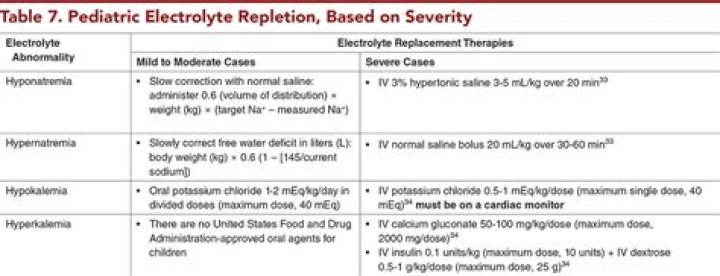
Is d5w normal saline isotonic

D5W. D5W is unique. It can be categorized as an isotonic solution or a hypotonic solution. The amount of dextrose in the solution makes its tonicity, or solute concentration, similar to that of intravascular fluid, making it isotonic.
Is dextrose normal saline isotonic?
Dextrose in water is a crystalloid solution. In the bag, it’s isotonic, but the solution itself is physiologically hypotonic.
Is 5 dextrose isotonic or hypotonic?
A solution of 5% dextrose has zero nonpenetrating solutes, and therefore, it is hypotonic.
Is 5 dextrose water isotonic?
D5W (dextrose 5% in water) is a crystalloid isotonic IV fluid with a serum osmolality of 252 mOsm/L. D5W is initially an isotonic solution and provides free water when dextrose is metabolized (making it a hypotonic solution), expanding the ECF and the ICF.Is D5W isotonic in the bag?
0.9% NaCl (Normal Saline) Lactated Ringers. D5W (In the bag)
What is D5 saline?
Dextrose and Sodium Chloride Injection, USP (dextrose and sodium chloride inj) is a sterile, nonpyrogenic solution for fluid and electrolyte replenishment and caloric supply in single dose containers for intravenous administration. It contains no antimicrobial agents.
What is the difference between normal saline and D5W?
Normal saline contain 0.9 % sodium chloride and lactated Ringer’s contain sodium chloride (6 g/L), sodium lactate (3.1 g/L), potassium chloride (0.3 g/L) and calcium chloride (0.2 g/L). Half-normal saline (1/2 NS) has half the concentration of NaCl i.e. 0.45 % NaCl. D5W composed of 5 % dextrose in water.
What is D5W IV solution?
Dextrose 5% in water is injected into a vein through an IV to replace lost fluids and provide carbohydrates to the body. Dextrose 5% in water is used to treat low blood sugar (hypoglycemia), insulin shock, or dehydration (fluid loss). … This helps your caregivers inject the medicine slowly and more safely into your body.Is D5W isotonic or hypertonic?
D5W is unique. It can be categorized as an isotonic solution or a hypotonic solution. The amount of dextrose in the solution makes its tonicity, or solute concentration, similar to that of intravascular fluid, making it isotonic.
What type of fluid is D5NS?KCL in D5NS (potassium chloride in 5% dextrose and sodium chloride injection) is a fluid and electrolyte replenishment and caloric supply used as a source of water, electrolytes and calories.
Article first time published onWhat is the difference between normal saline and dextrose saline?
Finally we would like to point out that while dextrose 5% in 0.9% normal saline is a hyperosmolar solution (560 mOsm/l) compared to the other crystalloids as well as plasma, it is nevertheless still isotonic. Once infused, the dextrose is rapidly metabolized, leaving isotonic normal saline12.
Is Plasmalyte isotonic?
Description of product. PL 148, also known as Plasma-Lyte A, is a sterile isotonic non-pyrogenic IV crystalloid solution used in clinical medicine to provide water, electrolytes and calories to patients.
Is 0.45 saline isotonic?
Sodium Chloride 0.45% Solution for Infusion is a hypotonic solution, with an approximate osmolarity of 154 mOsm/l. The pharmacodynamic properties of the solution are those of the sodium and chloride ions in maintaining the fluid and electrolyte balance.
Is D5W the same as D50?
Some examples of dextrose solutions that you might come across include: 5% Dextrose in Water (D5W) … 50% Dextrose (D50)
Which type of dextrose solution is considered isotonic?
