Is KBr a compound or molecule
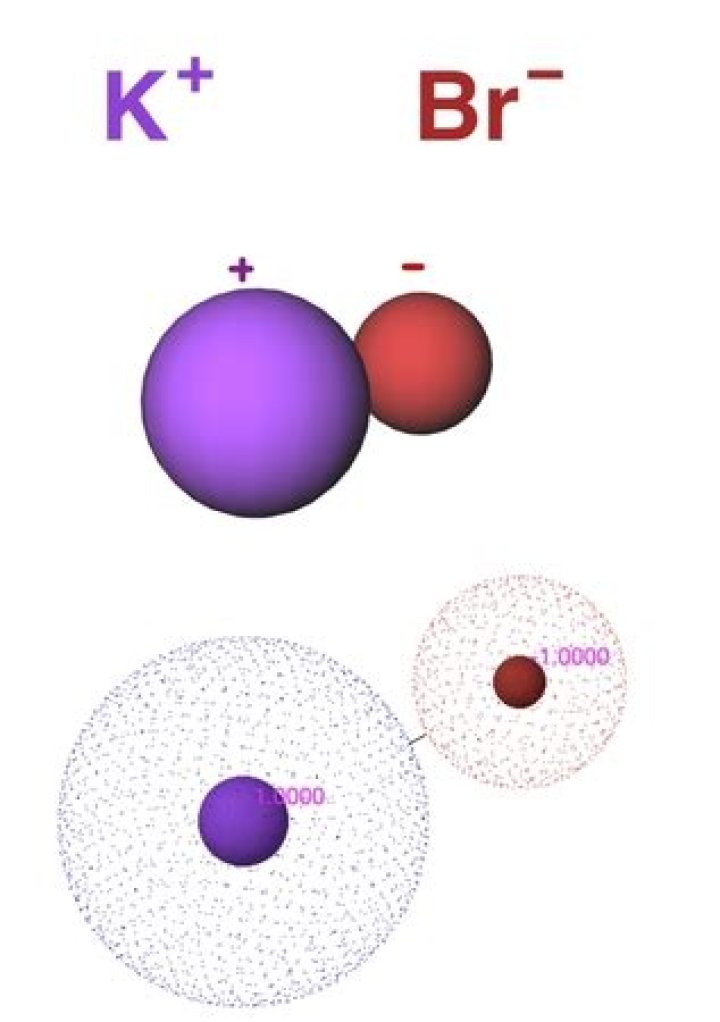
KBr is an ionic compound. Ionic compounds are also known as salts. You can identify KBr as an ionic compound because potassium is a metal and…
What type of structure is potassium bromide?
Potassium bromide is an ionic compound. It is labeled an ionic compound due to the type of bonding that occurs between the two elements – an electrostatic attraction between the two ions.
Is potassium bromide hygroscopic?
The substance is a highly water soluble inorganic salt so will partition to the aqueous environment. The solubility of potassium bromide in water is 687 g/L at 25°C. Potassium bromide is hygroscopic.
Is potassium bromide KBr ionic or covalent?
Potassium bromide has ionic bonding between its two elements potassium and bromine. Bromine has -1 charge, and potassium has a +1 charge.Is potassium bromide aqueous?
Potassium bromide, a typical ionic salt, is fully dissociated and near pH 7 in aqueous solution. It serves as a source of bromide ions.
Does potassium and bromide form precipitate?
For example, when silver nitrate and potassium bromide are mixed, a precipitate of silver bromide forms. … The complete ionic equation shows all aqueous ions as separate species, but pairs up the ions that form a solid precipitate on the products side.
Is potassium bromide inorganic?
Potassium bromide, also known as kaliumbromid or KBr, belongs to the class of inorganic compounds known as alkali metal bromides. These are inorganic compounds in which the largest halogen atom is Bromine, and the heaviest metal atom an alkali metal.
Is SO3 ionic or covalent?
ABSO3CovalentK2OIonicCH4Covalent(NH4)2CO3IonicIs potassium bromide a metal or nonmetal?
Potassium bromide is a metal bromide salt with a K(+) counterion.
