phthalo blue?
Prussian blue is, as a compound, Ferric hexacyanoferrate. Phthalo blue is a copper phthalocyanine compound that I can’t decisively find a name for. They’re chemically very different as pigments. Prussian blue is SUPPOSED to be darker and less saturated and bright than Phthalo blue.
Is Prussian blue watercolor transparent?
DANIEL SMITH Prussian Blue is lightfast and transparent, with medium to high staining strength. It disperses and diffuses easily and evenly and can be considered interchangeable with the higher staining Phthalo Blue in application, although Prussian Blue is slightly greener.
What color is Prussia?
Prussian blue, also known as Berlin blue, is a dark blue colour that is artificially made. It is one of the first pigments made synthetically. It was accidentally found in 1704 by two chemists in Berlin. The dark blue uniforms of the Prussian army were dyed this colour.