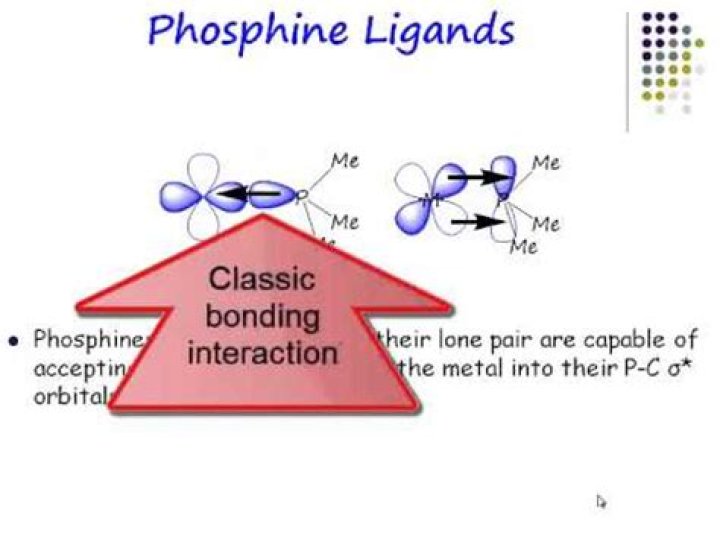
Is phosphine a neutral ligand
Lν(CO) cm−1P(OEt)32076.3PCl32097.0PF32110.8
Is phosphine an acid or base?
Phosphine is a Lewis base because there is a non-bonding pair of electrons on the P that can be donated.
What type of reagent is phosphine?
Phosphine (IUPAC name: phosphane) is a colourless, flammable, very toxic gas compound with the chemical formula PH3, classed as a pnictogen hydride.
What is the PH of phosphine?
phosphine (PH3), also called hydrogen phosphide, a colourless, flammable, extremely toxic gas with a disagreeable garliclike odour.Is phosphine polar or nonpolar?
Polarity or Nonpolarity of Phosphine or Phosphorus Trihydrate. PH3 is a polar molecule because it has a bent structure due to lone pairs of electrons and electron-electron repulsion.
Is phosphine gas lighter than air?
Phosphine is slightly (1.2 times) heavier than air so it will not move evenly through solid storage, such as a grain bin. … Magnesium phosphide also reacts faster with water to release phosphine gas but the process is faster than with aluminum phosphide.
Is phosphine a strong field ligand?
Phosphines are better spectator ligands than actor ligands. … Thus, a stronger σ−donor phosphine ligand would increase the electron density at the metal center leading to an enhanced metal to ligand π−back bonding and thereby lowering of the ν(CO) stretching frequencies in these complexes.
Is phosphine a covalent compound?
The atoms in the phosphine molecule are connected by single covalent bonds. A covalent bond is formed when atoms share electrons. … The phosphine molecule has a pyramidal shape. The hydrogen atoms form the base of the pyramid and the phosphorus atom is the apex.Is phosphine a gas?
Agent Characteristics. APPEARANCE: Colorless gas. DESCRIPTION: Phosphine is used in the semiconductor industry to introduce phosphorus into silicon crystals . It is also used as a fumigant, a polymerization initiator and as an intermediate for the preparation of several flame retardants.
How is phosphine made naturally?Phosphine is formed naturally via the anaerobic decay of phosphorus-containing organic matter. … Phosphine is made industrially from white phosphorus by hydrolysis with an alkali metal hydroxide or an aqueous acid–catalyzed disproportionation reaction.
Article first time published onIs phosphine organic or inorganic?
Phosphine, also known as [PH3] or fosfano, is a member of the class of compounds known as homogeneous other non-metal compounds. These compounds are inorganic nonmetals in which the largest atom belongs to the class of ‘other nonmetals’.
Can phosphine be produced naturally?
Phosphine (PH 3 ) is a natural gaseous carrier of phosphorus in its biogeochemical cycles, has been found ubiquitously present in the environment. … The tentative experiments show that phosphine gas could be produced by aqueous or acid corrosion from phosphorus-containing impurities in iron.
How does phosphine react with cuso4?
b) Phosphine gas is absorbed in copper sulphate solution. … Phosphine gas reacts with copper sulphate to give cupric phosphide and sulphuric acid.