Is N2O5 ionic or covalent?Binary molecular compounds are composed of two nonmetal elements, such as carbon and oxygen which form carbon dioxide, CO2 . Another molecular compound is dinitrogen pentoxide, N2O5 . We know that N2O5 could not be an ionic compound because nitrogen and oxygen are both nonmetals and would both be negative as ions.
Article first time published onIs potassium bromide an acid or base?
Name of MoleculePotassium bromidepH7
What is the correct name of KCL?
ChEBI Namepotassium chlorideChEBI IDCHEBI:32588DefinitionA metal chloride salt with a K(+) counterion.
What is the scientific name for NaBr?
Sodium bromide (NaBr)
What is the anode in potassium bromide?
Potassium bromideObservationsAnode (+)Orange-brown solution; maybe a little orange coloured gasTest used for productCathode (–)Gas ignited by burning splint; maybe a ‘squeak’Anode (+)Universal indicator paper turns red and is bleachedIdentity of products formedCathode (–)Hydrogen
Is potassium bromide a mixture?
Potassium bromine is an ionic compound with the chemical formula KBr. Potassium atoms lose one electron to become potassium ions. A bromine atom gains that electron to become bromide ion.
What Colour is aqueous potassium bromide?
Colour after shaking with hydrocarbon solventReaction with potassium bromide solutionBromine waterAqueous layer: yellow-orange to colourless Hydrocarbon layer: colourless to pale yellow-orangeNo reactionIodine solutionAqueous layer: brown to colourless Hydrocarbon layer: colourless to purpleNo reaction
What ions are in potassium bromide?
Thus, the formula of potassium bromide is given as KBr, but, when the potassium and bromide ions are shown individually, they are written K+ and Br−.
Is potassium bromide a reducing agent?
POTASSIUM BROMIDE is not in generally strongly reactive. A weak reducing agent, incompatible with oxidizing agents. Also incompatible with salts of mercury and silver.
What is the physical appearance of potassium bromide?
Compound FormulaBrKMolecular Weight119.01AppearanceWhite PowderMelting Point730 °C (1350 °F)Boiling Point1,435° C (2,615° F)
Does potassium acetate and ammonium bromide form a precipitate?
No, it does not.
When potassium and bromine atoms form chemical bonds what is produced?
Explanation: Ionic bonds result from the transfer of electrons. Potassium bromide may thus be regarded as K+Br− ; potassium bromide is thus non-molecular.
Does nabr form a precipitate?
Ammonium nitrate reacts with sodium bromide to form ammonium bromide and sodium nitrate. The reaction is a double displacement as well as a precipitation reaction. … Hence, the precipitate will form due to the combination of two soluble salts.
Is bromine a metal?
Smelly element No. 35, bromine, is a fairly abundant element but has a rare property: it is the only nonmetal to exist in liquid form at room temperature, and one of only two elements (the other being mercury) that is liquid at room temperature and pressure.
What is the name of NaI chemistry?
Sodium iodide | NaI – PubChem.
Is potassium nitrate ionic or covalent?
Potassium nitrate is an ionic compound that has the common name saltpeter.
Is potassium oxide a covalent compound?
Potassium oxide is an ionic compound formed by combining potassium and oxygen.
What type of compound is so3?
Sulfur trioxide is a sulfur oxide.
Is N2O5 a structure?
Dinitrogen pentoxide is the chemical compound with the formula N 2O 5, also known as nitrogen pentoxide or nitric anhydride. It is one of the binary nitrogen oxides, a family of compounds that only contain nitrogen and oxygen.
Is K2SO4 an ionic compound?
The bond between potassium ion (K+) and sulfate ion (SO42−) ( S O 4 2 − ) is ionic. Hence, K2SO4 K 2 S O 4 is considered as an ionic compound.
Is NaNO3 an acid or base?
Sodium nitrate is the salt that would be produced in the reaction of sodium hydroxide, a strong base, and nitric acid, a strong acid. As such, NaNO3 is neither an acid nor a base.
Is potassium hydroxide an acid or base?
Potassium hydroxide, also known as lye is an inorganic compound with the chemical formula KOH. Also commonly referred to as caustic potash, it is a potent base that is marketed in several forms including pellets, flakes, and powders. It is used in various chemical, industrial and manufacturing applications.