Is PCC a mild oxidizing agent
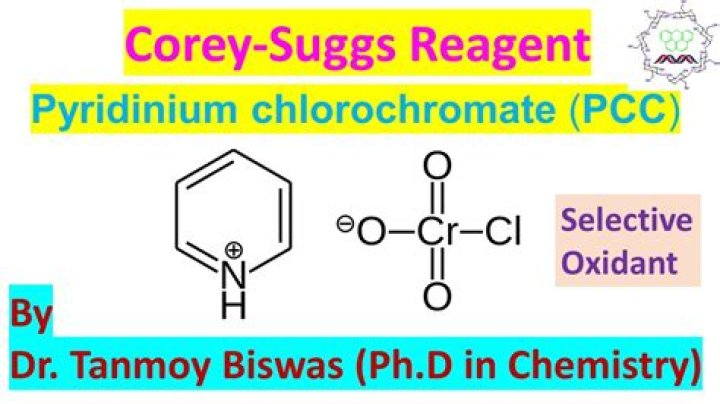
Pyridinium chlorochromate (PCC) is a milder version of chromic acid. PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. In contrast to chromic acid, PCC will not oxidize aldehydes to carboxylic acids. … Here are two examples of PCC in action.
What are the strong oxidizing agents?
Examples of the halogens that are good oxidizing agents include iodine, bromine, chlorine, and fluorine. Fluorine is said to be the strongest elemental oxidizing agent due to its highest electronegativity, as discussed earlier.
Is CrO3 a strong oxidizing agent?
Chromium trioxide is a strong oxidizing agent that is not soluble in most organic solvents and tends to explode in the presence of organic compounds and solvents. A solution of chromium trioxide in aqueous sulfuric acid can be safely mixed with acetone (Jones Reagent). …
Is PCC acidic or basic?
PCC is more acidic than PDC, but acid-labile compounds can be oxidized in the presence of sodium acetate or other buffers such as carbonates.Is PCC a weak oxidizing agent?
Pyridinium chlorochromate is a weak oxidizing agent and is often used to oxidize alcohols into carbony compounds. All of the other compounds are similar in that they function as reducing agents.
Is KMnO4 a strong oxidizing agent?
Potassium permanganate, KMnO4, is a powerful oxidizing agent, and has many uses in organic chemistry.
Can PCC oxidize ketone?
Oxidation of alcohols PCC is used as an oxidant. In particular, it has proven to be highly effective in oxidizing primary and secondary alcohols to aldehydes and ketones, respectively.
Which is strongest reducing agent?
Fluorine acts as a stronger reducing agent than oxygen.What is the weakest oxidizing agent?
H2O2 H 2 O 2 is a weakest oxidising agent because it can act as a reducing agent also.
What is PCC oxidation?Pyridinium chlorochromate (PCC) is a milder version of chromic acid. PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. … Similar to or the same as: CrO3 and pyridine (the Collins reagent) will also oxidize primary alcohols to aldehydes.
Article first time published onDoes PCC oxidize esters?
It belongs to the family of chromium-based oxidizing agents most of which are CrO3, Na2Cr2O7, and chromic acid but unlike those, it is a mild oxidizing agent. The reaction starts by converting the alcohol to its corresponding chromate ester, which then undergoes a deprotonation by a base to form a C=O.
Can PCC oxidize phenol?
Option A is nothing but phenol, which cannot be oxidized by using PCC. Option A is the correct one. Note: As PCC is a weak oxidizing agent, it cannot oxidize the primary alcohols directly to carboxylic acids but oxidizes primary alcohols to aldehydes only.
Does PCC oxidize alkenes?
As well as oxidizing (secondary alkyl)boranes to ketones,90,91 PCC oxidizes (primary alkyl)boranes to aldehydes in excellent yields (equation 47). The latter transformation cannot be accomplished with chromic acid. The reagent tolerates the presence of alkene, ester and acetal groups.
Is CrO3 a mild oxidizing agent?
CrO3 is a strong oxidising agent.
Is DMP a strong oxidizing agent?
Let’s call these “weak” oxidants. Examples of this are pyridinium chlorochromate (PCC), (DMP), the Swern oxidation [(COCl)2, DMSO, NEt3)] and CrO3/pyridine (the “ “) all shown below.
