Is a binary compound always Ionic

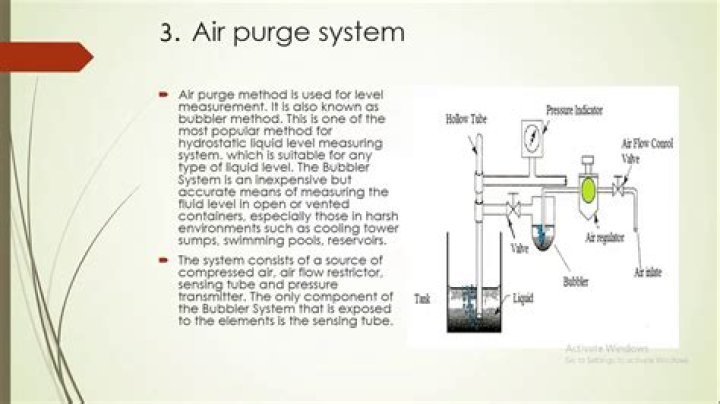
Number of Atoms4tetra-5penta-6hexa-7hepta-
Is binary compound ionic or covalent?
Binary molecular (covalent) compounds are formed as the result of a reaction between two nonmetals. Although there are no ions in these compounds, they are named in a similar manner to binary ionic compounds.
What is a binary non ionic compound?
A second type is a binary nonionic compound. These compounds are composed of two different nonmetallic elements, although the number of atoms of the individual elements can vary. … The prefixes mono, di, tri, tetra, penta, etc., are used to indicate 1, 2, 3, 4, 5, etc., atoms of a particular element.
Are binary compounds and ionic compounds the same?
The key difference between ionic and binary compounds is that ionic compounds contain two charged components whereas binary compounds contain two different chemical elements. Ionic compounds are binary compounds that come under two different categories.What compounds are considered binary?
Examples of binary compounds include water (H2O), carbon monoxide (CO), hydrochloric acid (HCl), sodium chloride (NaCl), and silicon dioxide (SiO2).
What is the difference between binary and non binary ionic compounds?
Ionic binary compounds- compounds in which two types of ions (metal cation and nonmetallic anion) combine to form an ionic compound. … Non binary compounds are those made up of either same elements such as O2, H2 etc or more than two different elements such as NH4Cl, H3PO4, etc.
What constitutes binary compound?
In chemistry, a binary compound is something consisting of precisely two elements. … A few examples of chemical elements are hydrogen, oxygen, and iron. This means that a binary compound will be composed of two different chemical elements. In a binary compound, there may be only one of each element.
What is the difference between binary ionic and binary molecular compounds?
Binary ionic compounds contain two elements: one metal and one nonmetal. … Binary molecular compounds contain two elements and both are nonmetals.How do you tell if a compound is binary or polyatomic?
BRIAN M. A polyatomic ion is a group of atoms bond by covalent bonds with an unbalanced charge. A binary ionic compound is composed of atoms of two elements bond by an ionic bond.
What makes a binary compound ionic?A binary ionic compound is composed of ions of two different elements – one of which is a metal, and the other a nonmetal. For example, sodium iodide, NaI, is composed of sodium ions, Na+ (elemental sodium is a metal), and iodide ions, I- (elemental iodine is a nonmetal).
Article first time published onIs NaCl a binary ionic compound?
Binary Compounds – They are the compounds formed from the metal cation and the nonmetallic anion, such as sodium chloride – NaCl. Most ionic compounds are binary compounds, or compounds formed from just two elements.
How do you tell if a compound is ionic or molecular?
As a general rule of thumb, compounds that involve a metal binding with either a non-metal or a semi-metal will display ionic bonding. Compounds that are composed of only non-metals or semi-metals with non-metals will display covalent bonding and will be classified as molecular compounds.
Are binary ionic compounds neutral?
