How many enantiomers are possible
There are two pairs of enantiomers. Any given molecule has its enantiomer; the two other molecules are its diastereomers.
How do you find the number of enantiomers?
- If ‘n’ is even (here n is the number of chiral centres): Number of enantiomers=2n−1. Number of meso compounds=2n/2−1. …
- If ‘n’ is odd: Number of enantiomers=2n−1−2(n−1)/2. Number of meso compounds=2(n−1)/2.
How are enantiomers possible?
Like platin, it has two each of two groups attached to the central atom. … However, if four different groups are attached to a tetrahedral atom, the four groups can be arranged in two possible ways. The two compounds that result are mirror images of each other. These two isomers are called enantiomers.
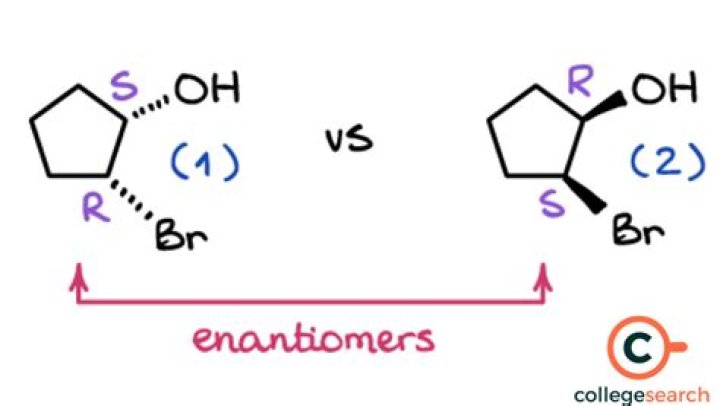
Can there be more than two enantiomers for a molecule?
(2) There can never be more than two enantiomers for a molecule. (1) Complete hydrolysis of a oligosaccharide produces only monosaccharides. (2) Fructose, galactose, and ribose are all hexoses.Is there only one enantiomer?
In some jurisdictions, single-enantiomer drugs are separately patentable from the racemic mixture. It is possible that only one of the enantiomers is active. Or, it may be that both are active, in which case separating the mixture has no objective benefits, but extends the drug’s patentability.
How many stereoisomers are possible for Ketohexose?
Answer: There are three nonequivalent stereogenic centers, so a total of eight stereoisomers are possible.
How many enantiomers are possible for glucose?
There are two enantiomers of glucose, called D-glucose and L-glucose. The D-enantiomer is the common sugar that our bodies use for energy. It has n = 4 stereocenters, so therefore there are 2n = 24 = 16 possible stereoisomers (including D-glucose itself).
How many chiral centers do enantiomers have?
D-glucose and L-glucose are enantiomers, because they differ at all four chiral centers.Which compound can exist as enantiomers?
Cis-2-butene.
Can enantiomers never be Epimers?(1) Cellulose and amylose are glucose polymers which differ in the type of glycosidic linkage present. (2) All monosaccharides are either aldoses and ketoses. (3) Enantiomers can never be epimers.
Article first time published onCan enantiomers have more than one chiral center?
Can enantiomers have more than one chiral center? – Quora. Yes, indeed. If a molecule has n chiral centres, it can exist in the form of a maximum of 2^n stereoisomers, arranged as half that number pairs of enantiomers.
How do you find enantiomers?
Bottom line for today: you can tell if molecules are enantiomers or diastereomers by looking at their (R,S) designations. Enantiomers are non-superimposable mirror images of each other. Pardon me while I push the Caps Lock button: ENANTIOMERS ALWAYS HAVE OPPOSITE R,S DESIGNATIONS.
How are enantiomers different?
Enantiomers differ in their configuration (R or S) at the stereogenic center. … Enantiomers have identical chemical and physical properties in an achiral environment. Enantiomers rotate the direction of plane polarized light to equal, but opposite angles and interact with other chiral molecules differently.
What are enantiomers made of?
