What are fullerenes of carbon
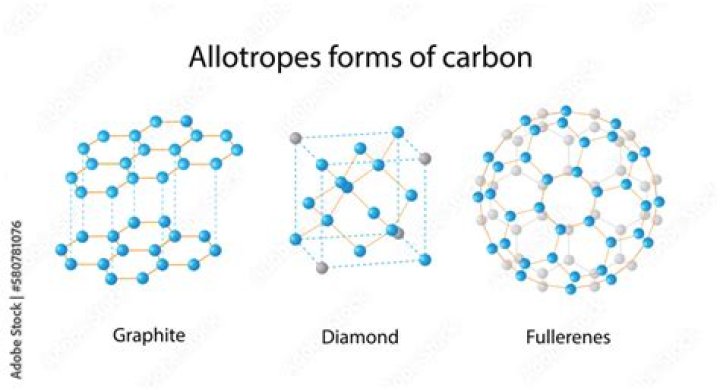
Fullerenes are molecules of carbon atoms with hollow shapes. Their structures are based on hexagonal rings of carbon atoms joined by covalent bonds. Some fullerenes include rings with five or seven carbon atoms. Two examples of fullerenes are buckminsterfullerene and nanotubes .
What is fullerene formula?
The general formula for a fullerene is C20+2n (n > 1).
How many carbons are in a buckyball?
Buckyballs are the rule-breakers of chemistry. These strange molecules are made up of 60 carbon atoms, fused together in a soccer-ball shape.
How many faces are in a fullerene?
Right: a highly asymmetric fullerene with 44 vertices and 24 faces. Despite their variety of appearances, they all have one thing in common: All fullerenes have exactly 12 pentagons!What is fullerene class 11?
Fullerene is an allotrope of carbon like graphite and diamond. They are molecules that are composed entirely of carbon, usually in the form of hollow sphere, ellipsoid or tube.
What is fullerene class 10th?
SOLUTION: Fullerene are allotropes of carbon. The fullerene C-60 (C – sixty) has carbon atoms arranged in the form of a football and it looks like a geodesic dome design by the US architect Buckminster fuller. Thus the molecule was named fullerene.
Is fullerene a allotrope?
A fullerene is an allotrope of carbon of a hollow sphere, ellipsoid, and other shapes. Carbon nanotubes are also considered members of the fullerene family. Fullerenes are closed three-dimensional allotropes of carbon.
Is fullerene crystalline form of carbon?
Therefore, the crystalline forms of carbon are diamond, graphite and fullerene. Thus, the correct option is B, that is, fullerene.How many bonds does fullerene have?
There are ninety covalent (also called molecular) bonds between them. To be more precise there are sixty single bonds and thirty double bonds.
Why is fullerene so called Class 10?This molecule got its name after the American architect Buck minster Fuller because its structure resembles the frame work of dome shaped halls designed by Fuller for large industrial exhibition.
Article first time published onHow do you make a fullerene?
Fullerenes are synthesized by the arc method from the fullerene-containing soot. By this process there are produced gram-sized quantities. In fact it is based on the Huffman Kratschmer method. There is struck an arc between two graphite electrodes in helium atmosphere (100~200 Torr).
What is a fullerene and its uses?
Fullerene is able to fit inside the hydrophobic cavity of HIV proteases, inhibiting the access of substrates to the catalytic site of enzyme. It can be used as radical scavenger and antioxidant. … In addition, fullerenes have been used as a carrier for gene and drug delivery systems.
Is graphene a fullerene?
Graphene is an allotrope of carbon that occurs as sheets of carbon while fullerene is an allotrope of carbon which occurs as spheres of carbon. The key difference between graphene and fullerene is that graphene has a two-dimensional structure, while fullerene has a three-dimensional structure.
What is fullerene used for?
Fullerene molecule can be used as an antioxidant because it can easily react with radicals due to the high affinity of the electron. At the same time, Fullerene is used as an anti-aging and anti-damage agent in the cosmetic sector. Fullerenes are used as antiviral agents.
What are Graphenes properties?
- High thermal conductivity.
- High electrical conductivity.
- High elasticity and flexibility.
- High hardness.
- High resistance. …
- Ionizing radiation is not affected.
- Able to generate electricity by exposure to sunlight.
- Transparent material.
How are fullerenes prepared Class 11?
Preparation: Fullerenes are made by heating of graphite in an electric are in the presence of inert gases such as helium or argan. … Fullerene molecule has a spherical shape and contains 60 vertices-with sp2 hybridised carbon at each vertex and are called buckyballs.
What are carbon nanotubes?
Carbon nanotubes (CNTs) are cylindrical large molecules consisting of a hexagonal arrangement of hybridized carbon atoms, which may by formed by rolling up a single sheet of graphene (single-walled carbon nanotubes, SWCNTs) or by rolling up multiple sheets of graphene (multiwalled carbon nanotubes, MWCNTs).
How many allotropes of carbon are there?
Diamond, graphite and fullerenes (substances that include nanotubes and ‘buckyballs’ , such as buckminsterfullerene) are three allotropes of pure carbon.
What allotropes are carbon?
When an element exists in more than one crystalline form, those forms are called allotropes; the two most common allotropes of carbon are diamond and graphite.
What are carbons properties?
Atomic number6Electronegativity according to Pauling2.5Density2.2 g.cm-3 at 20°CMelting point3652 °CBoiling point4827 °C
What are fullerenes examples?
Fullerenes are allotropes of carbon. For example, Buckminster fullerene is an allotrope of carbon with 60 carbon atoms arranged in the shape of a football.
Which type of hybridisation is present in carbon atom in fullerene?
As a result similar to graphite, each carbon atom in fullerene is sp2 hybridized.
Which type of hybridization is present in carbon atom in fullerene?
In fullerene, all the carbon atoms are equal and they undergo sp2-hybridisation.
How many types of fullerenes are there?
There are three important types of Fullerenes: C60, C70, and Fullerenols. This article aims to discuss these three types of Fullerenes and their uses. In 1985, Professors Kroto and Smalley were conducting experiments on the conversion of gaseous carbon into particles (soot), a phenomenon that occurs in the stars.
What are fullerenes physics?
fullerene, also called buckminsterfullerene, any of a series of hollow carbon molecules that form either a closed cage (“buckyballs”) or a cylinder (carbon “nanotubes”).
What is the shape of fullerenes?
Fullerene is a hollow sphere, and it also has other shapes. In spherical form, it is known as a buckyball. The fullerene molecule was discovered by Richard Smalley.
How many crystalline forms are there in carbon?
There are three crystalline forms of carbon: Diamond. Graphite. Fullerene.
What are the three forms of carbon?
These common forms are diamond, graphite, and buckminsterfullerene (buckyball). These three allotropes are of pure carbon.
Which is non crystalline form of carbon?
Amorphous carbon is the name used for carbon that does not have any crystalline structure.
What are fullerenes and nanotubes?
Fullerenes are forms of carbon, and include nanotubes and buckyballs . A nanotube resembles a layer of graphene, rolled into a tube shape. Nanotubes have high tensile strength , so they are strong in tension and resist being stretched.
Are fullerenes conductive?
To reiterate Ivan’s comment fullerene is a bad conductor because that’s what the measured properties produce as a result. The mechanism that makes it a bad conductor is that it has shorter range continuity than graphite. In graphite the carbon is made of sheets that can be as long as the sample.


