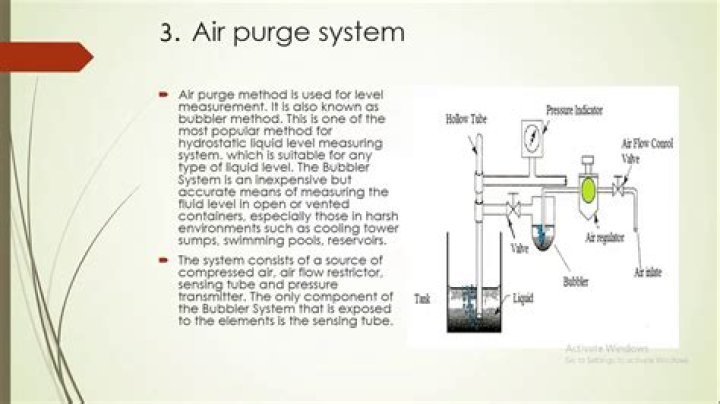
Is alcohol soluble in benzene
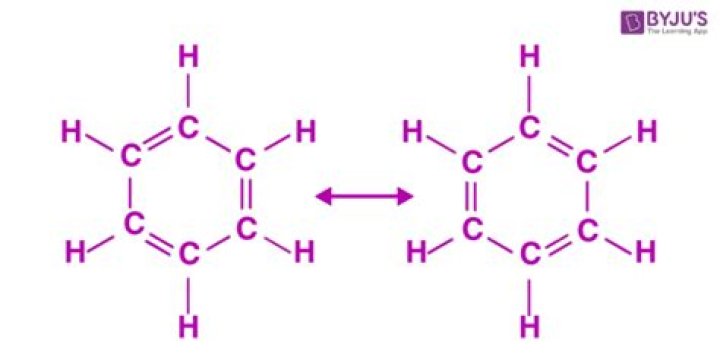
Ethanol is to some degree soluble in benzene. In fact, benzene is used to help to strip off small quantities of water from wet ethanol. This is called azeotropic distillation. This will result in the denatured ethanol
Which is more soluble in benzene?
Benzene is non polar and is therefore insoluble in polar solvents. Water is very polar, so benzene is insoluble in it. Ether is less polar than water, so benzene should be more soluble in ether.
Is methanol soluble in benzene?
AlcoholSolubility (mol/100 g of H 2O)methanolcompletely miscibleethanolcompletely misciblen-propanolcompletely misciblen-butanol0.11
What is not soluble in benzene?
Similarly, a few water molecules will enter the hexane layer because of the water-hexane London forces. But, for the most part, water and benzene are immiscible. They do not dissolve in each other.What is alcohol soluble in?
Because alcohols form hydrogen bonds with water, they tend to be relatively soluble in water. The hydroxyl group is referred to as a hydrophilic (“water-loving”) group, because it forms hydrogen bonds with water and enhances the solubility of an alcohol in water.
Is ethanol or benzene more soluble in water?
Benzene because it can form ion-dipole interactions with water molecules, a bonding interaction much like dipole-dipole interactions but stronger. Ethanol because it can form London forces with water. Ethanol because it can form dipole-dipole interactions with water molecules.
Is benzene miscible in ethanol?
Under standard conditions, ethanol is miscible with water and all common organic solvents (e.g., benzene, hexane, diethyl ether, THF, DMSO, acetonitrile, …) so it will dissolve in arbitrary amounts in either of the two.
Why benzene is not soluble in water?
Water is a polar compound with dipole moment, whereas benzene is a symmetrical compound with zero dipole moment and so is non-polar. When benzene is mixed with water, the molecular attraction increases, and the benzene molecules are no longer able to interact with the water molecules. As a result, it is insoluble.Which pair of liquids are likely to miscible?
Two liquids that appear to mix completely together are said to be miscible. Water and ethanol are one example of a pair of miscible liquids, because you can take any amount of ethanol and mix it with any amount of water and you will always end up with a clear, colorless liquid just like the ones you started with.
Is c6h6 soluble or insoluble?CCDC Number298305Associated ArticleDOI:10.1107/S010876810503747X
Article first time published onWhat is ethanol soluble in?
Ethanol is soluble in water primarily because of the presence of -OH group that allows or enables it to form hydrogen bonds with water molecules. In other words, ethanol is soluble in water because it is a polar solvent.