How is phosphine prepared from Aluminium?
Methods to prepare phosphine: Phosphine can be obtained by the hydrolysis of metallic phosphides with water, or dilute mineral acids like HCl orH2SO4. Calcium phosphide or sodium phosphide on being hydrolysed with water produce phosphine. Aluminium phosphide is hydrolysed with hydrochloric acid, it gives phosphine.
Is phosphine an element?
Present in the chemical group called ‘organophosphorus’, Phosphine (IUPAC name: Phosphane) is an element that is a highly toxic gas with flammable properties.
Why is phosphine not planar?
PH3 has a lone pair and does not have a trigonal planar geometry–for this reason it is not symmetrical. … When one or more lone pairs are present on a central atom, the VSEPR model (correctly) predicts the geometry. If the geometry is not totally symmetrical around its center, the molecule will be polar.
What type of bonding is present in phosphine?
PH3 is a covalent polar compound. Phosphorous is bonded to three hydrogen atoms and has a lone pair of electrons. Since the electronegativity of phosphorous and hydrogen is nearly same so the covalent bond is non-polar.
Is SO2 polar or nonpolar?
Sulfur dioxide (SO2) is polar in nature. The electronegativity difference between sulfur (2.58) and oxygen (3.44) atoms makes it a polar molecule. In addition, due to the presence of unbonded electrons on the sulfur and oxygen atoms, SO2 has a bent shape.
Is ch3 a neutral ligand?
Types of ligands A few examples of this type of ligand are H, halogens (Cl, Br, F, etc.), OH, CN, CH3, and NO (bent). L-type ligands are neutral ligands that donate two electrons to the metal center regardless of the electron counting method being used.
Is phosphine a good pi acceptor?
Phosphines are two electron donors that engage a lone pair for binding to metals. These are thus considered as good σ−donors and poor π−acceptors and they belong to the same class with the aryl, dialkylamino and alkoxo ligands.
Is PPh3 a strong ligand?
Yes, as per the crystal field theory and the spectrochemical series, the PPh3 group can be categorized as a strong field ligand since it has the ability to produce strong splitting.
What happens if you breathe phosphine gas?
Acute (short-term) inhalation exposure to phosphine may cause headaches, dizziness, fatigue, drowsiness, burning substernal pain, nausea, vomiting, cough, labored breathing, chest tightness, pulmonary irritation, pulmonary edema, and tremors in humans. Convulsions may ensue after an apparent recovery.
Is phosphine heavier than air?
Phosphine gas is heavier than air and may travel along the ground to an ignition source. When phosphine is heated to decomposition, it produces highly toxic fumes of phosphorus oxides.
How does phosphine react with oxygen?
Phosphine, an extremely poisonous and highly reactive gas, will react with oxygen to form tetraphosphorus decaoxide and water. PH3(g) + O2(g) → P4O10(s) + H2O(g) Calculate the mass of P4O10(s) formed when 225 g of PH3 reacts with excess oxygen.
Is phosphine a greenhouse gas?
Phosphine is a reactive atmospheric trace gas, which competes with methane and other greenhouse gases for hydroxyl radicals that are produced by the light degradation of ozone22.
Is there phosphine gas on Venus?
The reanalysis found that phosphine concentrations in Venus’s atmosphere occasionally peak at five parts per billion. That means levels of the gas might wax and wane over time at different places on the planet, said Greaves — a situation similar to methane spikes appearing on Mars.
Where is phosphine gas found?
In the outer solar system, phosphine is made deep in the interiors of Jupiter and Saturn. Near the giant planets’ cores, the temperatures and pressures are extreme enough to craft the molecule, which then rises through the atmosphere.
Is magnesium oxide ionic or covalent?
Magnesium oxide is considered ionic. This means that the bonds that hold magnesium and oxygen together are formed between atoms that give electrons or…
Is B2H6 ionic or covalent?
The two types of bonds present in B2H6 are covalent and ionic bond.
What bond is SO2?
Sulfur dioxide (SO2) is a covalent molecule. As a result of the small variation in electronegativity between the two atoms, electron bonds are shared, producing covalent bonds.
What is phosphine made of?
Phosphine is a chemical compound made up of one atom of phosphorus and three atoms of hydrogen, and scientists have also spotted it on Earth, Jupiter and Saturn.