How many shells does lithium have
Energy shellMaximum number of electronsSecond8Third8
How many shells are present in lithium?
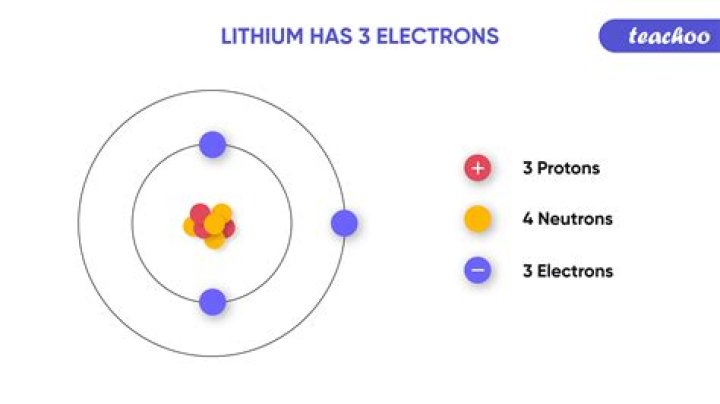
So… for the element of LITHIUM, you already know that the atomic number tells you the number of electrons. That means there are 3 electrons in a lithium atom. Looking at the picture, you can see there are two electrons in shell one and only one in shell two.
Does lithium have outer shell?
Therefore, the third electron in lithium goes into a second shell, that is, an outer shell. As a consequence of its electronic structure, lithium is the lowest atomic number element that is a metal. In a general sense, metals are elements that normally have only 1–3 electrons in their outer shells.
How many shells does each element have?
Energy shellMaximum number of electronsFirst2Second8Third8What Subshells does Li have?
Two of the lithium electrons can fit into the 1s subshell, but the third electron must go into the second shell. The second shell has two subshells, s and p, which fill with electrons in that order. The 2s subshell holds a maximum of 2 electrons, and the 2p subshell holds a maximum of 6 electrons.
How many shells does neon have?
Because neon has two atomic shells, it needs two electrons in the first and eight to fill the second.
How many shells does be have?
So… for the element of BERYLLIUM, you already know that the atomic number tells you the number of electrons. That means there are 4 electrons in a beryllium atom. Looking at the picture, you can see there are two electrons in shell one and two in shell two. ► More about the history and places to find beryllium.
Can an atom have 8 shells?
ElementElectrons per shell42, 262, 474, 384, 2, 2Are there only 4 shells?
There is no upper limit to the number of shells an atom can have. This even applies to a hydrogen atom. That’s right, a hydrogen atom has n = 1 , 2, 3, 4 all the way to infinity.
Are orbitals shells?The main difference between shell subshell and orbital is that shells are composed of electrons that share the same principal quantum number and subshells are composed of electrons that share the same angular momentum quantum number whereas orbitals are composed of electrons that are in the same energy level but have …
Article first time published onHow many valence shells Does lithium have?
Lithium has a single electron in the second principal energy level and so we say that lithium has one valence electron. Beryllium has two valence electrons.
How many atoms are in lithium?
1 mole of Lithium contains 6.022140857×1023 atoms.
What is the group number of lithium?
Group1180.50°C, 356.90°F, 453.65 KBlocks0.534Atomic number36.94State at 20°CSolid7LiElectron configuration[He] 2s17439-93-2
How many bonds can Lithium make?
Lithium has 2 valence electrons in the product because it can make only 1 bond with hydrogen. Beryllium has 4 valence electrons in the product because it can only make 2 bonds. It has fewer electrons than neon and can react with electron donors.
What does lithium need to do to get a full outer shell?
Atoms with very few VALENCE electrons, such as Lithium, would need to gain many electrons for a full shell so it is easier for them to LOSE electrons and become POSITIVE.
How many shells does hydrogen have?
Consequently, hydrogen only has 1 outer shell as it has less than 2 electrons so it does not need another shell.
How many shells are there in the world?
Estimates range from 70,000 to 120,000 known species of shell dwellers. Usually, these creatures can be broken down into two groups: bivalves, which have two shells connected by a hinge, and gastropods, which have one shell and no hinge.
What is the M shell?
Definition of M-shell : the third innermost shell of electrons surrounding an atomic nucleus — compare k-shell , l-shell.
How many shells does fluorine have?
That means there are 9 electrons in a fluorine atom. Looking at the picture, you can see there are two electrons in shell one and seven in shell two. ► More about the history and places to find fluorine.
How many shells are in helium?
Helium only has one atomic shell, which fills up when it has two electrons.
How many shells does radon have?
Classification:Radon is a noble gas and a nonmetalProtons:86Neutrons in most abundant isotope:136Electron shells:2,8,18,32,18,8Electron configuration:[Xe] 4f14 5d10 6s2 6p6
What is SPDF rule?
There are different orbital shapes (s,p,d,f) Each orbital can only hold 2 electrons max. There is a hierarchy, i.e. s orbitals will be filled before p orbitals which will be filled before d orbitals and so on. (s<p<d<f) (note, this is a general rule but there are exceptions)
How many atoms can the 4th shell hold?
ShellSubshellTotal Number of Electrons in Shell3rd Shell3s, 3p, 3d2 + 6 + 10 = 184th Shell4s, 4p, 4d, 4f2 + 6 + 10 + 14 = 32
Why is KLM shell named?
He named the innermost shell has k shell because he noticed that the X-rays emitted two types energies. These energies were named as type A that is higher energy X-ray and type B that is lower energy X-ray. … He noticed that K type X-rays emitted the highest energy. Therefore, he named the innermost shell as the K shell.
Is Lithium unstable?
Lithium is a relatively unstable element because its stable isotopes have a very low binding energy level. This means they are not prone to fission, which is breaking down.
Which element is usually nonreactive?
Noble gases are nonreactive, nonmetallic elements in group 18 of the periodic table. As you can see in the periodic table in the figure below, noble gases include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn).
How many shells does iodine have?
Atomic Number53Mass Number127Number of Neutrons74Shell structure (Electrons per energy level)[2, 8, 18, 18, 7]Electron Configuration[Kr] 4d10 5s2 5p5
What are the 4 types of orbitals?
There are four basic types of orbitals: s, p, d, and f. An s orbital has a spherical shape and can hold two electrons.
What are the 4 orbitals letters?
Named for their energy sublevels, there are four types of orbitals: s, p, d, and f. Each orbital type has a unique shape based on the energy of its electrons.
What are sub shells?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.
How many valence E does CL have?
A: An atom of a group 17 element such as chlorine has seven valence electrons. It is “eager” to gain an extra electron to fill its outer energy level and gain stability.