Enantiomer. Enantiomers are mirror images of each other. That is, the same parts of the molecule are located in opposite positions, like the thumbs on a glove, so that they cannot be superimposed on each other but look the same in a mirror.
Are enantiomers isomers?
Enantiomers are two optical isomers (i.e. isomers that are reflections of each other). … Compounds that are enantiomers of each other have the same physical properties except for the direction in which they rotate polarized light and how they interact with different optical isomers of other compounds.
Why do enantiomers have different smells?
Figure 2: R and S enantiomers of carvone. … The chirality of carvone is directly translated into a discrepancy in smell because several olfactory receptors in your nose are chiral and will register certain enantiomers more strongly than others. Thus, (R) carvone smells like spearmint and (S) carvone smells like caraway.
How many total stereoisomers are possible?
Each time we add a chiral center to a molecule, we double the possible number of stereoisomers. With 1 chiral center, there are 2 isomers, 2 chiral centers, 4 possible isomers, 3 centers, 8 isomers and 4 centers, 16 possible stereoisomers.
How many diastereomers of D-glucose are possible?
Therefore, 14 diastereomers are there of glucose.
How many stereoisomers are possible for fructose?
For fructose, there are only three asymmetric carbons, so only 8 or 23 stereoisomers can be produced.
How many enantiomeric pairs are possible for an aldohexose?
Aldohexoses have four chiral centers and, therefore, 24 = 16 isomers (Figure 3). There will be eight d-isomers and eight l-isomers. Therefore, the number of enantiomer pairs is 8 (2n−1).
How many enantiomeric pairs are possible for a linear ketohexose?
In its linear structure, a ketohexose contains the carbonyl carbon center at the position 2 or 3. Therefore, in the linear structure, a ketohexose contains three chiral centers and eight possible stereoisomers. This means four pairs of enantiomers are possible for this compound.
How many stereoisomers are possible for aldohexose?
The aldohexoses have four chiral centres for a total of 16 possible aldohexose stereoisomers (24).
Do enantiomers have the same solubility?
Enantiomers have the same solubility in achiral solvents (ethanol), but different solubility in chiral solvent (D-2-butanol). Rate & Extent of Reaction of Enantiomer is the same with an achiral reactant but different with another chiral reactant.
What are enantiomers examples?
Enantiomers are chemical isomers that are non-superimposable mirror images of each other. … Moreover, these types of stereoisomers can be considered as mirror images of each other. A common example of a pair of enantiomers is dextro lactic acid and laevo lactic acid, whose chemical structures are illustrated below.
Do enantiomers have the same density?
All Enantiomers have the same boiling point, the same melting points, and the density, they have the same chemical and physical properties.
Do all chiral molecules have enantiomers?
There are two types of stereoisomers: enantiomers and diastereomers. Enantiomers are pairs of stereoisomers which are mirror images of each other: thus, A and B are enantiomers. It should be self-evident that a chiral molecule will always have one (and only one) enantiomer: enantiomers come in pairs.
Can a meso compound have a diastereomer?
Meso compounds are achiral (optically inactive) diastereomers of chiral stereoisomers.
Is erythro syn or anti?
Thus, the substituents in the erythro isomer have an anti orientation, but are syn in the threo isomer. The syn-anti nomenclature may be applied to acyclic compounds having more than two chiral centers, as illustrated by the example in the colored box.
What is the most important monosaccharide?
The most important monosaccharide for living things is glucose. It is the main source of chemical energy used for cell functions in all organisms, from bacteria to humans.
How many drugs are chiral?
In pharmaceutical industries, 56% of the drugs currently in use are chiral molecules and 88% of the last ones are marketed as racemates (or racemic mixtures), consisting of an equimolar mixture of two enantiomers.
How many stereoisomers are possible for dihydroxyacetone?
1). A carbon atom that has four different groups attached is a chiral carbon. If a molecule contains one or more chiral carbons, it is likely to exist as two or more stereoisomers. Dihydroxyacetone does not contain a chiral carbon and thus does not exist as a pair of stereoisomers.



