How many electrons are in a shell
Energy shellMaximum number of electronsThird8
How many electrons are there in the shell Answer?
So, for each element K shell can hold up to 2 elements, L shell can hold up to 8, M shell can hold up to 18 electrons and N shell can hold up to 32 electrons.
How many electrons are in each shell Class 9?
Each shell can hold only a specific number of electrons. The general formula to determine the number of electrons is 2(n2), where n is the shell number. The first shell can have two electrons, the second shell can contain up to 8 electrons and the third shell can hold 18 electrons and so on.
How many electrons are in the 5 shell?
Energy Level (Principal Quantum Number)Shell LetterElectron Capacity2L83M184N325O50How many electrons are in the oxygen shell?
There are 6 electrons present in L shell of oxygen element.
What is the M shell?
Definition of M-shell : the third innermost shell of electrons surrounding an atomic nucleus — compare k-shell , l-shell.
Is there a 5th shell?
The 5th shell has 5s, 5p, 5d, 5f, and 5g orbitals. An s orbital can hold 2, p holds 6, d holds 10, f holds 14, and g holds 18 (which you are not expected to know for this class). If you add them up, you get 50 electrons that the 5th shell can hold. This also follows the 2n^2 rule.
Why does the third shell have 8 electrons?
The electron capacity of the third shell is 8, when there are no shells above it. And that is the case for all elements in the third period. It is only when there are outer shells surrounding it that the third (or higher) shell has a higher capacity.How many electrons are in the 7th shell?
In reality, there aren’t any atoms on the periodic table that have 50 electrons in their fifth shell (nor any that have 72 electrons in their 6th shell, or 98 electrons in their 7th shell, etc.) Due to the peculiarities of quantum mechanics, electrons start filling higher shells before the lower shells are “complete”.
How many electrons can m shell hold?M-shell is the third energy level and can have a maximum of 18 electrons. Complete step by step answer: In an atom, the electrons surround the nucleus in different orbitals or energy levels.
Article first time published onHow are atom shells filled?
- Determine the number of electrons that the atom has.
- Fill the s orbital in the first energy level (the 1s orbital) with the first two electrons.
- Fill the s orbital in the second energy level (the 2s orbital) with the second two electrons.
How do you find number of electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How many electrons are in the 4th Shell?
n=ShellMaximum Number of Electrons11st Shell222nd Shell833rd Shell1844th Shell32
How many 5s orbitals exist in a given atom?
That is 25 orbitals in level n=5.
How many individual orbitals are there in the fifth shell?
3 subshells in 3rd shell and 25 orbitals in 5th shell.
What is the fifth shell called?
Shell nameSubshell nameSubshell max electronsO5s25p65d105f14
How many shells do atoms have?
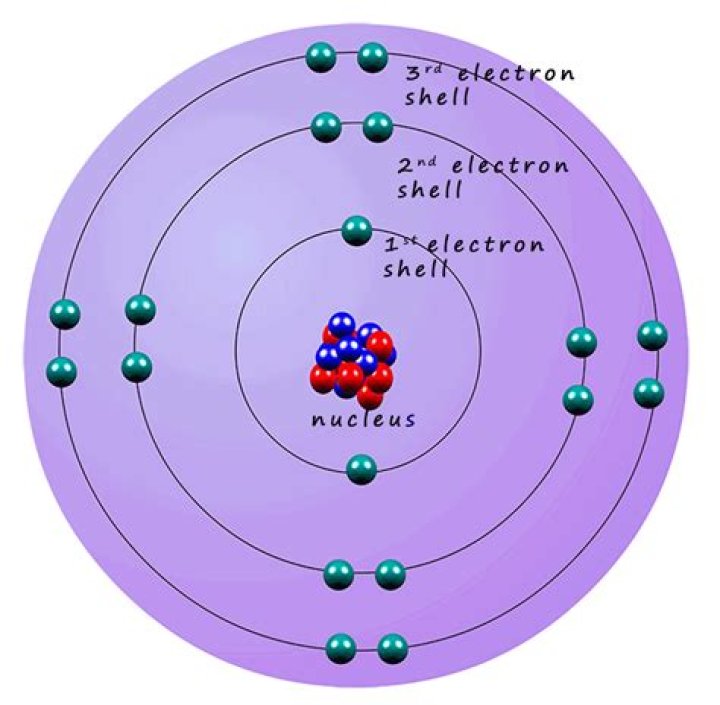
Energy shellMaximum number of electronsFirst2Second8Third8
What is N shell?
the fourth shell of electrons surrounding the nucleus of an atom and containing, when filled, 32 electrons having principal quantum number 4. …
Why is it called K shell?
The names of the electron shell were given by a spectroscopist named Charles G Barkla. He named the innermost shell has k shell because he noticed that the X-rays emitted two types energies. … He noticed that K type X-rays emitted the highest energy. Therefore, he named the innermost shell as the K shell.
What sublevel comes after 5s?
The 4d sublevel is filled next, after the 5s sublevel.
How many electrons can level 2?
For example, energy level I can hold a maximum of two electrons, and energy level II can hold a maximum of eight electrons. The maximum number depends on the number of orbitals at a given energy level. An orbital is a volume of space within an atom where an electron is most likely to be found.
Does 7f orbital exist?
For any atom, there are seven 7f orbitals. … Three of the orbitals are common to both sets. These are are the 7fxyz, 7fz 3, and 7fz(x 2-y 2) orbitals. The higher f-orbitals (8f, 9f, …) are more complex since they have more spherical nodes while the lower orbitals (4f, 5f, and 6f) have fewer.
What is the 2 8 8 rule in chemistry?
There is a 2-8-8 rule for these elements. The first shell is filled with 2 electrons, the second is filled with 8 electrons, and the third is filled with 8. You can see that sodium (Na) and magnesium (Mg) have a couple of extra electrons. They, like all atoms, want to be happy.
Is the M shell full?
the third shell of electrons surrounding the nucleus of an atom and containing, when filled, 18 electrons having principal quantum number three. … Compare K-shell, L-shell, N-shell.
How many electrons can KLMN shells fill?
In K, L,M, N shells,in the N shell it can accommodate 32 electrons.
Is shell and Orbit same?
Orbit is a well-defined circular path around the nucleus in which electrons revolve around the nucleus. It is also called a shell. It is denoted by the principal quantum number ‘n’. Electrons can jump from one orbit to another by either absorbing energy or releasing energy.
How many electrons are in a atom?
If we gots an electrically neutral atom, then if there are the 8 positively charged, massive particles, i.e. protons, and there are by SPECIFICATION, THERE MUST be 8 electrons in the NEUTRAL ATOM….and these electrons are conceived to whizz about the nuclear core in all sorts of fancy orbits, with fancy energies.
How many electrons are in a mole?
One mole of anything is 6.022 x 10^23 particles. That’s what a mole is and 6.022 x 10^23 is the Avogadro number. So, 1 mole of electrons is 6.022 x 10^23 electrons.
What is the 4th energy level?
The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons. There is a 4d orbital with 10 electrons which coincides with the 5th energy level of the periodic table.
How many electrons does N 4 L 3 have?
Fourteen electrons can be represented by the values n = 4 and l = 3.
What is an outer shell electron?
The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons.



