Is ice melt covalent or ionic
For example, water is covalently bonded and ice melts at a low temperature. However, an ionic material like salt has a lower melting point because its whole structure is composed of strong ionic bonds. There are many other differences between bonds.
Is melting points ionic or covalent?
Ionic latticeDiscrete covalent moleculesBoiling and melting pointsHighLowState at room temperatureSolidLiquid or gasConduction of electricityOnly when molten or in solutionNever
Is it covalent or ionic?
Ionic BondsCovalent BondsShapeNo definite shapeDefinite shapeMelting PointHighLowBoiling PointHighLowState at Room TemperatureSolidLiquid or Gas
Do covalent bonds form when ice melts?
water is less dense than solid water. d) Only covalent bonds are broken when ice melts. … Assuming constant temperature and pressure, the vapor pressure of the water decreases as the water evaporates.Is ice ionic solid?
Some molecular crystals, such as ice, have molecules held together by hydrogen bonds. … In all cases, the intermolecular forces holding the particles together are far weaker than either ionic or covalent bonds. As a result, the melting and boiling points of molecular crystals are much lower.
What are the melting points of ionic compounds?
Ionic compoundMelting pointBoiling pointNaCl801°C1,413°CMgO2,852°C3,600°C
Is ice a covalent solid?
Many solids are of this type. Examples are water (ice) and solid carbon dioxide (dry ice). … Covalent Network solid (Network Solid): A solid that consists of atoms held together in large networks or chains by covalent bonds. Diamond is an example of a network solid.
What happen when ice melts?
Changing states of matter and energy A block of ice is solid water. When heat (a form of energy) is added, the ice melts into liquid water. … When heat is added, the snowflake melts and changes state to become liquid water. If heat is removed from water vapour, the gas cools down and it condenses back into liquid water.Is ice covalent compound?
In the solid state (ice), intermolecular interactions lead to a highly ordered but loose structure in which each oxygen atom is surrounded by four hydrogen atoms; two of these hydrogen atoms are covalently bonded to the oxygen atom, and the two others (at longer distances) are hydrogen bonded to the oxygen atom’s …
Is ice covalent crystal?Rock salt is sodium chloride as it possesses ionic bonds, while ice is crystallized water and thus is made up of hydrogen bonding as a means of crystal formation. … These atoms are in turn bonded to two other silicon atoms and thus forms a covalent network of bonds. The compound is represented by the formula SiO2.
Article first time published onIs NH3 ionic or covalent?
NH3 has a covalent single bond among its nitrogen and hydrogen atoms. A covalent bond means the N and H atoms share valence electrons while creating…
Is Sugar ionic or covalent?
They are also very different chemically. Salt is made up of sodium and chloride and is ionically bonded. Sugar, on the other hand, is composed of carbon, oxygen, and hydrogen and has covalent bonds. A salt molecule is made up of one sodium atom and one chlorine atom.
Is Covalent stronger than ionic?
They tend to be stronger than covalent bonds due to the coulombic attraction between ions of opposite charges. … To maximize the attraction between those ions, ionic compounds form crystal lattices of alternating cations and anions.
What type of molecule is ice?
As a naturally occurring crystalline inorganic solid with an ordered structure, ice is considered to be a mineral. It possesses a regular crystalline structure based on the molecule of water, which consists of a single oxygen atom covalently bonded to two hydrogen atoms, or H–O–H.
Which type of solid is ice?
Explain why ice, which is a crystalline solid, has a melting temperature of 0 °C, whereas butter, which is an amorphous solid, softens over a range of temperatures.
Is ice a crystalline solid?
Ice has a crystalline structure stabilized by hydrogen bonding. These intermolecular forces are of comparable strength and thus require the same amount of energy to overcome. As a result, ice melts at a single temperature and not over a range of temperatures.
Can ionic compounds melt ice?
When the ionic compound salt is added to the equation, it lowers the freezing point of the water, which means the ice on the ground can’t freeze that layer of water at 32 °F anymore. The water, however, can still melt the ice at that temperature, which results in less ice on the roads.
