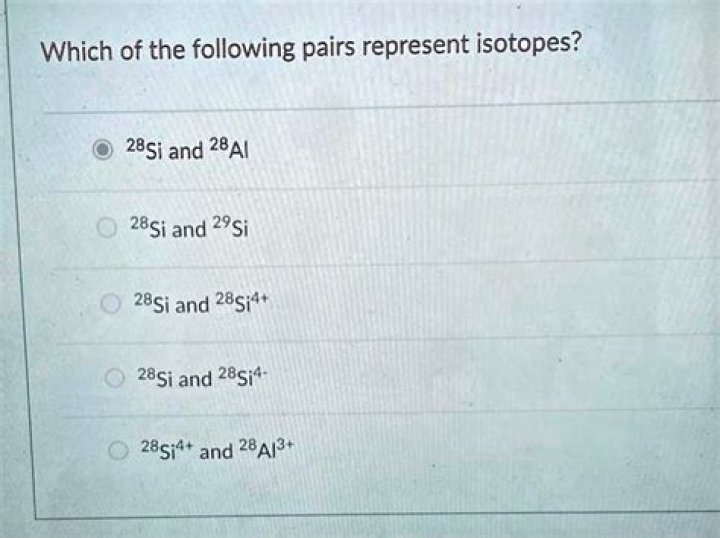
Is 28si and 28si 4 an isotope
Publisher Summary. There are four isotopes of silicon (Si) exist in the natural environment—28Si, 29Si, 30Si and 32Si. The first three isotopes are stable isotopes and the last one is radiogenic. The relative abundance of 28Si, 29Si and 30Si is 92.23%, 4.67% and 3.10%, respectively.
What is 14si?
Silicon (14Si) has 23 known isotopes, with mass numbers ranging from 22 to 44. … The longest-lived radioisotope is 32Si, which is produced by cosmic ray spallation of argon.
What is the mass of 29si?
Silicon-29 atom is the stable isotope of silicon with relative atomic mass 28.9764947, 4.683 atom percent natural abundancy, and nuclear spin (1)/2.
Is SI-30 the most abundant?
Silicon has nine isotopes, with mass numbers from 25-33. Si (the most abundant isotope, at 92.23%), 29Si (4.67%), and 30Si (3.1%) are stable; 32Si is a radioactive isotope produced by argon decay.What is the normal phase of silicon?
Classified as a metalloid, Silicon is a solid at room temperature.
Is oxygen a radioactive element?
Oxygen isotope species Stable isotopes are variants of atoms that differ in mass but do not decay over time, that is, they are not radioactive. The element oxygen (O) is found in three naturally occurring stable isotopes, 18O, 17O, and 16O.
Is Aluminium an isotope?
Isotopes: Natural aluminum is comprised of two isotopes: 26Al and 27Al. Twenty radioactive isotopes have been produced under laboratory conditions ranging from 21Al to 42Al. Aluminum-26 is a radioactive isotope containing 13 neutrons.
Is silicon and silica the same thing?
Silica, or Silicon dioxide, is an oxide of silicon, as the name suggests. It has a chemical formula SiO₂ and is mainly found naturally in Quartz and diverse living organisms. You will discover silica as one of the significant constituents of sand in many parts of the world.What are the isotopes of chlorine?
There are two stable isotopes, 35Cl (75.77%) and 37Cl (24.23%), giving chlorine a standard atomic weight of 35.45. The longest-lived radioactive isotope is 36Cl, which has a half-life of 301,000 years. All other isotopes have half-lives under 1 hour, many less than one second.
What is the proton electron and neutron of the silicon 28 28Si atom?Properties of Silicon-28 Isotope:SILICON-28Relative Isotopic Mass27.97692654Neutron Number (N)14Atomic Number (Z)14Mass Number (A)28
Article first time published onWhat is the difference between silica and silicon?
Silicon and silica are two terms often used in inorganic chemistry. Silicon is the second most abundant element on the earth, second only to oxygen. … The main difference between silicon and silica is that silicon is an element whereas silica is a compound.
What is the natural abundance of 28si?
Natural abundance of Si-28 is 92.23% and of Si-30 is 3.1%.
What are the three silicon isotopes?
Silicon has three stable isotopes: 28Si, 29Si, and 30Si with respective abundances of 92.23%, 4.67%, and 3.1%. Si is a primary nucleus that exists owing to the nuclear oxygen-burning processes within stars whose initial compositions are hydrogen and helium rich, the two left elements from the Big Bang.
What is the half life of silicone?
