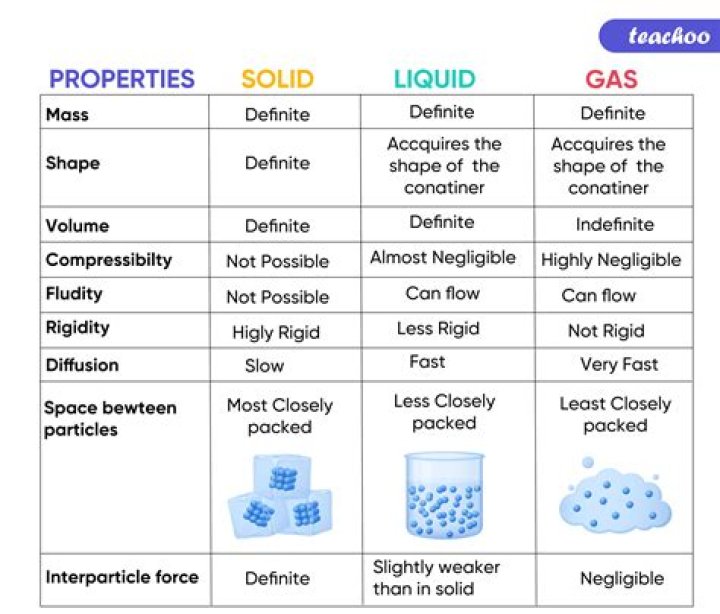
Is CsOH a solid liquid or gas
Cesium hydroxide is a colorless to yellow crystalline solid. Harmful to skin and eyes. Used in electric storage batteries. Caesium hydroxide is an alkali metal hydroxide and a caesium molecular entity.
Is CsOH dissolved in water?
Alkali metal hydroxides LiOH, NaOH, KOH, CsOH are soluble, and their solutions are basic. Hydroxides of alkali earth metals are much less soluble. For example, quicklime (CaO) reacts with water to give slaked lime, which is slightly soluble.
Is caesium a metal?
Caesium is a soft, gold-coloured metal that is quickly attacked by air and reacts explosively in water. The most common use for caesium compounds is as a drilling fluid.
Is CsOH a strong base or weak base?
LiOH lithium hydroxideNaOH sodium hydroxideKOH potassium hydroxideCa(OH)2 calcium hydroxideRbOH rubidium hydroxideSr(OH)2 strontium hydroxideCsOH cesium hydroxideBa(OH)2 barium hydroxideWhat is CsOH classified as?
Cesium hydroxide is a colorless to yellow crystalline solid. … Caesium hydroxide is an alkali metal hydroxide and a caesium molecular entity.
Why is cesium so reactive?
Cesium has a large valence electron shell and a low effective nuclear charge. The size of the valence shell affects how tightly bound the outermost electrons are to the nucleus. … Both of these factors make cesium extremely reactive.
Is strontium hydroxide a solid?
Strontium hydroxide has the molecular formula of Sr(OH)2 and the molecular weight of 121.6324 g/mol. It occurs as an anhydrate whose prismatic, colorless crystals are deliquescent. A monohydrate and octahydrate are also known. … Sr(OH)2 crystals will precipitate out as a fine white powder.
What is the common name of caoh2?
calcium compounds Calcium hydroxide, also called slaked lime, Ca(OH)2, is obtained by the action of water on calcium oxide.What is in cesium sulfate?
Caesium sulfate or cesium sulfate is the inorganic compound and salt with the formula Cs2SO4. It is a white water-soluble solid that is used to prepare dense aqueous solutions for use in isopycnic (or “density-gradient”) centrifugation. It is isostructural with potassium salt.
Is br2 a compound or element?What is Diatomic Bromine? Bromine compound is a molecule formed when two bromine atoms combine together. Bromine (Br2) is a red-brown liquid at ordinary temperature. It is very volatile.
Article first time published onIs CsOH a strong electrolyte?
Strong ElectrolyteNameCa(OH)2Calcium HydroxideSr(OH)2Strontium HydroxideRbOHRubidium HydroxideCsOHCaesium Hydroxide
Is LiH a strong base?
