How much acid is in acid rain
Typical acid rain has a pH value of 4.0. A decrease in pH values from 5.0 to 4.0 means that the acidity is 10 times greater. There are many high-tech devices that are used to measure pH in laboratories.
How many acids are present in acid rain?
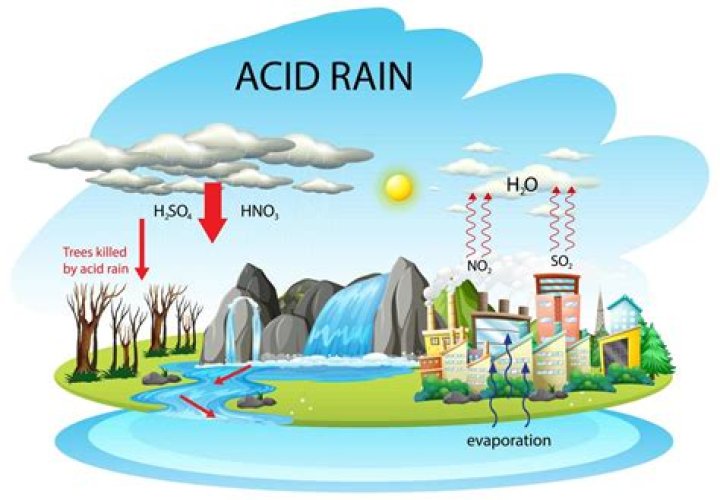
The two strong acids present in the acid rain are nitrogen oxide and sulphur dioxide. These two gases react with water in the atmosphere and oxygen in the air to form sulphuric acid and nitric acid.
Is acid rain a strong acid?
Acid rain is a result of air pollution. … Some of these gases (especially nitrogen oxides and sulphur dioxide) react with the tiny droplets of water in clouds to form sulphuric and nitric acids. The rain from these clouds then falls as very weak acid – which is why it is known as “acid rain”.
Is there acid in acid rain?
Acid rain describes any form of precipitation that contains high levels of nitric and sulfuric acids. It can also occur in the form of snow, fog, and tiny bits of dry material that settle to Earth. Normal rain is slightly acidic, with a pH of 5.6, while acid rain generally has a pH between 4.2 and 4.4.What happens if acid rain touches you?
Very strong acids will burn if they touch your skin and can even destroy metals. Acid rain is much, much weaker than this; it is never acidic enough to burn your skin. Rain is always slightly acidic because it mixes with naturally occurring oxides in the air.
Which acid is present in rainwater?
Most is accounted for by the presence of sulfuric acid (H2SO4) in rainwater.
What is acid rain in BYJU's?
Acid Rain as the name suggests can be said to be the precipitation of acid in the form of rain in the simplest manner. When atmospheric pollutants like oxides of nitrogen and sulphur react with rainwater and come down with the rain, then this results in Acid Rain.
What is acid rain for kids?
Acid rain is a form of air pollution. When coal and petroleum are burned in automobiles, electric power plants, and factories, they release certain harmful gases into the air. … When the water in the air comes down as rain, sleet, hail, or snow, it carries with it these gases. This is known as acid rain.What pH is rainwater?
Normal, clean rain has a pH value of between 5.0 and 5.5, which is slightly acidic. However, when rain combines with sulfur dioxide or nitrogen oxides—produced from power plants and automobiles—the rain becomes much more acidic. Typical acid rain has a pH value of 4.0.
What is pH full form?The letters pH stand for potential of hydrogen, since pH is effectively a measure of the concentration of hydrogen ions (that is, protons) in a substance. The pH scale was devised in 1923 by Danish biochemist Søren Peter Lauritz Sørensen (1868-1969).
Article first time published onIs lemon juice more acidic than acid rain?
Each level of the pH scale represents the power of 10. Sulfuric acid measures 1 on the pH scale. It is 10 times more acidic than lemon juice and 100 times more acidic than orange juice (10 x 10). … Normal rain is slightly acidic naturally with a pH of 5.6. This is because the rain combines with carbon dioxide in the air.
What pH level is milk?
Cow’s milk Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it’s acid-forming or alkaline-forming.
What is the pH of human blood?
Blood is normally slightly basic, with a normal pH range of about 7.35 to 7.45. Usually the body maintains the pH of blood close to 7.40.