Does PCC affect double bond?
PCC oxidizes the alcohol (OH) group and does not affect any other functional group or double bond present in the compound [1-4].
Can PCC oxidize a tertiary alcohol?
A common reagent that selectively oxidizes a primary alcohol to an aldehyde (and no further) is pyridinium chlorochromate, PCC. E.g. Tertiary Alcohols These are resistant to oxidation because they have no hydrogen atoms attached to the oxygen bearing carbon (carbinol carbon).
Why does PCC oxidation stop at the aldehyde?
PCC is soluble in dichloromethane, thus it can be used under conditions that exclude water, allowing for the oxidization of primary alcohols to aldehydes and stop at that stage because the aldehyde hydrate is not present under anhydrous conditions. So the oxidation stops at the aldehyde stage.
What are the advantages of PDC over PCC?
PDC is less acidic than PCC and is therefore more suitable for the oxidation of acid-sensitive substrates. A full review of chromium-based reagents can be found in the book written by Tojo and Fernández (Oxidation of Alcohols to Aldehydes and Ketones, Springer Berlin, 2006, 1-97.).
What does Na2Cr2O7 do in a reaction?
Sodium dichromate (Na2Cr2O7) or chromium trioxide (CrO3) will oxidize a primary alcohol to the carboxylic acid. Oxidation of primary alcohols to aldehydes can be accomplished by using pyridinium chlorochromate (PCC) as the oxidizing agent. PCC is a mixture is chromium trioxide, pyridine, and HCl.
What is PCC and PDC?
Pyridinium chlorochromate (PCC) and Pyridinium dichromate (PDC) are two reagents used for the oxidation of alcohols. … -Oxidation of primary alcohols always gives aldehyde (RCHO) and secondary and tertiary alcohols gives ketone (RCOR’); the carboxylic acid is never produce.
How is KMnO4 an oxidizing agent?
KMnO4Potassium permanganateOxidation State+7
Is K2Cr2O7 a strong oxidizing agent?
Potassium dichromate is a powerful oxidizing-agent, especially in an acidic medium. This chemical compound is commonly used to oxidize alcohols.
Which is better oxidising agent KMnO4 or K2Cr2O7?
KMnO4 is slightly stronger oxidising agent than K2Cr2O7 in acidic medium due to its higher reduction potential since the compound having higher reduction potential behaves as best oxidizing agent.
What is the strongest and weakest oxidizing agent?
Explanation: Here’s a typical table of standard reduction potentials. The species at the top left have the greatest “potential” to be reduced, so they are the strongest oxidizing agents. The strongest oxidizing agent in the list is F2 , followed by H2O2 , and so on down to the weakest oxidizing agent, Li+ .
What is the strongest and weakest reducing agent?
For example, among sodium (Na), chromium (Cr), cuprous (Cu+) and chloride (Cl−), it is Na that is the strongest reducing agent while Cl− is the weakest; said differently, Na+ is the weakest oxidizing agent in this list while Cl is the strongest.
What is the weakest reducing agent?
Fluorine is at the bottom of the table has highest reduction potential. It is the weakest reducing agent or the strongest oxidizing agent with lowest ability to lose electron or highest ability to gain electron and get reduced.
How do you identify oxidizing agent and reducing agent?
Break the reaction down into a net ionic equation and then into half-reactions. The substance that loses electrons is being oxidized and is the reducing agent. The substance that gains electrons is being reduced and is the oxidizing agent.
What is an example of an oxidizing agent?
An oxidizing agent, or oxidant, gains electrons and is reduced in a chemical reaction. … Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid. A reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction.
Can PCC oxidize amines?
Oxidation with chromium(VI) amines has two primary limitations. Operationally, the tarry byproducts of chromium oxidations cause reduced yields and product sequestration. In addition, Cr(VI)-amines (particularly PCC) may react with acid-labile functionality.
What is PCC in chemistry class 12?
Hint: We know that PCC stands for pyridinium chlorochromate. Pyridinium chlorochromate is formed by the reaction of pyridine and chromium oxide, hydrochloric acid. You can now protonate the nitrogen atom present on the pyridine molecule and then perform the reaction.