Why ice is a crystalline compound?
Answer: Water molecules are arranged in cage like structure orderly array resulting in a crystalline structure. Crystalline compounds are made up of particles or molecules that are organized in a specific order, such as ice particles or water molecules, resulting in an ordered structure known as a crystalline compound.
What type of bond is in ice?
In ice, the crystalline lattice is dominated by a regular array of hydrogen bonds which space the water molecules farther apart than they are in liquid water. This accounts for water’s decrease in density upon freezing.
Is ice simple molecular?
Ice is a molecular lattice where the small covalent water molecules are held together by hydrogen bonds. … Low melting/boiling points and low state change enthalpies due to weak intermolecular bonding.
Why does ice melt in salt?
The actual reason that the application of salt causes ice to melt is that a solution of water and dissolved salt has a lower freezing point than pure water. … Ice in contact with salty water therefore melts, creating more liquid water, which dissolves more salt, thereby causing more ice to melt, and so on.
Is ice a hexagonal?
Ice Ih (hexagonal ice crystal) (pronounced: ice one h, also known as ice-phase-one) is the hexagonal crystal form of ordinary ice, or frozen water. … The crystal structure is characterized by the oxygen atoms forming hexagonal symmetry with near tetrahedral bonding angles.
Is NaCl ionic or covalent?
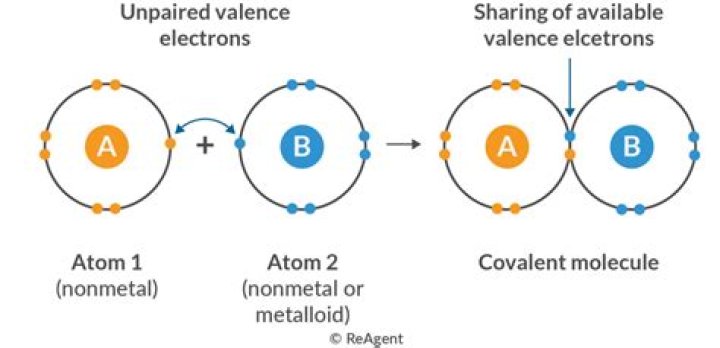
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
What is an example of a covalent compound?
Covalent compound examples include water, ammonia, chlorine gas, and nitrogen gas. Covalent compounds or molecular compounds are chemical compounds made of elements connected by covalent bonds.
Which is a covalent compound?
Covalent Compound Definition When a molecule is formed from the covalent bond, such compounds are known as a covalent compound. These compounds share one or more pair of valence electrons. The covalent compound is a result of two non-metals reacting with each other such that they are electrically neutral.
What type of melting points do covalent compounds have?
Covalent compounds usually have low melting points. An exception to this include molecules of silica and diamonds that have a high melting point. These compounds have low boiling points. This can be attributed to their weak force of attraction between the various bonded atoms.
Is high solubility ionic or covalent?
High Melting Points Because of the strong ionic bonds that hold ions together, ionic compounds have high melting points. C. Solubility and Electrical Conductivity Many ionic compounds are highly soluble.
Do ionic compounds have higher melting points than covalent?
Key Points Ionic compounds are formed from strong electrostatic interactions between ions, which result in higher melting points and electrical conductivity compared to covalent compounds.
Why does ice have a low melting point?
Ice (solid H 2 O) is a molecular compound whose molecules are held together by hydrogen bonds. Though hydrogen bonds are the strongest of the intermolecular forces, the strength of hydrogen bonds is much less than that of ionic bonds. The melting point of ice is 0°C. … For water, this equilibrium occurs at 0°C.
How are molecules arranged in ice?
Ice has a very regular pattern with the molecules rigidly apart from one another connected by the hydrogen bonds that form a crystalline lattice. These crystals have a number of open regions and pockets making ice less dense than liquid water. This is why ice floats on water.
What caused the ice to melt?
As energy is transferred to the water molecules in the ice, the motion of the molecules increases. The motion of the molecules increases enough that it overcomes the attractions the water molecules have for each other causing the ice to melt.