Is warfarin warfarin a sodium
Warfarin sodium is an anticoagulant medication. “Anti” means against and “coagulant” means causing blood clotting. Warfarin controls the way that blood clots (thickens into a lump) inside your blood vessels. The brand names of warfarin are Coumadin® and Jantoven®.
What class of medication is warfarin?
Warfarin is also used to treat or prevent venous thrombosis (swelling and blood clot in a vein) and pulmonary embolism (a blood clot in the lung). Warfarin is in a class of medications called anticoagulants (‘blood thinners’). It works by decreasing the clotting ability of the blood.
What is the makeup of warfarin?
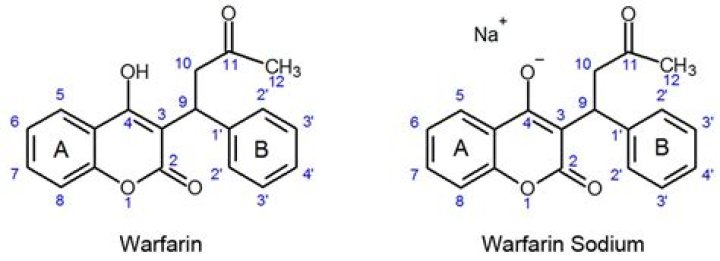
PubChem CID16204922Molecular FormulaC19H15NaO4SynonymsWarfarin sodium 129-06-6 Sodium warfarin Jantoven Marevan More…Molecular Weight330.3Parent CompoundCID 54678486 (Warfarin)
What is an acidic drug?
Abstract. Strong acidic drugs are a class of chemical compounds that normally have high hydrophilicity and large negative charges, such as organophosphatic compounds and organosulphonic compounds.Is warfarin a LMWH?
LMWH is often used temporarily to help patients transition – or bridge – to long-term oral anticoagulant therapy, most commonly warfarin or Coumadin®.
Why is warfarin being discontinued?
The most commonly reported reasons for warfarin discontinuation were physician preference (47.7%), patient refusal/preference (21.1%), bleeding event (20.2%), frequent falls/frailty (10.8%), high bleeding risk (9.8%), and patient inability to adhere to/monitor therapy (4.7%).
Is warfarin an anticoagulant?
Warfarin is an anticoagulant. It is most likely to be the drug popularly referred to as a “blood thinner,” yet this is a misnomer, since it does not affect the thickness or viscosity of blood. Instead, it acts on the liver to decrease the quantity of a few key clotting factors in blood that allow blood to clot.
Is warfarin water soluble?
Its empirical formula is C19H15NaO4, and its structural formula may be represented by the following: 1 Page 2 Crystalline warfarin sodium occurs as a white, odorless, crystalline powder, is discolored by light and is very soluble in water; freely soluble in alcohol; very slightly soluble in chloroform and in ether.Is warfarin an enzyme inducer or inhibitor?
Drug(s)/productEnzyme inhibitor or inducerDrug(s)Amiodarone (Cordarone)CYP2C9 and CYP3A4 inhibitorWarfarin (Coumadin)Carbamazepine (Tegretol), phenobarbital, phenytoin (Dilantin)CYP3A4 inducerEthinyl estradiol-containing contraceptives
Is ibuprofen acidic or basic?Ibuprofen is a relatively weak acid (pka 4.4) and its solubility in water or at acid pHis very low. This results in a relatively long residence time in the acid environment of the stomach, which slows down absorption of the substance.
Article first time published onIs aspirin basic or acidic?
Aspirin itself is an acidic medicine and causes gastric irritation and regurgitation which can lead to low oral pH levels [7].
Which drugs are weak bases?
- Diazepam (3.0)
- Lignocaine (7.9)
- Codeine (8.2)
- Cocaine (8.5)
- Adrenaline (8.7)
- Atropine (9.7)
- Amphetamine (9.8)
- Metoprolol (9.8)
What is a common side effect of warfarin?
The main side effect of warfarin is bleeding. While the risk of major bleeding is low, you need to be aware of potential problems. For example, you might have trouble stopping the bleeding from a cut on your hand or a nosebleed. More-serious bleeding may occur inside the body (internal).
Is warfarin natural or synthetic?
Warfarin, the most widely used oral anticoagulant, is a synthetic compound available as a racemic mixture of 50% R-warfarin and 50% S-warfarin.
Why do you take warfarin at 6pm?