Is ethyl alcohol and isopropyl alcohol miscible?
Properties. Isopropyl alcohol is miscible in water, ethanol, and chloroform.
Are all alcohols soluble in water?
NameFormulaSolubilityHexanolC6H13OH0.0058HeptanolC7H15OH0.0008MethanolCH3OHmiscibleEthanolC2H5OHmiscible
Are alcohols polar or nonpolar?
Alcohols are polar, since they have oxygen-hydrogen bonds, which allow alcohol molecules to attract each other through hydrogen bonds. Since oxygen atoms are much more electronegative than hydrogen atoms, the oxygen-hydrogen bond is especially polar.
Which is more soluble in water ethanol or propanol?
Methanol is more soluble. As number of carbon atoms increase, solubility decreases. So, Methanol>Ethanol>Propanal>…..and so on.
Which alcohol is least soluble in water?
According to this, 1 − p e n t a n o l will be the least soluble in water, among the other given alcohols.
Is benzene polar or nonpolar?
Benzene is a nonpolar molecule due to the presence of many nonpolar carbon-hydrogen bonds spaced out in equal proportion around the molecule ring. It is a symmetrical molecule in which all the bond dipoles cancel.
Why ethanol is completely soluble in water but benzene is not?
Answer: This is because the water is able to form hydrogen bonds with the hydroxyl group in these molecules, and the combined energy of formation of these water-alcohol hydrogen bonds is more than enough to make up for the energy that is lost when the alcohol-alcohol hydrogen bonds are broken up.
Is ethyl alcohol miscible in water?
Ethanol has small alkyl group. Also like dissolves like. Both ethanol and water are polar molecules are miscible. Therefore, ethanol is miscible with water in all the proportions even if it is an organic compound.
What affects the solubility of alcohols?
Alcohols are soluble in water. … The reason why the solubility decreases as the length of hydrocarbon chain increases is because it is requires more energy to overcome the hydrogen bonds between the alcohol molecules as the molecules are more tightly packed together as the size and mass increases.
Is propanol soluble in water?
You find that the smaller alcohols – methanol, ethanol, and propanol – dissolve easily in water.
Which is more soluble in water?
Complete answer: Among these given compounds, sodium chloride is more soluble in water than potassium chloride. Because the solubility of a compound mainly depends upon the hydration energy and lattice energy. If the hydration energy of the compound is greater than lattice energy, the solubility is more.
Why ethanol and water are miscible but oil and water are immiscible?
Because water molecules are polar, any liquid that does not have polar molecules—such as oil—is usually immiscible with water. Rubbing alcohol molecules have a polar and nonpolar part, which means they are able to form hydrogen bonds with water and therefore able to mix with it.
Is Honey miscible in water?
Honey is soluble in water. Thus, honey and water are miscible liquids.
Is alcohol and oil miscible or immiscible?
When you try to mix water and oil or alcohol and oil, the polar molecules stick together, keeping the oil molecules from getting between them-and the two don’t mix. When you try to mix water and alcohol, they mix fine, since both are made of polar molecules.
Why do sugars dissolve in water and not in benzene?
Why? Sugar dissolves only in polar solvents and since benzene is not a polar solvent , sugar does not dissolve in it. Sugar is soluble only in polar covalent compounds like water, where sugar molecules form hydrogen bonds with water.
Does benzene mix with water?
No such strong intermolecular interaction can exist between benzene and water, so benzene is insoluble in water.
Why is benzene solubility in organic solvents?
Benzene is more able to dissolve many organic substances than a saturated hydrocarbon such as hexane, this is becuase the pi cloud of the benzene ring is able to interact with the pi clouds of various solutes. For example polystyrene will be solvated better by benzene than by hexane for this reason.
What type of solid is C6H6?
The compound C6(CH3)6 is a hydrocarbon (hexamethylbenzene), which consists of isolated molecules that stack to form a molecular solid with no covalent bonds between them.
Is benzene a liquid?
Benzene is a chemical that is a colorless or light yellow liquid at room temperature. It has a sweet odor and is highly flammable.
Why is benzene banned?
Benzene has been banned as an ingredient in products intended for use in the home, including toys. Benzene has a sweet, aromatic, gasoline-like odor. … The odor threshold generally provides adequate warning for acutely hazardous exposure concentrations but is inadequate for more chronic exposures.