How is van der Waals force formed
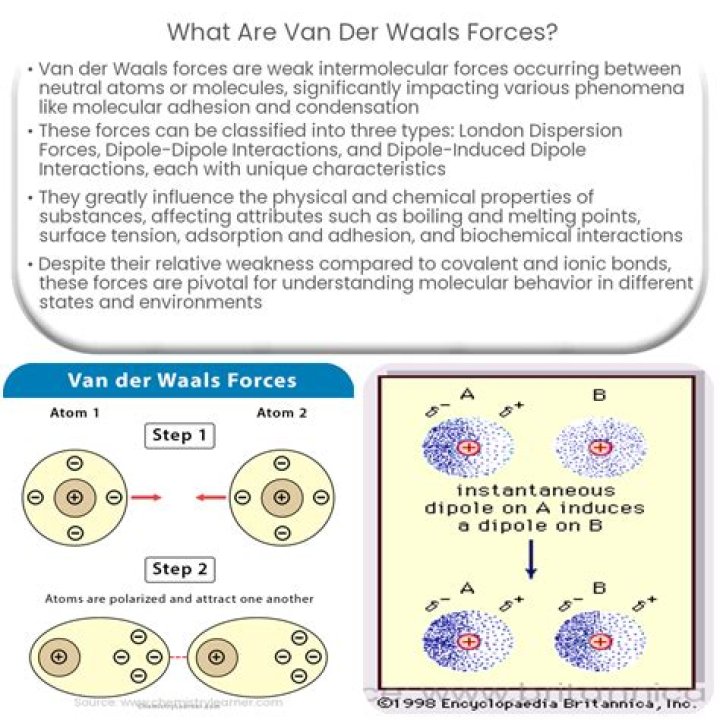
Van der Waals forces are electrostatic forces caused by a temporarily fluctuating dipole moment arising from a brief shift of orbital electrons to one side of an atom or molecule, which creates a similar shift in adjacent atoms or molecules (Lutzow et al., 2006).
Why are van der Waals forces?
15.1. Van der Waals forces occur because while neutral molecules have no net charge or permanent dipole moment, they do have a dynamic distribution of charge. As two molecules approach, this charge distribution can become favorable for interaction between the two molecules (Figure 15.8A).
How do van der Waals forces hold molecules together?
Van der Waals forces form electrostatic bonds between molecules. … The intermolecular forces are much weaker than the internal forces that hold atoms together in molecules, but they are still strong enough to affect the behavior and properties of many materials.
What is van der Waals force with example?
Van der Waals forces are the sum of the attractive and repulsive electrical forces between atoms and molecules. … Examples of van der Waals forces include hydrogen bonding, dispersion forces, and dipole-dipole interactions.What causes a dipole dipole force?
Dipole -dipole interactions occur when the partial charges formed within one molecule are attracted to an opposite partial charge in a nearby molecule. Polar molecules align so that the positive end of one molecule interacts with the negative end of another molecule.
What is van der Waals forces for kids?
Van der Waals’ forces are the weakest type of intermolecular force. They are named after the Dutch scientist Johannes Diderik van der Waals (1837–1923). Negatively charged electrons orbit molecules or ions. The electrons create slightly different charges from one end of the molecule to the other.
What is Vander Waal's force of attraction explain their types?
Van Der Waals forces are the interactions between atoms and molecules that result in a pull between them. … The molecules do not contain any charge. These interactions or bonds comprise of three types, such as dipole-dipole, hydrogen bonds and London dispersion forces.
How are ionic bonds and van der Waals forces different?
Ionic bonds are strong electrostatic attraction forces formed between positive and negative ions. … Like hydrogen bonds, van der Waals forces rely on dipoles, a difference in charge between two molecules.How do Van der Waal forces work?
van der Waals forces, relatively weak electric forces that attract neutral molecules to one another in gases, in liquefied and solidified gases, and in almost all organic liquids and solids. … An additional attractive force results from the interaction of a permanent dipole with a neighbouring induced dipole.
What is weak van der Waal forces?Van der Waals forces are weak intermolecular forces that are dependent on the distance between atoms or molecules. These forces arise from the interactions between uncharged atoms/molecules. … They are known to rapidly vanish when the distance between the interacting molecules increases.
Article first time published onWhich molecules have dipole-dipole forces?
Dipole–dipole forces occur between molecules with permanent dipoles (i.e., polar molecules). For molecules of similar size and mass, the strength of these forces increases with increasing polarity. Polar molecules can also induce dipoles in nonpolar molecules, resulting in dipole–induced dipole forces.
Are dipole-dipole forces intermolecular or intramolecular?
Dipole–dipole interactions are a type of intermolecular attraction—attractions between two molecules.