It’s usual to take warfarin in the evening. This is so that if you need to change the dose after a routine blood test, you can do this the same day rather than waiting until the following morning.
Why does warfarin cause Hypercoagulability?
Warfarin loading doses may paradoxically result in a hypercoagulable state and potential clot formation because of significant reductions in protein C and protein S levels.
Is apixaban a LMWH?
The anticoagulant function of agents such as Apixaban and Rivaroxaban is derived from the inhibition of coagulation factor Xa, and is therefore similar to that of a number of LMWH.
What is the difference between UFH and LMWH?
Compared with UFH, the LMWH enoxaparin binds less avidly to plasma proteins, and therefore has increased bioavailability and duration of action. When coupled with antithrombin III, enoxaparin has weaker activity against thrombin, but unlike UFH, it has more potent inhibition of factor Xa.
Is warfarin a DOAC?
Vitamin K antagonists, such as warfarin, have been the anticoagulants of choice for many years for patients with AF and other thrombotic conditions. The introduction of direct oral anticoagulants (DOACs) as alternatives represents a major advance in anticoagulation.
How does warfarin inhibit vitamin K?
Warfarin competitively inhibits the vitamin K epoxide reductase complex 1 (VKORC1), which is an essential enzyme for activating the vitamin K available in the body. Through this mechanism, warfarin can deplete functional vitamin K reserves and therefore reduce the synthesis of active clotting factors.
What is the difference between aspirin and warfarin?
Aspirin prevents clotting and warfarin thins the blood, thus reducing the risk of stroke due to a clot or blockage in a cerebral artery. Unlike aspirin, warfarin requires a prescription and regular blood work to monitor clotting levels and adjust drug dosages.
What is the safest blood thinner medication?
Safer Blood-Thinning Drugs to Prevent Stroke The newer medications are Pradaxa (dabigatran), Xarelto (rivaroxaban), Eliquis (apixaban), and most recently Savaysa (edoxaban) — which work by preventing pooled blood in the heart from clotting. Unlike warfarin, the newer drugs are safer and easier for patients to use.
Do doctors still prescribe warfarin?
Warfarin is still the most prescribed anticoagulant today, but NOACs as a whole have been quickly gaining ground. With several warfarin alternatives to choose from, patients and their physicians can now compare factors like cost, side effects, and hassle to decide which one is best for them.
Is 5mg of warfarin a lot?
Warfarin induction at 5 mg daily is safe with a low risk of anticoagulant overdose: results of an audit of patients with deep vein thrombosis commencing warfarin.
Is warfarin an inducer?
Upon initiation of phenytoin, the INR may increase due to the displacement of warfarin from protein binding sites. Long-term phenytoin use with warfarin can decrease the INR since it is a CYP inducer.
Is warfarin a substrate?
Warfarin has been shown to be a moderate inhibitor of P-GP activity in human hepatocytes, which indicates that warfarin is a P-GP substrate in the liver. In theory, low P-GP activity in hepatocytes may lead to a high level of warfarin in the target cell, causing warfarin sensitivity in some people.
Is warfarin a CYP inhibitor?
The anticoagulant drug warfarin occurs as a pair of enantiomers that are differentially metabolized by human cytochromes P450 (CYP). R-warfarin is metabolized primarily by CYP1A2 to 6- and 8-hydroxywarfarin, by CYP3A4 to 10-hydroxywarfarin, and by carbonyl reductases to diastereoisomeric alcohols.
What is warfarin soluble in?
2. SUMMARY AND EVALUATION 2.1 Identity, Physical and Chemical Properties, and Analytical Methods Warfarin is a colourless, crystalline compound with a melting point of 151-161°C. It is practically insoluble in water, readily soluble in acetone and dioxane, and moderately soluble in alcohols.
Is warfarin a lipid?
Warfarin is highly lipid soluble (logP 5 3.5)3; therefore, it is possible that it could distribute into the plasma bulk lipid phase created by infusion of TPN.
Does warfarin affect PT or PTT?
Results Warfarin markedly affected APTT; for each increase of 1.0 in the international normalized ratio, the APTT increased 16 seconds (95% confidence interval, 10-22 seconds).
Is naproxen acidic or basic?
Naproxen is a propionic acid derivative and a non-steroidal anti-inflammatory drug (NSAID) with anti-inflammatory, antipyretic and analgesic activities. Naproxen inhibits the activity of the enzymes cyclo-oxygenase I and II, resulting in a decreased formation of precursors of prostaglandins and thromboxanes.