Lithium hydride that is LiH is basic in nature. It is a proton abstracting base. It reacts with acids like HCl. The heat of neutralisation is not for any particular base or acid.
Which is strong base NaOH or CsOH?
NaOH and CsOH are both fully ionised hydroxides so there is no difference. For a stronger base something stronger than the hydroxide is needed such as an alkoxide ion. Sodium ethoxide (Na OC2H5), for example, is a stronger base than sodium hydroxide.
Is Caesium a liquid at room temperature?
Cesium is located in row 6 of the periodic table and is also an alkali metal. Cesium’s atomic number is 55. It is liquid at room temperature and reacts explosively with cold water. Also, it is a soft and ductile element.
Is gallium and Caesium metal?
HydrogenCaesiumBariumLutetiumHafniumTantalum
How is Caesium extracted?
The ore is crushed, hand-sorted, but not usually concentrated, and then ground. Caesium is then extracted from pollucite primarily by three methods: acid digestion, alkaline decomposition, and direct reduction.
Is mnoh2 a strong base?
manganese hydroxide is a strong base.
Is cesium hydroxide soluble?
Cesium Hydroxide Hydrate is a water soluble Cesium source for use in oxygen-sensitive applications, such as metal production.
Is CsOH amphoteric?
CsOH is an alkaline hydroxide and it is strongly basic.
Is strontium hydroxide a liquid?
IdentifiersBoiling point710 °C (1,310 °F; 983 K) decomposes (anhydrous)Solubility in water0.41 g/100 mL (0 °C) 1.77 g/100 mL (40 °C) 21.83 g/100 mL (100 °C)
Is CsOH an acid or base?
Caesium hydroxide or cesium hydroxide (CsOH) is a chemical compound consisting of caesium ions and hydroxide ions. It is a strong base (pKa= 15.76), much like the other alkali metal hydroxides such as sodium hydroxide and potassium hydroxide.
Is SR aqueous or solid?
Strontium is water insoluble, but it does react with water. Strontium compounds can be water soluble.
Is caesium flammable?
FIRE HAZARDS * Cesium is a FLAMMABLE LIQUID or SOLID which MAY IGNITE SPONTANEOUSLY IN AIR.
Is fluorine a metal?
Fluorine (F) is the first element in the Halogen group (group 17) in the periodic table. … It is the most electronegative element, given that it is the top element in the Halogen Group, and therefore is very reactive. It is a nonmetal, and is one of the few elements that can form diatomic molecules (F2).
Is antimony a metal?
Antimony is a semi-metal. In its metallic form it is silvery, hard and brittle. Antimony is used in the electronics industry to make some semiconductor devices, such as infrared detectors and diodes. … A lead-antimony alloy is used in batteries.
Is cesium sulfate a gas?
Flammability (solid, gas) No data available.
What does Caesium do in water?
Caesium (cesium in USA) metal reacts rapidly with water to form a colourless basic solution of caesium hydroxide (CsOH) and hydrogen gas (H2). The reaction continues even when the solution becomes basic. The resulting solution is basic because of the dissolved hydroxide.
What does Caesium do when it reacts with water?
When caesium makes contact with water, it reacts very rapidly, and forms a colourless solution of caesium hydroxide (CsOH) and hydrogen gas (H2). This reaction is so fast, that if you tried pouring water into a test tube containing caesium (don’t do it), the glass container would shatter all over the place.
What is Chuuna?
chuna or choona, an Indian term for calcium hydroxide as used in a digestive aid.
Is Ca Oh 2 aqueous or solid?
Calcium hydroxide, commonly referred to as slaked lime, is described by the chemical formula Ca(OH)2. It is an inorganic compound which has a white, powdery appearance in its solid-state.
Is calcium a metal?
Calcium is a silvery-white, soft metal that tarnishes rapidly in air and reacts with water. Calcium metal is used as a reducing agent in preparing other metals such as thorium and uranium. It is also used as an alloying agent for aluminium, beryllium, copper, lead and magnesium alloys.