How are dispersion forces different from dipole-dipole interactions?
The main difference between dipole-dipole and London dispersion forces is that dipole-dipole forces occur among molecules with dipole moment whereas London dispersions occur due to instantaneous dipoles that form in atoms or nonpolar molecules.
What are van der Waals dispersion forces?
What are Van der Waals Forces? Van der Waals forces are weak forces between molecules that occur due to either temporary or permanent dipoles. They are also called dispersion forces. The dipoles (either temporary or permanent) cause attraction between the molecules.
Which interaction produces dispersive forces by creating temporary dipoles that induce dipoles in other molecules?
Which interaction produces dispersive forces by creating temporary dipoles that induce dipoles in other molecules? D. Induced dipole-induced dipole.
Is Van der Waals a covalent bond?
Covalent compounds exhibit van der Waals intermolecular forces that form bonds of various strengths with other covalent compounds. The three types of van der Waals forces include: 1) dispersion (weak), 2) dipole-dipole (medium), and 3) hydrogen (strong). … These compounds typically form medium to strong bonds.
Are van der Waals forces the same as London forces?
London dispersion force is the weak intermolecular force that results from the motion of electrons that creates temporary dipoles in molecules. The London dispersion force is sometimes called a ‘Van der Waals force.
Are van der Waals forces intermolecular or intramolecular?
Van der Waals forces are a category of intermolecular forces that includes London dispersion and dipole-dipole interactions. Some sources also consider hydrogen bonding to be a Van der Waals force.
Do Van der Waals forces occur in polar molecules?
Van der Waals forces are weak interactions between molecules that involve dipoles. Polar molecules have permanent dipole-dipole interactions. Non-polar molecules can interact by way of London dispersion forces.
Why are intermolecular forces such as Van der Waals forces important to biological function?
Like hydrogen bonds, van der Waals interactions are weak attractions or interactions between molecules. … These bonds—along with ionic, covalent, and hydrogen bonds—contribute to the three-dimensional structure of proteins that is necessary for their proper function.
Is an ionic bond a van der Waal force?
van der Waals forces are short-range intermolecular forces, specifically the interaction of loose, fluctuating PARTIAL charges, i.e. temporary dipoles… … Ionic bonds are interactions of FULL charges, i.e. due to complete transfer of valence electrons. They are permanent charges, and mostly static in time.
Is electrostatic force an intermolecular force?
Though we consider electrostatic forces as intermolecular forces (like dipole-dipole interaction and hydrogen bond), but they’re not strong enough to make a bond. Whereas the strong electrostatic forces between ions result in formation of ionic compounds.
Why is hydrogen bond stronger than Wonderwall forces?
Hydrogen bonds are stronger than the van der Waals forces because H-bonds are considered as an extreme form of dipole-dipole interaction.
How strong are van der Waals forces?
A van der Waals interaction is a relatively weak force ranging from 0.5 to 1 kcal/mol and is nonionic in nature. Neutral molecules containing electronegative atoms, like oxygen and nitrogen, have a tendency to draw the electron cloud toward itself through the covalent bond from its less electronegative neighbor atom.
What affects the strength of van der Waals forces?
Factors affecting van der Waals forces Size of atoms: (strength of VWF increases as size increases e.g., He, Ne, Ar, Kr and Ra) Boiling point and melting point increases down the group because of increase in strength in these forces. 2. Electronegativity of elements.
Why do van der Waals forces increase with size of molecule?
– When a dipole molecule and polar molecules come closer to each other than the positive pole of the dipole attracts negative charge of polar molecules. … The strength of van der waal’s forces increases due to the increase in the number of electrons present and the size of the molecules.
Which molecule will engage in the strongest dispersion forces?
The dispersion forces are strongest for iodine molecules because they have the greatest number of electrons. The relatively stronger forces result in melting and boiling points that are the highest of the halogen group.
How do you identify dipole-dipole forces?
You have a dipole moment when there is a difference in electronegativity between two atoms.
Do dipole-dipole forces only occur in polar molecules?
Dipole-dipole forces are the attraction forces that exist between the partially positive pole of one polar molecule and the partially negative pole of another. So, yes all polar molecules will form dipole-dipole forces (non-polar molecules only form weak London dispersion forces).
What is the difference between van der Waals and dipole-dipole?
Dipole-dipole force are a type of Van Der Waals force. When two polar molecules interact, opposite partial charges attract, similarly to ionic bonding, but generally weaker, because of the smaller charge magnitude.
How are intermolecular forces different from covalent bonds?
This is why many explanations usually take covalent bonds vs intermolecular forces, as covalent bonds rely on sharing of a pair of electrons to form a “physical” bond. Whereas intermolecular forces rely on a “force” to bring atoms or molecules together.