PubChem CID6337619Molecular Weight30.9753632DatesModify 2021-12-11 Create 2005-08-09DescriptionSilicon-31 atom is the radioactive isotope of silicon with relative atomic mass 30.975363, half-life of 2.62 hours and nuclear spin (3)/2. ChEBI
How many protons are in 29si?
Properties of Silicon-29 Isotope:SILICON-29Nucleon Number (A)29Proton Number (Z)14Half-lifeStableSpin0.5
What is the atomic number of uranium 235?
GroupActinides1135°C, 2075°F, 1408 KPeriod74131°C, 7468°F, 4404 KBlockf19.1Atomic number92238.029State at 20°CSolid234U, 235U, 238U
What is gallium mass number?
Atomic mass of Gallium is 69.723 u.
What is the flammability of silicon?
Silicone is not considered to be flammable. It can catch fire and burn but requires high temperatures to do so. The ignition temperature of silicone is 842 degrees Fahrenheit (450 Celsius).
Why is silicon called silicon?
The name silicon derives from the Latin silex or silicis, meaning “flint” or “hard stone.” Amorphous elemental silicon was first isolated and described as an element in 1824 by Jöns Jacob Berzelius, a Swedish chemist. Impure silicon had already been obtained in 1811.
Is Si a conductor?
Silicon is a semiconductor, meaning that it does conduct electricity. Unlike a typical metal, however, silicon gets better at conducting electricity as the temperature increases (metals get worse at conductivity at higher temperatures).
Is aluminum a metal?
Aluminium is a silvery-white, lightweight metal. It is soft and malleable. Aluminium is used in a huge variety of products including cans, foils, kitchen utensils, window frames, beer kegs and aeroplane parts. … It is the second most malleable metal and the sixth most ductile.
What are the 3 isotopes of aluminum?
Aluminium or aluminum (13Al) has 22 known isotopes from 22Al to 43Al and 4 known isomers. Only 27Al (stable isotope) and 26Al (radioactive isotope, t1/2 = 7.2×105 y) occur naturally, however 27Al comprises nearly all natural aluminium.
Does aluminum have many isotopes?
Aluminum has nine isotopes whose mass numbers range from 23 to 30. Only 27Al (stable isotope) and 26Al (radioactive isotope; t1/2 = 0.72×106 yr) occur naturally. Al is produced from argon in the atmosphere by spallation caused by cosmic-ray protons.
What are the 4 radioactive elements?
- Alpha Radiation. Alpha radiation is a type of energy released when certain radioactive elements decay or break down. …
- Uranium. Uranium is a radioactive element that can be found in soil, air, water, rocks, plants and food. …
- Radium. …
- Radon. …
- Polonium.
What does radioactive mean?
1 : the giving off of rays of energy or particles by the breaking apart of atoms of certain elements (as uranium) 2 : the rays or particles that are given off when atoms break apart. radioactivity. noun.
What is the most common radioactive element?
Polonium. Because it is a naturally-occurring element that releases a huge amount of energy, many sources cite polonium as the most radioactive element.
How do chlorine isotopes differ?
There are three isotopes of chlorine that differ from each other according to the number of neutrons in their atomic nucleus. The key difference between chlorine 35 and 37 is that chlorine 35 has 18 neutrons per atomic nuclei, whereas chlorine 37 has 20 neutrons per atomic nuclei.
Why does chlorine have two different isotopes?
Adding or removing a proton from an atom’s nucleus changes that atom’s atomic number and creates a different element. Chlorine-35 and chlorine-37 are both isotopes of the element chlorine. … Since all atoms of chlorine contain 17 protons, chlorine-35 and chlorine-37 differ in the number of neutrons each one has.
What is Cl 36 used for?
Basic Principles of 36Cl Chlorine-36 has been widely used for dating old groundwater. The 36Cl used for this purpose is produced in the atmosphere by the interaction of cosmic rays with argon atoms. The 36Cl then mixes with ordinary atmospheric chloride (mostly derived from the ocean).
Is silicon harmful to humans?
Silicon is non-toxic as the element and in all its natural forms, nameli silica and silicates, which are the most abundant. … Silicon may cause chronic respiratory effects. Crystalline silica (silicon dioxide) is a potent respiratory hazard.
Is silica harmful to humans?
Inhaling crystalline silica can lead to serious, sometimes fatal illnesses including silicosis, lung cancer, tuberculosis (in those with silicosis), and chronic obstructive pulmonary disease (COPD). In addition, silica exposure has been linked to other illnesses including renal disease and other cancers.