Has acid rain killed anyone?
Acid rainfall can cause serious repertory problems and greatly impact human health. It has been estimated that around 550 premature deaths each year occur due to acid rain.
Can you drink acid rain?
Humans are affected when we breathe in air pollution, this can cause breathing problems, and even cancer. Drinking water which has been contaminated with acid rain can cause brain damage over time.
Is acid rain rare?
Acid rain was rare among environmental problems in that it had a viable solution, and these days it’s often hailed as an environmental success story. The market worked as intended, sulfur and nitrogen emissions declined and rain became less acidic.
What is acid rain Vedantu?
Rainfall is declared as acid rain when its pH is less than 5.6 because natural and unpolluted rainwater actually has a pH between 5.6-6.5 (acidic) due to the reaction of water with the presence of CO2. … The pH of acid rain can range between 5.6 to 3.5 and in some cases, pH can go even lower than 2.
What is acid rain for class 11th?
(i) Acid rain refers to the rainfall and other forms of precipitation with a pH of less than 5. (ii) Oxides of sulphur and nitrogen are released from automobile exhausts, industries, power plants, etc.
What is acid rain Class 7 Ncert?
What is the effect of acid rain? Answer: The rain containing excess of acids is called acid rain. It is very acidic because carbon dioxide, sulphur dioxide and nitrogen dioxide present in it which dissolve in rain drops to form carbonic acid, sulphuric acid and nitric acid respectively.
Why is rain water acidic?
The extra acidity in rain comes from the reaction of air pollutants, primarily sulfur oxides and nitrogen oxides, with water in the air to form strong acids (like sulfuric and nitric acid). The main sources of these pollutants are vehicles and industrial and power-generating plants.
Is acid rain still a problem in 2020?
The quick version: Yes, acid rain is still around, and yes it’s still a problem. … Rain is naturally slightly acidic, since picks up carbon dioxide in the air, producing carbonic acid. But when it starts absorbing industrial pollutants like sulfur dioxide and nitrogen oxide, the acidity becomes troublesome.
Which acid is present in Apple?
Fruit acidity in cultivated apples is majorly determined by malic acid, which accounts for up to 90% of total organic acids [6]. Citric acid also exists in mature apple fruits; however, it exhibits a very low to undetectable concentration in cultivated apple [14,15].
Is tap water basic or acidic?
Water, including tap water, has a pH of 7, or neutral. Alkaline water has a pH of up to 8 or 9. It can be naturally alkaline (for example, spring water can contain minerals that make it more alkaline), or it can be ionized to make it that way.
Is DI water acidic?
The pH of distilled water is actually slightly acidic. This is because as soon as distilled water comes into contact with the air, carbon dioxide gas dissolves in it and creates a dilute solution of carbonic acid.
Is seawater acidic or basic?
Seawater is slightly basic (meaning pH > 7), and ocean acidification involves a shift towards pH-neutral conditions rather than a transition to acidic conditions (pH < 7).
Is acid fog real?
The pollutants are picked up by atmospheric moisture that condenses into fog. The nitrogen and sulfur compounds then oxidize into nitric acid and sulfuric acid.” There are people right now who live in the acid fog belt in places like Salinas Valley, CA.
How do we stop acid rain?
A great way to reduce acid rain is to produce energy without using fossil fuels. Instead, people can use renewable energy sources, such as solar and wind power. Renewable energy sources help reduce acid rain because they produce much less pollution.
What is acid rain 4th grade?
Acid rain is rain that has been made acidic by certain pollutants in the air. Acid rain is a type of acid deposition, which can appear in many forms. Wet deposition is rain, sleet, snow, or fog that has become more acidic than normal.
Who invented pH?
Søren Sørensen. In 1909 Sørensen, a Danish chemist, introduced the concept of pH as a convenient way of expressing acidity.
Why is the pH of water 7?
pH is a measure of the amount of Hydrogen ions (H+) in a solution. … Even in pure water ions tend to form due to random processes (producing some H+ and OH- ions). The amount of H+ that is made in pure water is about equal to a pH of 7. That’s why 7 is neutral.
Is a pH of 7 acidic?
pH is a measure of how acidic/basic water is. The range goes from 0 – 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. … pHs less than 7 are acidic while pHs greater than 7 are alkaline (basic).