Is bismuth stable or unstable
Bismuth was long considered the element with the highest atomic mass that is stable, but in 2003 it was discovered to be extremely weakly radioactive: its only primordial isotope, bismuth-209, decays via alpha decay with a half-life more than a billion times the estimated age of the universe.
Is bismuth the most stable?
At ultralow temperatures, bismuth germanate is more sensitive to the small amount of energy released by exiting alpha particles and creates more noticeable changes in the crystal’s temperature. With bismuth dethroned, lead, the previous element in the periodic charts, now claims the title of heaviest stable element.
Is bismuth a stable nucleus?
Bismuth-209 was long thought to have the heaviest stable nucleus of any element, but in 2003, a research team at the Institut d’Astrophysique Spatiale in Orsay, France, discovered that 209Bi undergoes alpha decay with a half-life of approximately 19 exayears (1.9×1019, approximately 19 quintillion years), over a …
Is lead-214 stable?
Uranium is widely distrib- uted in rocks and soils throughout the earth’s crust. … Thus, radium-226 is one of the decay products of uranium-238. Polonium-218 and lead-214 are decay products of radon-222. The final isotope, lead-206, is stable (non-radioactive) and its formation ends the series.Why is bismuth stable?
Bismuth compounds are more stable in +3 oxidation state because ‘Bi’ exhibits +3 stable oxidation state instead of +5 due to inert pair efffect.
When the nuclide bismuth 214 decays to polonium 214 what kind of decay does bismuth 214 undergo?
The bismuth-214 decays to polonium-214 by beta emission.
Is bismuth a solid at room temperature?
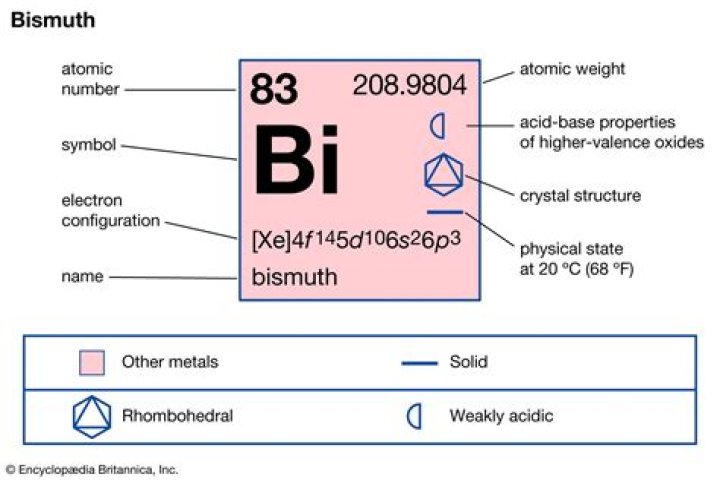
Bismuth is a chemical element with symbol Bi and atomic number 83. Classified as a post-transition metal, Bismuth is a solid at room temperature.
How strong is bismuth?
Bismuth is a post-transition metal. It is one of the strongest diamagnetic metals. It is almost as heavy as lead. Its melting point is quite low 271.5 °C (520.7 °F), which is normal for post-transition metals.Why is bismuth unstable?
Bismuth was long considered the element with the highest atomic mass that is stable, but in 2003 it was discovered to be extremely weakly radioactive: its only primordial isotope, bismuth-209, decays via alpha decay with a half-life more than a billion times the estimated age of the universe.
Is uranium 238 stable?Uranium’s most stable isotope, uranium-238, has a half-life of about 4,468,000,000 years. It decays into thorium-234 through alpha decay or decays through spontaneous fission. Uranium-238 is not fissile, but is a fertile isotope, because after neutron activation it can produce plutonium-239, another fissile isotope.
Article first time published onIs lead 209 stable?
Lead is the element with the heaviest stable isotope, 208Pb. (The more massive 209Bi, long considered to be stable, actually has a half-life of 2.01×1019 years.) A total of 43 lead isotopes are now known, including very unstable synthetic species.
Which process turns RN 218 into Po 214?
The decay of radon into polonium 218 is followed by further alpha decay into lead 214.
When bismuth 214 undergoes beta decay its balanced nuclear equation is?