Binary Covalent Compounds (Type III) These compounds are always neutral (not ions which have charges), and consist of only two elements (see acid naming below for compounds containing only non-metal elements, but with more than two elements.
How do you name non binary ionic compounds?
- Name the metal by its elemental name.
- Name the nonmetal by its elemental name and an -ide ending.
- Name metals that can have different oxidation states using roman numerals to indicate positive charge. Example Fe2+ is Iron(II) …
- Name polyatomic ions by their names.
How do you identify a binary covalent compound?
- Name the non-metal furthest to the left on the periodic table by its elemental name.
- Name the other non-metal by its elemental name and an -ide ending.
- Use the prefixes mono-, di-, tri-…. to indicate the number of that element in the molecule.
How are all the cations different in each type of binary compounds?
How are the cations different in each type of binary compounds? In Type 1 cations, the compounds are named after their parent atom. Type 2 binary compounds the cation have a Roman numeral that indicates the charge as well as the name of its parent atom.
Which of the following are examples of ionic compounds?
- LiF – Lithium Fluoride.
- LiCl – Lithium Chloride.
- LiBr – Lithium Bromide.
- LiI – Lithium Iodide.
- NaF – Sodium Fluoride.
- NaCl – Sodium Chloride.
- NaBr – Sodium Bromide.
- NaI – Sodium Iodide.
Is CaS A binary compound?
alkaline earth chalcogenides form ionic binary crystals such as barium oxide (BaO), calcium sulfide (CaS), barium selenide (BaSe), or strontium oxide (SrO). They have the same structure as sodium chloride, with each atom having six neighbours.
Why are compounds in Table 1 called binary acids?
Binary acids are acids composed of two atoms, a hydrogen element and a nonmetal element. Hydroiodic acid, hydrofluoric acid, and hydrobromic acid are binary acids because they have a hydrogen element and nonmetal element (iodine, fluorine and bromine) bonded together.
Can binary ionic compounds have polyatomic ions?
Indeed, most ionic compounds contain polyatomic ions. Well-known examples are sodium hydroxide (NaOH) with OH- as the polyatomic anion, calcium carbonate (CaCO3), and ammonium nitrate (NH4NO3), which contains two polyatomic ions: NH+ and NO3-.
What is the difference between binary and multivalent?
Be sure these charges are written on your periodic table! Example- Show the formation of Na2O. Show the formation of CaCl2. … Also write the names of the compounds.
Is CH4 a binary compound?
Binary Ionic Compounds Containing a Metal and a Nonmetal. A binary compound is a compound formed from two different elements. There may or may not be more than one of each element. … CH4 Binary, since there are only two types of atoms. CHCl3 Not binary or diatomic.
What is the only polyatomic ion that is a cation?
What is the only polyatomic ion that is a cation? Ammonium, NH + .
What is the difference between a binary acid and an Oxyacid?
A binary acid consists of hydrogen and one other element. Oxyacids contain hydrogen, oxygen, and one other element. The name of the acid is based on the anion attached to the hydrogen.
Is MgSO4 a binary ionic compound?
Magnesium sulfate is an ionic compound as there is a bond between a metal and a non-metal.
How do you identify ionic compounds?
To find the formula of an ionic compound, first identify the cation and write down its symbol and charge. Then, identify the anion and write down its symbol and charge. Finally, combine the two ions to form an electrically neutral compound.
Is KF ionic or covalent?
The oppositely charged ions form an electrostatic attraction, which is the ionic bond. The compound potassium fluoride (KF) results, and since the potassium and fluoride ions have equal but opposite charges, the compound is neutral (but not the individual ions in the compound).
Which of the following are not ionic compounds?
Answer : Non-ionic compounds are those that have covalent bonds. Hydrochloric acid (HCl) and carbon tetrachloride (CCl4) form covalent due to sharing of a pair of electrons between two atoms. Whereas sodium chloride (NaCl) and potassium chloride (KCl) form ionic compounds as they consist of positive and negative ions.