TypeIV SolutionIsotonic5% Dextrose in Water (D5W) *starts as isotonic and then changes to hypotonic when dextrose is metabolizedHypotonic0.45% Sodium Chloride (0.45% NaCl)Hypotonic5% Dextrose in Water (D5W)Hypertonic3% Sodium Chloride (3% NaCl)
Is saline isotonic?
0.9% saline is a perfectly isotonic solution that is isoosmolar to human plasma and is not associated with hypernatremia [2].
Is D5W and D5 the same?
D5W is 5% dextrose in water is hypotonic so it moves fluid into the cells out of the circulation. D5NS is 5% dextrose in normal saline. … D5W is technically isotonic, but it becomes hypotonic once in the body so it pulls fluid out of the vasculature and into the cells.
Is glucose 10% isotonic?
Glucose 10% w/v Solution for Infusion is a hypertonic solution, with an approximate osmolarity of 555 mOsm/l.
Does normal saline contain dextrose?
Salt contentNa (mEq/L)Normal saline0.9%154D5 0.50% normal (D5 half-normal)0.45%75D5 0.33% normal (D5 third-normal)0.33%50
Does D5W have sodium?
5% dextrose in water (D5W): 0 mmol/L. 0.2% sodium chloride in 5% dextrose in water (D 5 2NS): 34 mmol/L. 0.45% sodium chloride in water (0.45NS): 77 mmol/L.
What is D5 lactated Ringer's?
5% Dextrose in Lactated Ringer’s Injection provides electrolytes and calories, and is a source of water for hydration. It is capable of inducing diuresis depending on the clinical condition of the patient. This solution also contains lactate which produces a metabolic alkalinizing effect.
What class of drug is normal saline?
What is Normal Saline and how is it used? Normal Saline is a prescription medicine used for fluid and electrolyte replenishment for intravenous administration. Normal Saline may be used alone or with other medications. Normal Saline belongs to a class of drugs called Crystalloid Fluid.
Which type of dextrose solution is considered isotonic quizlet?
D5W is classified as a ISOTONIC fluid BUT after adminstration the body metabolizes the dextrose and the fluid left over is a hypotonic solution.
Which IV fluids are hypertonic?
- 3% Saline.
- 5% Saline.
- 10% Dextrose in Water (D10W)
- 5% Dextrose in 0.9% Saline.
- 5% Dextrose in 0.45% saline.
- 5% Dextrose in Lactated Ringer’s.
Is 3 saline hypertonic?
3% and 5% Sodium Chloride Injection, USP is a sterile, nonpyrogenic, hypertonic solution for fluid and electrolyte replenishment in single dose containers for intravenous administration. The pH may have been adjusted with hydrochloric acid. It contains no antimicrobial agents.
What is dextrose saline solution?
Intravenous sugar solution, also known as dextrose solution, is a mixture of dextrose (glucose) and water. It is used to treat low blood sugar or water loss without electrolyte loss.
How do you make a D5W solution?
- Withdraw 100 mL from a 1L bag of Ringer’s Lactate.
- Add 100 mL of Dextrose 50%
- Resulting solutions 1 L of D5W-Ringer’s Lactate.
Is D5 1/2 ns isotonic or hypertonic?
Notice that sodium plays a bigger part in determining your osmolality than glucose. That’s why D5 1⁄2 NS is not an isotonic solution. As such, D5 1⁄2 NS is NOT appropriate for most medical patients who are hypovolemic. Such patients need isotonic fluids (normal saline or Lactated Ringers).
How much NaCl is in D5NS?
Each 100 mL of 5% Dextrose and 0.45% Sodium Chloride Injection, USP contains dextrose, hydrous 5 g and sodium chloride 0.45 g in water for injection. Electrolytes per 1000 mL: sodium (Na+), 77 mEq; chloride (Cl–) 77 mEq. The osmolarity is 406 mOsmol/L (calc.), which is hypertonic.
Is 0.9 Sodium Chloride normal saline?
Normal saline is 0.9% saline. This means that there is 0.9 G of salt (NaCl) per 100 ml of solution, or 9 G per liter. This solution has 154 mEq of Na per liter. In fact, all the other solutions listed on the previous screen will be compared to normal saline as if it has 150 mEq of Na/L.
What is the difference between D5 and DNS?
The main difference between 5 dextrose and dextrose saline is that 5 dextrose does not contain sodium chloride whereas dextrose saline contains sodium chloride in its chemical composition.