21483Bi→21484Po+0−1e .
Is Lithium stable or unstable?
Lithium is a relatively unstable element because its stable isotopes have a very low binding energy level. This means they are not prone to fission, which is breaking down. This is why, lithium is less common in the galaxy than some other elements with a higher atomic weight.
What is the half life of bismuth 214?
Half life:19.9 m 4ENSDF citation:NDS 76,127 (1995)Literature cut-off date:1-Jun-1995Author(s):Y.A. AkovaliReferences since cut-off:214Bi decay from 1995-98 (NSR)
Which oxidation state of bismuth is more stable?
The most stable oxidation state of bismuth is +3 due to inert pair effect.
Which is more stable Bi2O3 or Bi2O5?
There fore,the oxides of Bi are basic compound. The oxidation number of Bi in Bi2O3 is +3 and the oxidation number of Bi in Bi2O5 is +5. Higher the oxidation number of the metallic element lower the basicity of the compound. Hence,Bi2O3 is more basic than Bi2O5.
What is the malleability of bismuth?
Fourthly, many bismuth alloys are relatively soft and malleable. Malleable means that a metal can be hammered into thin sheets. Bismuth is alloyed with iron to create what is known as “malleable irons.”
What does bismuth look like at room temperature?
Bismuth is a soft, silvery metal with a bright, shiny surface and a yellowish or pinkish tinge. The metal breaks easily and cannot be fabricated (worked with) at room temperature.
Is bismuth Reactive or nonreactive?
Bismuth itself is not very reactive; it is sometime found in ore deposits as the native metal. But surprisingly there is little evidence that it was known to the ancients. Aristotle doesn’t list it among his seven metals and Pliny is silent on the matter.
Is polonium-214 stable?
Polonium is a chalcogen. A rare and highly radioactive metal with no stable isotopes, polonium is chemically similar to selenium and tellurium, though its metallic character resembles that of its horizontal neighbors in the periodic table: thallium, lead, and bismuth.
When the nuclide polonium-214 decays to lead 210 what kind of decay does polonium-214 undergo?
This corresponds to a alpha particle (42α). Polonium-214 undergoes d. alpha decay.
What is the daughter product for the alpha decay of bismuth 214?
In the bismuth-214 natural decay series, Bi-214 initially undergoes β decay, the resulting daughter emits an α particle, and the succeeding daughters emit a β and a β particle in that. order.
Who discovered bismuth 213?
The element bismuth was officially discovered in 1753 by Claude Geoffrey Junine, but has been utilized since the Middle Ages and in Ancient Egypt. It is a hard and brittle metallic element found in Group 15 of the periodic table.
What is bismuth 213 used for?
Further promising applications of the alpha emitters 225Actinium and its daughter nuclide 213Bismuth include the therapy of brain tumors, bladder cancer, neuroendocrine tumors, and leukemia.
What is the longest half life?
The half-life of xenon-124 — that is, the average time required for a group of xenon-124 atoms to diminish by half — is about 18 sextillion years (1.8 x 10^22 years), roughly 1 trillion times the current age of the universe. This marks the single longest half-life ever directly measured in a lab, Wittweg added.
Does bismuth oxidize in air?
Although it does not tarnish in air at ordinary temperatures, bismuth forms an oxide coating when heated and is oxidized rapidly at its boiling point of 1,560 °C. The yellow colour of this oxide distinguishes it from those formed by other metals.
Does bismuth float in water?
Elements such as silicon,germanium,gallium,arsenic and bismuth are also known to float in their own liquid forms. Any solid substance whose density is lower than that of its liquid form can float in its own liquid.
Is bismuth as heavy as lead?
Number 83 on the periodic table, it is mainly a byproduct of lead ore processing; yet among the heavy metals, it is the heaviest and the only non-toxic. …
Why is U-238 more stable than U-235?
Both U-235 and U-238 have 92 protons. But U-238 has 3 more neutrons than does U-235. The neutrons provide the nuclear force that holds the nucleus together. So the repulsive forces are the same, but the glue is stronger for U-238.
Is uranium-239 stable?
Uranium (92U) is a naturally occurring radioactive element that has no stable isotope.