How would water h20 be classified
It is an oxygen hydride, a mononuclear parent hydride and an inorganic hydroxy compound. It is a conjugate base of an oxonium. It is a conjugate acid of a hydroxide. Water is h2O, a clear, colorless, odorless, tasteless liquid that freezes into ice below 0 degrees centigrade and boils above 100 degrees centigrade.
Is water H2O an element or compound?
For example, water (H2O) is a compound consisting of two hydrogen atoms bonded to an oxygen atom. The atoms within a compound can be held together by a variety of interactions, ranging from covalent bonds to electrostatic forces in ionic bonds.
Is H2O a basic compound?
As the proton donor, H2O acts as a Brønsted-Lowry acid. In this reaction, the water molecule accepts a proton from HC2H3O2, becoming H3O+(aq). As the proton acceptor, H2O is a Brønsted-Lowry base.
Is h20 pure substance or mixture?
Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. Although water is the most abundant substance on earth, it is rarely found naturally in its pure form. Most of the time, pure water has to be created.What is structure of H2O?
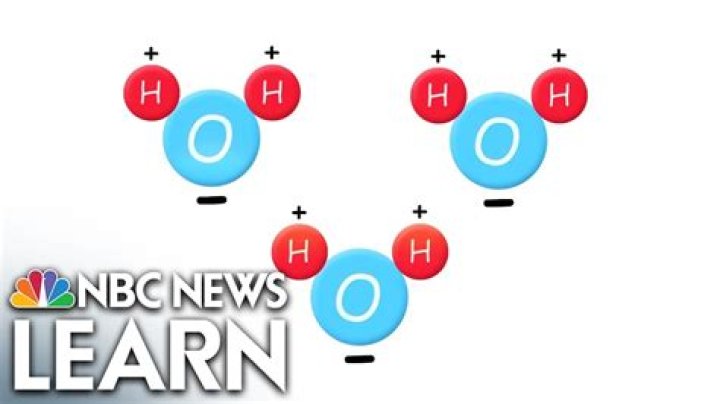
H2O is the molecular formula of water, one of the major constituents of the Earth. A single molecule is made up of two hydrogen atoms and one oxygen atom, which are bonded through the covalent bond. Moreover, two or more H2O molecules connect with the help of hydrogen bonds to form a compound.
Is water homogeneous or heterogeneous?
Water – another example of homogeneous mixture; all but the purest water contains dissolved minerals and gases; These are dissolved throughout the water, so the mixture presents in the same phase and is homogeneous.
How do you read H2O in chemistry?
H2O is the chemical formula of water. It means that each molecule of water is made up of two hydrogen atoms, indicated by the letter H, and a single oxygen atom, represented by the letter O.
Is water considered a chemical?
water, a substance composed of the chemical elements hydrogen and oxygen and existing in gaseous, liquid, and solid states. It is one of the most plentiful and essential of compounds. A tasteless and odourless liquid at room temperature, it has the important ability to dissolve many other substances.How many compounds are in H2O?
How many different elements are in a water molecule quizlet? The two elements that make up a molecule of water are hydrogen and oxygen; there are two hydrogen atoms and one oxygen atom, resulting in the chemical formula H2O.
Why water is considered as a pure substance?Water is a pure substance because it only contains one type of molecule. Pure water contains only molecules that are a combination of one oxygen atom…
Article first time published onWhy water is considered as a compound not a mixture?
Answer: Water is a compound and not a mixture because Its properties are completely different from that of oxygen and hydrogen, its constituent elements.
Is water H2O a mixture?
The true pure water (H2O) is a compound because the constituents are bond chemically in a certain fixed proportion. H2O is not a mixture because a mixture doesn’t have a definite ratio of composition.
Is H20 a neutral?
Water is considered neutral because the concentration of hydrogen and hydroxide ions is the same.
Is H20 neutral oxide?
H2O , NO , CO are neutral oxides.
Is H20 neutral or acidic?
Because it has a pH of 7, water is considered to be neutral. It is neither an acid nor a base but is the reference point for acids and bases.
Why is h20 not linear?
In water, the oxygen atom has two lone pairs. These two lone pairs repel the hydrogen-oxygen bonded pairs so much that the molecule is at its lowest energy arrangement when the H-O-H bond angle is 104.5 degrees. As a result, the water molecule can be classified as non-linear.
How does the water molecule differ from other compounds?
Water behaves differently from most other chemical compounds. In almost all substances the atoms and molecules move closer together as they get colder. … Its oxygen atom (O) and the two hydrogen atoms (H) are asymmetrically arranged. This produces a dipole, a molecule with one negatively and one positively charged end.
What is h20 commonly known as?
H. 2. O is the chemical formula for water, meaning that each of its molecules contains one oxygen and two hydrogen atoms.
Is H2O ionic or covalent?
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. When you look at a diagram of water (see Fig. 3-2), you can see that the two hydrogen atoms are not evenly distributed around the oxygen atom.
How is the compound H2O different from the elements hydrogen and oxygen?
The difference is due to the fact that one water molecule has two hydrogen atoms to one oxygen atom, as explained above. This means it takes two water molecules (2 H2O) to make one oxygen molecule (O2). At the same time, however, two molecules of water (2 H2O) can make two molecules of hydrogen (2 H2).
What is difference between homogenous and heterogenous?
1. A homogenous mixture is that mixture in which the components mix with each other and its composition is uniform throughout the solution. A heterogenous mixture is that mixture in which the composition is not uniform throughout and different components are observed. 2.
Is water an example of heterogeneous mixture?
Examples of heterogeneous mixtures include sand, oil and water, and chicken noodle soup.
Is water a element compound heterogeneous or homogeneous?
MaterialPure Substance or MixtureElement, Compound, Homogeneous, Heterogeneouspure water (H2O)Pure SubstanceCompound,chromium (Cr)Pure SubstanceElement,Chex mixMixtureHeterogeneoussalt + pure water (NaCl + H2O)MixtureHomogeneous
What is h20 on the periodic table?
H2O is the chemical formula of water. It means that each molecule of water is made up of two hydrogen atoms, indicated by the letter H, and a single oxygen atom, represented by the letter O.
What are the chemical properties of H2O?
PropertiesChemical formulaH2OMolar mass18.01528(33) g/molOdourNoneDensitySolid: 0.9167 g/ml at 0 °C Liquid: 0.961893 g/mL at 95 °C 0.9970474 g/mL at 25 °C 0.9998396 g/mL at 0 °C
Is water wet article?
Answer 1: Liquid water is not itself wet, but can make other solid materials wet. Wetness is the ability of a liquid to adhere to the surface of a solid, so when we say that something is wet, we mean that the liquid is sticking to the surface of a material.
What is water made of elements?
A water molecule has three atoms: two hydrogen (H) atoms and one oxygen (O) atom. That’s why water is sometimes referred to as H2O.
How is water different from its elements in its properties?
Hydrogen is a gas , oxygen is a gas whereas water formed is in liquid state. Also hydrogen does not support combustion, oxygen supports combustion whereas water stops combustion.
Which of the following samples of water can be termed as pure water?
Pure water is called distilled water or deionized water.
Is water an example of pure substance?
A substance that has a fixed chemical composition throughout is called a pure substance such as water, air, and nitrogen. A pure substance does not have to be of a single element or compound.
How is water a compound class 9?
Water is a compound because it is made up of water molecules. There is no such thing as water atoms. Water molecules are made of hydrogen and oxygen atoms, in the definite proportion of two hydrogens for one oxygen.